Abstract
Purpose
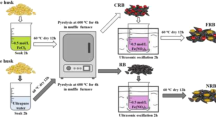
Aluminum- and iron-impregnated food waste biochar (Al-FWBC and Fe-FWBC) have been suggested for the remediation of dredged sediments contaminated with arsenic (As).
Materials and methods
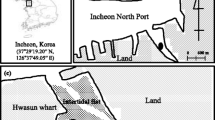
Sediment was obtained from a reservoir surrounded by several abandoned gold mines. High As concentration (102.7 mg kg−1) of the sediment corresponded to a severely contaminated level, but other heavy metals were in acceptable ranges without toxic concerns. Al-FWBC and Fe-FWBC, successfully synthesized in a previous study to remove As from aqueous solutions, were used as stabilization/solidification (S/S) agents in the current study. A sequential extraction procedure was applied to investigate the fractionation and stability of As in sediments.
Results and discussion
Amendment of Al-FWBC and Fe-FWBC for 6 months reduced the non-specifically and specifically sorbed fractions but increased the residual fraction. Fe-FWBC 1% was most efficient in stabilizing As in the sediment and it decreased 45.4% of non-specifically and specifically sorbed fractions. The effect of Al-FWBC and Fe-FWBC amendments was further confirmed by measuring potential ecological risk, and the contamination factor decreased from a considerably polluted to moderately polluted level. Arsenic and other heavy metals that leached from the S/S-treated sediments via toxicity characteristic leaching procedure did not exceed the values prescribed by the US Environmental Protection Agency.
Conclusions
Stabilization/solidification treatment using Al-FWBC and Fe-FWBC, converted from waste to value-added substances for environmental pollution control, can be applied to remediate sediments contaminated with As.


Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Agius SJ, Porebski L (2008) Towards the assessment and management of contaminated dredged materials. Integr Environ Assess Manag 4(2):255–260. https://doi.org/10.1897/IEAM_2007-058.1
Agrafioti E, Kalderis D, Diamadopoulos E (2014) Arsenic and chromium removal from water using biochars derived from rice husk, organic solid wastes and sewage sludge. J Environ Manage 133:309–314. https://doi.org/10.1016/j.jenvman.2013.12.007
Akcil A, Erust C, Ozdemiroglu S, Fonti V, Beolchini F (2015) A review of approaches and techniques used in aquatic contaminated sediments: metal removal and stabilization by chemical and biotechnological processes. J Clean Prod 86:24–36. https://doi.org/10.1016/j.jclepro.2014.08.009
Aredes S, Klein B, Pawlik M (2013) The removal of arsenic from water using natural iron oxide minerals. J Clean Prod 60:71–76. https://doi.org/10.1016/j.jclepro.2012.10.035
Bettoschi A, Marrucci A, Marras B, Atzori M, Schintu M (2018) Arsenic speciation in marine sediments: a comparison between two sequential extraction procedures. Soil Sediment Contam 27(8):723–735. https://doi.org/10.1080/15320383.2018.1512081
Bortone G, Arevalo E, Deibel I, Detzner HD, de Propris L, Elskens F, van Veen J (2004) Synthesis of the Sed Network package 4 outcomes. J Soils Sediments 4(4):225–232. https://doi.org/10.1007/BF02991117
Carabante I, Grahn M, Holmgren A, Kumpiene J, Hedlund J (2009) Adsorption of As (V) on iron oxide nanoparticle films studied by in situ ATR-FTIR spectroscopy. Colloids Surf a: Physicochem Eng Asp 346(1–3):106–113. https://doi.org/10.1016/j.colsurfa.2009.05.032
Chang Q, Lin W, Ying WC (2010) Preparation of iron-impregnated granular activated carbon for arsenic removal from drinking water. J Hazard Mater 184(1–3):515–522. https://doi.org/10.1016/j.jhazmat.2010.08.066
Ding S, Chen M, Gong M, Fan X, Qin B, Xu H, Zhang C (2018) Internal phosphorus loading from sediments causes seasonal nitrogen limitation for harmful algal blooms. Sci Total Environ 625:872–884. https://doi.org/10.1016/j.scitotenv.2017.12.348
EPA, U.S. (2013) Environmental protection agency. http://www.epa.gov
Förstner U, Apitz SE (2007) Sediment remediation: US focus on capping and monitored natural recovery. J Soils Sediments 7(6):351–358. https://doi.org/10.1065/jss2007.10.256
Frihy O, Moufaddal W, Deabes E, Helmy EED (2016) Economic evaluation of using marine dredged material for erosion control along the northeast coast of the Nile Delta. Egypt Arab J Geosci 9(14):1–19. https://doi.org/10.1007/s12517-016-2660-y
Fu H, Quan X (2006) Complexes of fulvic acid on the surface of hematite, goethite, and akaganeite: FTIR observation. Chemosphere 63(3):403–410. https://doi.org/10.1016/j.chemosphere.2005.08.054
Goldberg S, Johnston CT (2001) Mechanisms of arsenic adsorption on amorphous oxides evaluated using macroscopic measurements, vibrational spectroscopy, and surface complexation modeling. J Colloid Interface Sci 234(1):204–216. https://doi.org/10.1006/jcis.2000.7295
Guo J, Yin Z, Zhong W, Jing C (2022) Immobilization and transformation of co-existing arsenic and antimony in highly contaminated sediment by nano zero-valent iron. J Environ Sci 112:152–160. https://doi.org/10.1016/j.jes.2021.05.007
Hakanson L (1980) An ecological risk index for aquatic pollution control. A Sedimentological Approach Water Res 14(8):975–1001. https://doi.org/10.1016/0043-1354(80)90143-8
Hashimi SQ, Hong SH, Lee CG, Park SJ (2022) Adsorption of arsenic from water using aluminum-modified food waste biochar: optimization using response surface methodology. Water 14(17):2712. https://doi.org/10.3390/w14172712
Hong SH, Lyonga FN, Kang JK, Seo EJ, Lee CG, Jeong S, Hong SG, Park SJ (2020) Synthesis of Fe-impregnated biochar from food waste for Selenium (VI) removal from aqueous solution through adsorption: Process optimization and assessment. Chemosphere 126475. https://doi.org/10.1016/j.chemosphere.2020.126475
Hu Q, Liu Y, Gu X, Zhao Y (2017) Adsorption behavior and mechanism of different arsenic species on mesoporous MnFe2O4 magnetic nanoparticles. Chemosphere 181:328–336. https://doi.org/10.1016/j.chemosphere.2017.04.049
Hwang S, Her Y, Jun SM, Song JH, Le G, Kang M (2019) Characteristics of arsenic leached from sediments: agricultural implications of abandoned mines. Appl Sci 9(21):4628. https://doi.org/10.3390/app9214628
International Biochar Initiative (2012) Standardized product definition and product testing guidelines for biochar that is used in soil. IBI Biochar Standards
Jamali MK, Kazi TG, Afridi HI, Arain MB, Jalbani N, Memon AR (2007) Speciation of heavy metals in untreated domestic wastewater sludge by time saving BCR sequential extraction method. J Environ Sci Health A 42(5):649–659. https://doi.org/10.1080/10934520701244433
Jeffery S, Bezemer TM, Cornelissen G, Kuyper TW, Lehmann J, Mommer L, Sohi SP, van de Voorde TFJ, Wardle DA, van Groenigen JW (2015) The way forward in biochar research: targeting trade-offs between the potential wins. Gcb Bioenergy 7(1):1–13. https://doi.org/10.1111/gcbb.12132
Jeong Y, Fan M, Singh S, Chuang CL, Saha B, Van Leeuwen JH (2007) Evaluation of iron oxide and aluminum oxide as potential arsenic (V) adsorbents. Chem Eng Process Intensif 46(10):1030–1039. https://doi.org/10.1016/j.cep.2007.05.004
Jiang J, Dai Z, Sun R, Zhao Z, Dong Y, Hong Z, Xu R (2017) Evaluation of ferrolysis in arsenate adsorption on the paddy soil derived from an Oxisol. Chemosphere 179:232–241. https://doi.org/10.1016/j.chemosphere.2017.03.115
Kang JK, Seo EJ, Lee CG, Jeong S, Park SJ (2022) Application of response surface methodology and artificial neural network for the preparation of Fe-loaded biochar for enhanced Cr(VI) adsorption and its physicochemical properties and Cr(VI) adsorption characteristics. Environ Sci Pollut Res 29:60852–60866. https://doi.org/10.1007/s11356-022-20009-3
Kang JK, Seo EJ, Lee CG, Park SJ (2021) Fe-loaded biochar obtained from food waste for enhanced phosphate adsorption and its adsorption mechanism study via spectroscopic and experimental approach. J Environ Chem Eng 9(4):105751. https://doi.org/10.1016/j.jece.2021.105751
Kim JY, Kim KW, Lee JU, Lee JS, Cook J (2002) Assessment of As and heavy metal contamination in the vicinity of Duckum Au-Ag mine. Korea Environ Geochem Health 24(3):213–225. https://doi.org/10.1023/A:1016096017050
Lee CG, Hong SH, Hong SG, Choi JW, Park SJ (2019) Production of biochar from food waste and its application for phenol removal from aqueous solution. Water Air Soil Pollut 230(3):1–13. https://doi.org/10.1007/s11270-019-4125-x
Lee SH, Kim EY, Park H, Yun J, Kim JG (2011) In situ stabilization of arsenic and metal-contaminated agricultural soil using industrial by-products. Geoderma 161(1–2):1–7. https://doi.org/10.1016/j.geoderma.2010.11.008
LeMonte JJ, Stuckey JW, Sanchez JZ, Tappero R, Rinklebe J, Sparks DL (2017) Sea level rise induced arsenic release from historically contaminated coastal soils. Environ Sci Technol 51(11):5913–5922. https://doi.org/10.1021/acs.est.6b06152
Li H, Shi WY, Shao HB, Shao MA (2009) The remediation of the lead-polluted garden soil by natural zeolite. J Hazard Mater 169(1–3):1106–1111. https://doi.org/10.1016/j.jhazmat.2009.04.067
Li R, Li Q, Gao S, Shang JK (2012) Exceptional arsenic adsorption performance of hydrous cerium oxide nanoparticles: Part A. Adsorption capacity and mechanism. Chem Eng J 185–186:127–135. https://doi.org/10.1016/j.cej.2012.01.061
Li XD, Poon CS, Sun H, Lo IMC, Kirk DW (2001) Heavy metal speciation and leaching behaviors in cement based solidified/stabilized waste materials. J Hazard Mater 82(3):215–230. https://doi.org/10.1016/S0304-3894(00)00360-5
Li X, Qin Y, Jia Y, Li Y, Zhao Y, Pan Y, Sun J (2021) Preparation and application of Fe/biochar (Fe-BC) catalysts in wastewater treatment: a review. Chemosphere 274:129766. https://doi.org/10.1016/j.chemosphere.2021.129766
Liu T, Wu K, Xue W, Ma C (2015) Characteristics and mechanisms of arsenate adsorption onto manganese oxide-doped aluminum oxide. Environ Prog Sustain 34(4):1009–1018. https://doi.org/10.1002/ep.12091
Lyonga FN, Hong SH, Cho EJ, Kang JK, Lee CG, Park SJ (2021) As (III) adsorption onto Fe-impregnated food waste biochar: experimental investigation, modeling, and optimization using response surface methodology. Environ Geochem Health 43:3303–3321. https://doi.org/10.1007/s10653-020-00739-4
Ma X, Li C, Yang L, Ding S, Zhang M, Zhang Y, Zhao T (2020) Evaluating the mobility and labile of As and Sb using diffusive gradients in thin-films (DGT) in the sediments of Nansi Lake, China. Sci Total Environ 713:136569. https://doi.org/10.1016/j.scitotenv.2020.136569
MacDonald DD, Ingersoll CG, Berger TA (2000) Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch Environ Contam Toxicol 39(1):20–31. https://doi.org/10.1007/s002440010075
Mamindy-Pajany Y, Hurel C, Geret F, Roméo M, Marmier N (2013) Comparison of mineral-based amendments for ex-situ stabilization of trace elements (As, Cd, Cu, Mo, Ni, Zn) in marine dredged sediments: a pilot-scale experiment. J Hazard Mater 252:213–219. https://doi.org/10.1016/j.jhazmat.2013.03.001
Mamindy-Pajany Y, Hurel C, Marmier N, Roméo M (2011) Arsenic (V) adsorption from aqueous solution onto goethite, hematite, magnetite and zero-valent iron: effects of pH, concentration and reversibility. Desalination 281:93–99. https://doi.org/10.1016/j.desal.2011.07.046
Martin M, Violante A, Ajmone-Marsan F, Barberis E (2014) Surface interactions of arsenite and arsenate on soil colloids. Soil Sci Soc Am J 78(1):157–170. https://doi.org/10.2136/sssaj2007.0295
Meilani V, Lee JI, Kang JK, Lee CG, Jeong S, Park SJ (2021) Application of aluminum-modified food waste biochar as adsorbent of fluoride in aqueous solutions and optimization of production using response surface methodology. Microporous Mesoporous Mater 312:110764. https://doi.org/10.1016/j.micromeso.2020.110764
National Institute of Environmental Research (NIER) (2011) Baseline concentrations of heavy metals in river sediments in Korea, Korea Ministry of Environment
Ordonez-Loza J, Chejne F, Jameel AGA, Telalovic S, Arrieta AA, Sarathy SM (2021) An investigation into the pyrolysis and oxidation of bio-oil from sugarcane bagasse: kinetics and evolved gases using TGA-FTIR. J Environ Chem Eng 9(5):106144. https://doi.org/10.1016/j.jece.2021.106144
Rađenović D, Kerkez Đ, Pilipović DT, Dubovina M, Grba N, Krčmar D, Dalmacija B (2019) Long-term application of stabilization/solidification technique on highly contaminated sediments with environment risk assessment. Sci Total Environ 684:186–195. https://doi.org/10.1016/j.scitotenv.2019.05.351
Ren J, Li N, Li L, An JK, Zhao L, Ren NQ (2015) Granulation and ferric oxides loading enable biochar derived from cotton stalk to remove phosphate from water. Bioresour Technol 178:119–125. https://doi.org/10.1016/j.biortech.2014.09.071
Rijkenberg MJ, Depree CV (2010) Heavy metal stabilization in contaminated road-derived sediments. Sci Total Environ 408(5):1212–1220. https://doi.org/10.1016/j.scitotenv.2009.11.053
Rulkens W (2005) Introduction to the treatment of polluted sediments. Rev Environ Sci Bio/technol 4(3):213–221. https://doi.org/10.1007/s11157-005-2167-6
Siham K, Fabrice B, Edine AN, Patrick D (2008) Marine dredged sediments as new materials resource for road construction. Waste Manag 28(5):919–928. https://doi.org/10.1016/j.wasman.2007.03.027
Simon G, Huang H, Penner-Hahn JE, Kesler SE, Kao LS (1999) Oxidation state of gold and arsenic in gold-bearing arsenian pyrite. Am Mineral 84(7–8):1071–1079. https://doi.org/10.2138/am-1999-7-809
Taneez M, Marmier N, Hurel C (2016) Use of neutralized industrial residue to stabilize trace elements (Cu, Cd, Zn, As, Mo, and Cr) in marine dredged sediment from South-East of France. Chemosphere 150:116–122. https://doi.org/10.1016/j.chemosphere.2016.02.014
Thomas GW (1996) Soil pH and soil acidity. Methods Soil AnalPart 3 Chem Methods 5:475–490. https://doi.org/10.2136/sssabookser5.3.c16
Thommes M, Kaneko K, Neimark AV, Olivier JP, Rodriguez-Reinoso F, Rouquerol J, Sing KS (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87(9–10):1051–1069. https://doi.org/10.1515/pac-2014-1117
Tomasevic DD, Dalmacija MB, Prica MD, Dalmacija BD, Kerkez DV, Bečelić-Tomin MR, Roncevic SD (2013) Use of fly ash for remediation of metals polluted sediment–green remediation. Chemosphere 92(11):1490–1497. https://doi.org/10.1016/j.chemosphere.2013.03.063
Torasso N, Vergara-Rubio A, Rivas-Rojas P, Huck-Iriart C, Larrañaga A, Fernández-Cirelli A, Cerveny S Goyanes S (2021) Enhancing arsenic adsorption via excellent dispersion of iron oxide nanoparticles inside poly (vinyl alcohol) nanofibers. J Environ Chem Eng 9(1):104664. https://doi.org/10.1016/j.jece.2020.104664
US Environmental Protection Agency (1986) Test methods for evaluating solid waste. Laboratory Manual Physical/Chemical Methods, vol. 1A. US Govt. Print. Office, Washington, DC (SW-846)
USEPA (2002) Toxicity characterization leaching procedure (TCLP) EPA method 1311
Wang L, Chen L, Tsang DC, Li JS, Baek K, Hou D, Poon CS (2018a) Recycling dredged sediment into fill materials, partition blocks, and paving blocks: technical and economic assessment. J Clean Prod 199:69–76. https://doi.org/10.1016/j.jclepro.2018.07.165
Wang L, Chen L, Tsang DC, Zhou Y, Rinklebe J, Song H, Ok YS (2019) Mechanistic insights into red mud, blast furnace slag, or metakaolin-assisted stabilization/solidification of arsenic-contaminated sediment. Environ Int 133:105247. https://doi.org/10.1016/j.envint.2019.105247
Wang M, Zhu Y, Cheng L, Andserson B, Zhao X, Wang D, Ding A (2018b) Review on utilization of biochar for metal-contaminated soil and sediment remediation. J Environ Sci 63:156–173. https://doi.org/10.1016/j.jes.2017.08.004
Wen J, Yi Y, Zeng G (2016) Effects of modified zeolite on the removal and stabilization of heavy metals in contaminated lake sediment using BCR sequential extraction. J Environ Manage 178:63–69. https://doi.org/10.1016/j.jenvman.2016.04.046
Wenzel WW, Kirchbaumer N, Prohaska T, Stingeder G, Lombi E, Adriano DC (2001) Arsenic fractionation in soils using an improved sequential extraction procedure. Anal Chim Acta 436(2):309–323. https://doi.org/10.1016/S0003-2670(01)00924-2
Wildt C, Gibert O, Cortina JL, Coscera G (2004) On-site remediation of chromium-contaminated sediments by combination of sediment washing and stabilization with magnesium oxide/limestone mixtures. J Soils Sediments 4(3):184–191. https://doi.org/10.1007/BF02991139
Woo NC, Choi MJ (2001) Arsenic and metal contamination of water resources from mining wastes in Korea. Environ Geology 40(3):305–311. https://doi.org/10.1007/s002540000161
World Health Organization (2008) Guidelines for drinking-water quality: second addendum. Recommendations 1
Wu K, Liu T, Xue W, Wang X (2012) Arsenic (III) oxidation/adsorption behaviors on a new bimetal adsorbent of Mn-oxide-doped Al oxide. Chem Eng J 192:343–349. https://doi.org/10.1016/j.cej.2012.03.058
Yang Y, Ye S, Zhang C, Zeng G, Tan X, Song B, Zhang P, Yang H, Li M, Chen Q (2021) Application of biochar for the remediation of polluted sediments. J Hazard Mater 404:124052. https://doi.org/10.1016/j.jhazmat.2020.124052
Zeng H, Fisher B, Giammar DE (2008) Individual and competitive adsorption of arsenate and phosphate to a high-surface-area iron oxide-based sorbent. Environ Sci Technol 42(1):147–152. https://doi.org/10.1021/es071553d
Zhang S, Li XY, Chen JP (2010) Preparation and evaluation of a magnetite-doped activated carbon fiber for enhanced arsenic removal. Carbon 48(1):60–67. https://doi.org/10.1016/j.carbon.2009.08.030
Zhang WL, Zhao LY, Yuan ZJ, Li DQ, Morrison L (2021) Assessment of the long-term leaching characteristics of cement-slag stabilized/solidified contaminated sediment. Chemosphere 267:128926. https://doi.org/10.1016/j.chemosphere.2020.128926
Zhu J, Pigna M, Cozzolino V, Caporale AG, Violante A (2013) Higher sorption of arsenate versus arsenite on amorphous Al-oxide, effect of ligands. Environ Chem Lett 11(3):289–294. https://doi.org/10.1007/s10311-013-0405-7
Funding
This work was supported by Korea Environment Industry & Technology Institute (KEITI) through Aquatic Ecosystem Conservation Research Program, funded by Korea Ministry of Environment (grant number: RE202201970).
Author information
Authors and Affiliations
Contributions
Seung-Hee-Hong: experiment, data analysis, writing—original draft; Soonho Hwang: conceptualization; Chang-Gu Lee: writing—review and editing; Seong-Jik Park: conceptualization, writing—review and editing, supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Research involving human participants and/or animals
Not applicable.
Additional information
Responsible editor: Jos Brils
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hong, SH., Hwang, S., Lee, CG. et al. Stabilization of As-contaminated dredged sediment using Al- and Fe-impregnated food waste biochar. J Soils Sediments 23, 2628–2640 (2023). https://doi.org/10.1007/s11368-023-03520-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-023-03520-z