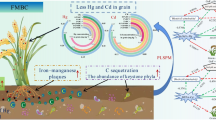
Abstract
Purpose
Diphenylarsinic acid (DPAA) tends to mobilize in flooded soil due to iron (Fe) reduction and further thionate under sulfate-reducing conditions. Fe and sulfate reduction are natural processes in paddy soil, but their coupled effects on the behavior of DPAA remain unclear. This study investigated the coupled impacts of Fe and sulfate reduction on the mobilization, thionation, and sequestration of DPAA in a paddy soil.
Methods
Four treatments were prepared in the soil incubation experiment: (1) treatment S−C− was created without the addition of S or C sources; (2) treatment S+C- was only amended with 426 μg S g−1; (3) treatment S+C+ was amended with 426 μg S g−1 and 1300 μg C g−1; and (4) treatment S+C++ was amended with 426 μg S g−1 and 2170 μg C g−1.
Results
DPAA was significantly mobilized in treatments S−C− and S+C− due to Fe reduction. Treatments S−C− and S+C− exhibited a more rapid and final enhanced DPAA transformation than did treatments S+C+ and S+C++, with the final transformation rate reaching > 80% and < 15%, respectively, for the former and the latter two treatments, and the major products of this transformation being diphenylthioarsinic acid (DPTAA) and phenylarsonic acid (PAA). It appears that DPAA thionation was favored when sulfide was supplied at a low concentration but consistently, under conditions where no additional C source was added. However, when both Fe and sulfate reduction occurred prominently and simultaneously, Fe(II) produced consumed a significant amount of sulfide to form FeS, which subsequently reduced DPAA thionation under conditions where an additional C source was added. The produced DPTAA (> 95%) was mainly associated with the soil solid phase, and sulfate reduction appears to promote DPAA sequestration on the one hand by enhancing DPAA thionation and hence its sequestration, and on the other hand by producing FeS that sorbs DPAA.
Conclusion
Our results demonstrate the coupled effects of Fe and sulfate reduction on the mobilization, thionation, and sequestration of DPAA, and that sulfate addition may be a promising strategy to reduce DPAA contamination in Fe-rich paddy soil.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Arao T, Maejima Y, Baba K (2009) Uptake of aromatic arsenicals from soil contaminated with diphenylarsinic acid by rice. Environ Sci Technol 43:1097–1101
Burton ED, Bush RT, Sullivan LA, Mitchell DRG (2007) Reductive transformation of iron and sulfur in schwertmannite-rich accumulations associated with acidified coastal lowlands. Geochim Cosmochim Acta 71:4456–4473
Cai XL, ThomasArrigo LK, Fang X, Bouchet S, Cui YS, Kretzschmar R (2020) Impact of organic matter on microbially-mediated reduction and mobilization of arsenic and iron in arsenic(V)-bearing ferrihydrite. Environ Sci Technol 55:1319–1328
Chen C, Li LY, Huang K, Zhang J, Xie WY, Lu YH, Dong XZ, Zhao FJ (2019) Sulfate-reducing bacteria and methanogens are involved in arsenic methylation and demethylation in paddy soils. ISME J 13:2523–2535
Chen XM, Zeng XC, Kawa YK, Wu WW, Zhu XB, Ullah Z, Wang YX (2020) Microbial reactions and environmental factors affecting the dissolution and release of arsenic in the severely contaminated soils under anaerobic or aerobic conditions. Ecotox Environ Safe 189:109946
Cord-Ruwisch R (1985) A quick method for the determination of dissolved and precipitated sulfides in cultures of sulfate-reducing bacteria. J Microbiol Meth 4:33–36
Couture RM, Wallschläger D, Rose J, Van Cappellen P (2013a) Arsenic binding to organic and inorganic sulfur species during microbial sulfate reduction: a sediment flow-through reactor experiment. Environ Chem 10:285–294
Couture RM, Rose J, Kumar N, Mitchell K, Wallschlag̈er D, Van Cappellen P (2013) Sorption of arsenite, arsenate, and thioarsenates to iron oxides and iron sulfides: a kinetic and spectroscopic investigation. Environ Sci Technol 47:5652–5659
Daus B, Hempel M, Wennrich R, Weiss H (2010) Concentration and speciation of arsenic in groundwater polluted by warfare agents. Environ Pollut 158:3439–3444
Deng YM, Zheng TL, Wang YX, Liu L, Jiang HC, Ma T (2018) Effect of microbially mediated iron mineral transformation on temporal variation of arsenic in the Pleistocene aquifers of the central Yangtze River basin. Sci Total Environ 619:1247–1258
FAO-IUSS-ISRIC (2014) World Soil Resources Reports No. 106 World Reference Base for Soil Resources. Food and Agriculture Organization of the United Nations (FAO), Rome
Han YS, Park JH, Kim SJ, Jeong HY, Ahn JS (2019) Redox transformation of soil minerals and arsenic in arsenic-contaminated soil under cycling redox conditions. J Hazard Mater 378:120745
Handley KM, McBeth JM, Charnock JM, Vaughan DJ, Wincott PL, Polya DA, Lloyd JR (2013) Effect of iron redox transformations on arsenic solid-phase associations in an arsenic-rich, ferruginous hydrothermal sediment. Geochim Cosmochim Acta 102:124–142
Hempel M, Daus B, Vogt C, Weiss H (2009) Natural attenuation potential of phenylarsenicals in anoxic groundwaters. Environ Sci Technol 43:6989–6995
Herbel M, Fendorf S (2006) Biogeochemical processes controlling the speciation and transport of arsenic within iron coated sands. Chem Geol 228:16–32
Hisatomi S, Guan L, Nakajima M, Fujii K, Nonaka M, Harada N (2013) Formation of diphenylthioarsinic acid from diphenylarsinic acid under anaerobic sulfate-reducing soil conditions. J Hazard Mater 262:25–30
Hultman J, Waldrop MP, Mackelprang R, David MM, McFarland J, Blazewicz SJ, Harden J, Turetsky MR, McGuire AD, Shah MB, VerBerkmoes NC, Lee LH, Mavrommatis K, Jansson JK (2015) Multi-omics of permafrost, active layer and thermokarst bog soil microbiomes. Nature 521:208–212
Ishii K, Tamaoka A, Otsuka F, Iwasaki N, Shin K, Matsui A, Endo G, Kumagai Y, Ishii T, Shoji S, Ogata T, Ishizaki M, Doi M, Shimojo N (2004) Diphenylarsinic acid poisoning from chemical weapons in Kamisu, Japan. Ann Neurol 56:741–745
Jakobsen R, Postma D (1999) Redox zoning, rates of sulfate reduction and interactions with Fe-reduction and methanogenesis in a shallow sandy aquifer, Rømø, Denmark. Geochim Cosmochim Acta 63:137–151
Kappler A, Benz M, Schink B, Brune A (2004) Electron shuttling via humic acids in microbial iron(III) reduction in a freshwater sediment. FEMS Microbiol Ecol 47:85–92
Kerl CF, Ballaran TB, Planer-Friedrich B (2019) Iron plaque at rice roots: no barrier for methylated thioarsenates. Environ Sci Technol 53:13666–13674
Kinoshita K, Shida Y, Sakuma C, Ishizaki M, Kiso K, Shikino O, Ito H, Morita M, Ochi T, Kaise T (2005) Determination of diphenylarsinic acid and phenylarsonic acid, the degradation products of organoarsenic chemical warfare agents, in well water by HPLC-ICP-MS. Appl Organomet Chem 19:287–293
Kirk MF, Roden EE, Crossey LJ, Brealey AJ, Spilde MN (2010) Experimental analysis of arsenic precipitation during microbial sulfate and iron reduction in model aquifer sediment reactors. Geochim Cosmochim Acta 74:2538–2555
Kumar N, Noel̈ V, Planer-Friedrich B, Besold J, Lezama-Pacheco J, Bargar JR, Brown GE, Fendorf S, Boye K (2020) Redox heterogeneities promote thioarsenate formation and release into groundwater from low arsenic sediments. Environ Sci Technol 54:3237–3244
Lefroy RDB, Samosir SSR, Blair GJ (1993) The dynamics of sulfur, phosphorus and iron in flooded soils as affected by changes in Eh and pH. Aust J Soil Res 31:493–508
Li LN, Jia R, Qu Z, Li TL, Shen WS, Qu D (2020) Coupling between nitrogen-fixing and iron(III)-reducing bacteria as revealed by the metabolically active bacterial community in flooded paddy soils amended with glucose. Sci Total Environ 716:137056
Lin B, Hyacinthe C, Bonneville S, Braster M, Van Cappellen P, Roling WFM (2007) Phylogenetic and physiological diversity of dissimilatory ferric iron reducers in sediments of the polluted Scheldt estuary, Northwest Europe. Environ Microbiol 9:1956–1968
Liu CX, Kota S, Zachara JM, Fredrickson JM, Brinkman AK (2001) Kinetic analysis of the bacterial reduction of goethite. Environ Sci Technol 35:2482–2490
Lu RK (2000) Methods of soil and agrochemical analysis. China Agricultural Science and Technology Press, Beijing
Maejima Y, Arao T, Baba K (2011a) Transformation of diphenylarsinic acid in agricultural soils. J Environ Qual 40:76–82
Maejima Y, Murano H, Iwafune T, Arao T, Baba K (2011b) Adsorption and mobility of aromatic arsenicals in Japanese agricultural soils. Soil Sci Plant Nutr 57:429–435
Nawała J, Gordon D, Dziedzic D, Rodziewicz P, Popiel S (2021) Why does Clark I remain in the marine environment for a long time? Sci Total Environ 774:145675
Niemikoski H, Lehtonen KK, Ahvo A, Heiskanen I, Vanninen P (2021) Metabolism and cytotoxicity of diphenylarsinic acid, a degradation product of sea-dumped chemical warfare agents, in a rainbow trout liver cell line RTL-W1. Aquat Toxicol 241:105993
Noel̈ V, Boye K, Kukkadapu RK, Li Q, Bargar JR (2019) Uranium storage mechanisms in wet-dry redox cycled sediments. Water Res 152:251–263
O’Day PA, Vlassopoulos D, Root R, Rivera N (2004) The influence of sulfur and iron on dissolved arsenic concentrations in the shallow subsurface under changing redox conditions. Proc Natl Acad Sci USA 101:13703–13708
Planer-Friedrich B, London J, McCleskey RB, Nordstrom DK, Wallschläger D (2007) Thioarsenates in geothermal waters of Yellowstone National Park: determination, preservation, and geochemical importance. Environ Sci Technol 41:5245–5251
Pollock J, Weber KA, Lack J, Achenbach LA, Mormile MR, Coates JD (2007) Alkaline iron(III) reduction by a novel alkaliphilic, halotolerant, Bacillus sp isolated from salt flat sediments of Soap Lake. Appl Microbiol Biot 77:927–934
Poulton SW, Krom MD, Raiswell R (2004) A revised scheme for the reactivity of iron (oxyhydr)oxide minerals towards dissolved sulfide. Geochim Cosmochim Acta 68:3703–3715
Ravenschlag K, Sahm K, Pernthaler J, Amann R (1999) High bacterial diversity in permanently cold marine sediments. Appl Environ Microb 65:3982–3989
Saalfield SL, Bostick BC (2009) Changes in iron, sulfur, and arsenic speciation associated with bacterial sulfate reduction in ferrihydrite-rich systems. Environ Sci Technol 43:8787–8793
Savage KE, Tingle TN, O’Day PA, Waychunas GA, Bird DK (2000) Arsenic speciation in pyrite and secondary weathering phases, Mother Lode gold district, Tuolumne County, California. Appl Geochem 15:1219–1244
Schwertmann U, Cornell RM (2000) The iron oxides in the laboratory: preparation and characterization. Wiley-VCH, Weinheim
Shi WT, Song WJ, Zheng JL, Luo Y, Qile G, Lü SJ, Lü XM, Zhou B, Lü CW, He J (2021) Factors and pathways regulating the release and transformation of arsenic mediated by reduction processes of dissimilated iron and sulfate. Sci Total Environ 768:144697
Suess E, Planer-Friedrich B (2012) Thioarsenate formation upon dissolution of orpiment and arsenopyrite. Chemosphere 89:1390–1398
Vega AS, Planer-Friedrich B, Pastén PA (2017) Arsenite and arsenate immobilization by preformed and concurrently formed disordered mackinawite (FeS). Chem Geol 475:62–75
Wakeman KD, Erving L, Riekkola-Vanhanen ML, Puhakka JA (2010) Silage supports sulfate reduction in the treatment of metals- and sulfate-containing waste waters. Water Res 44:4932–4939
Wang HY, Byrne JM, Perez JPH, Thomas AN, Göttlichere J, Höferf HE, Mayanna S, Kontny A, Kappler A, Guo HM, Benning LG, Norra S (2020a) Arsenic sequestration in pyrite and greigite in the buried peat of As-contaminated aquifers. Geochim Cosmochim Acta 284:107–119
Wang N, Xue XM, Juhasz AL, Chang ZZ, Li HB (2016) Biochar increases arsenic release from an aerobic paddy soil due to enhanced microbial reduction of iron and arsenic. Environ Pollut 220:514–522
Wang JJ, Halder D, Wegner L, Brüggenwirth L, Schaller J, Martin M, Said-Pullicino D, Romani M, Planer-Friedrich B (2020b) Redox dependence of thioarsenates occurrence in paddy soil and the rice rhizosphere. Environ Sci Technol 54:3940–3950
Weber F, Hofacker AF, Voegelin A, Kretzschmar R (2010) Temperature dependence and coupling of iron and arsenic reduction and release during flooding of a contaminated soil. Environ Sci Technol 44:116–122
Wilkin RT, Ford RG (2006) Arsenic solid-phase partitioning in reducing sediments of a contaminated wetland. Chem Geol 228:156–174
Xia D, Yi XY, Lu Y, Huang WL, Xie YY, Ye H, Dang Z, Tao XQ, Li L, Lu GL (2019) Dissimilatory iron and sulfate reduction by native microbial communities using lactate and citrate as carbon sources and electron donors. Ecotox Environ Safe 174:524–531
Xu LY, Wu X, Wang SF, Yuan ZD, Xiao F, Ming Y, Jia YF (2016) Speciation change and redistribution of arsenic in soil under anaerobic microbial activities. J Hazard Mater 301:538–546
Xu XW, Wang P, Zhang J, Chen C, Wang ZP, Kopittke PM, Kretzschmar R, Zhao FJ (2019) Microbial sulfate reduction decreases arsenic mobilization in flooded paddy soil with high potential for microbial Fe reduction. Environ Pollut 251:952–960
Xue SG, Jiang XX, Wu C, Hartley W, Qian ZY, Luo XH, Li WC (2020) Microbial driven iron reduction affects arsenic transformation and transportation in soil-rice system. Environ Pollut 260:114010
Yao H, Conrad R, Wassmann R, Neue HU (1999) Effect of soil characteristics on sequential reduction and methane production in sixteen rice paddy soils from China, the Philippines, and Italy. Biogeochemistry 47:269–295
Zhang Y, Zhu YB, Zhang SP, Li DX, Xi HL, Wang Y (2022) Arsenic in soils contaminated by arsenic-containing chemical weapons in a site of Jilin, China: fraction and bioaccessibility. Environ Sci Pollut R 29:28957–28972
Zheng TL, Deng YM, Wang YX, Jiang HC, Xie XJ, Gan YQ (2020) Microbial sulfate reduction facilitates seasonal variation of arsenic concentration in groundwater of Jianghan Plain. Central China Sci Total Environ 735:139327
Zhou LM, Lü SL, Zhou JM, Lü CH, Liu B (2008) Study on speciation analysis method of arsenic in Japan’s abandoned chemical weapons contaminated soil (In Chinese). Chin J Anal Lab 27:84–88
Zhu M, Tu C, Hu XF, Zhang HB, Zhang LJ, Wei J, Li Y, Luo YM, Christie P (2016a) Solid-solution partitioning and thionation of diphenylarsinic acid in a flooded soil under the impact of sulfate and iron reduction. Sci Total Environ 569–570:1579–1586
Zhu M, Tu C, Zhang HB, Luo YM, Christie P (2016b) Simultaneous determination of diphenylarsinic and phenylarsinic acids in amended soils by optimized solvent extraction coupled to HPLC-MS/MS. Geoderma 270:109–116
Zhu M, Hu XF, Tu C, Zhang HB, Song F, Luo YM, Christie P (2019) Sorption mechanisms of diphenylarsinic acid on ferrihydrite, goethite and hematite using sequential extraction, FTIR measurement and XAFS spectroscopy. Sci Total Environ 669:991–1000
Zhu M, Luo YM, Cheng NN, Yang RY, Zhang JQ, Zhang MY, Christie P (2020a) Contrasting effects of iron reduction on thionation of diphenylarsinic acid in a biostimulated Acrisol. Environ Sci Pollut R 24:16646–16655
Zhu M, Luo YM, Yang RY, Zhou SB, Zhang JQ, Zhang MY, Christie P, Rylott EL (2020b) Diphenylarsinic acid sorption mechanisms in soils using batch experiments and EXAFS spectroscopy. Front Env Sci Eng 14:58
Zhu M, He YH, Wei XB, Qi HR, Zhang YP, Zhang YJ, Yang RY, Luo YM (2022) Sorption mechanisms of diphenylarsinic acid on natural magnetite and siderite: evidence from sorption kinetics, sequential extraction and extended X-ray absorption fine-structure spectroscopy analysis. Front Env Sci Eng 16:115
Acknowledgements
The STXM data were obtained at the BL08U1A beamline at the Shanghai Synchrotron Radiation Facility (SSRF). We would like to thank Dr. Xiangzhi Zhang at SSRF for his help with the STXM data analysis.
Funding
This work was supported by the National Natural Science Foundation of China (No. 41807117), the Doctoral Research Start-up Funds Project of Anhui Normal University (No. 2018XJJ50), the Cultivation Project on Excellent Undergraduates’ Thesis (design, create) of Anhui Normal University (No. pyjh2021772), and the Project of Innovation and Entrepreneurship Training for College Students (No. S202210370320).
Author information
Authors and Affiliations
Contributions
MZ and RYY designed the research. XBW and YHH completed the experiment and sample analysis. MZ wrote and edited the original draft of the paper. CXH, MXL, and SW revised the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Claudio Bini
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, M., Wei, X., He, Y. et al. Coupled effects of iron and sulfate reduction on the mobilization, thionation, and sequestration of diphenylarsinic acid in a paddy soil. J Soils Sediments 23, 2162–2174 (2023). https://doi.org/10.1007/s11368-023-03463-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-023-03463-5