Abstract
Purpose
NO3−-N reduction is the main way of soil nitrogen (N) loss, especially in saline-alkaline soil, and NO3−-N-reducing Fe(II) oxidation (NRFO) is an important but less known process for NO3−-N reduction. An anoxic cultivation experiment was carried out on coastal saline-alkaline paddy soil to determine the effect of the external environment on NRFO process and microbial community composition.
Methods
A laboratory cultivation experiment was studied to elucidate the effect of pH and acetate on NRFO in saline-alkaline soil. Nine different treatments were designed with two factors: (1) three pH levels: 7.5, 8.0, and 8.5; (2) three acetate concentrations: 15 (C1), 20 (C2), and 25 mM (C3). Concentration of Fe(II), Fe(III), NO3−-N, NO2−-N, and NH4+ and dynamic changes in microorganisms were assessed within 36 days.
Results
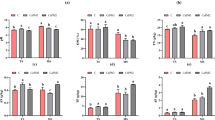
The reduction of NO3−-N showed a positive correlationship with Fe(II) oxidation and Fe(III) formation. In addition to Fe(II), acetate also participated in NO3−-N reduction as an electron donor. The reduction rate of NO3−-N was the highest in pH 8.5 + C3 treatment, in which the contribution of Fe(II) was 46.10%. The loss of NO3−-N was the least in pH 7.5 + C2 treatment, and the contribution of Fe(II) was 37.03%. Illumina high-throughput sequencing showed that the phyla of Proteobacteria and Firmicutes were enriched in all treatments. The classes of Alphaproteobacteria, Bacilli, and Gammaproteobacteria were found to be the dominant in all treatments. Pseudomonas, a NO3−-N reduction bacteria, was dominant in pH 8.5 + C3 treatment, in which the relative abundance was 24%. Meanwhile, a Fe(II) oxidizing bacteria, Acidovorax, was dominant in the pH 8.5 + C1 treatment, and the relative abundance was 15%.
Conclusion
In coastal saline-alkaline paddy field, high C input and high pH (pH 8.5 + C3 treatment) could increase NO3−-N reduction, which might be the important reasons for N loss. Therefore, proper application of organic fertilizer and N fertilizer should be paid attention to in actual production in the Yellow River Delta.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Buchwald C, Grabb K, Hansel C, Wankel S (2016) Constraining the role of iron in environmental nitrogen transformations: dual stable isotope systematics of abiotic NO2- reduction by Fe(II) and its production of N2O. Geochim Cosmochim Acta 186. https://doi.org/10.1016/j.gca.2016.04.041
Byrne-Bailey KG, Coates JD (2012) Complete genome sequence of the anaerobic perchlorate-reducing bacterium azospira suillum strain PS. J Bacteriol 194:2767–2768. https://doi.org/10.1128/JB.00124-12
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Chakraborty A, Picardal F (2013) Induction of nitrate-dependent Fe(II) oxidation by Fe(II) in Dechloromonas sp. strain UWNR4 and Acidovorax sp. strain 2AN. Appl Environ Microbiol 79:748–752. https://doi.org/10.1128/AEM.02709-12
Chen D, Liu T, Li X, Li F, Luo X, Wu Y, Wang Y (2018) Biological and chemical processes of microbially mediated nitrate-reducing Fe(II) oxidation by Pseudogulbenkiania sp. strain 2002. Chem Geol 476:59–69. https://doi.org/10.1016/j.chemgeo.2017.11.004
Chen D, Yuan X, Zhao W, Luo X, Li F, Liu T (2020) Chemodenitrification by Fe(II) and nitrite: pH effect, mineralization and kinetic modeling. Chem Geol 541:119586. https://doi.org/10.1016/j.chemgeo.2020.119586
Coby AJ, Picardal F, Shelobolina E, Xu H, Roden EE (2011) Repeated anaerobic microbial redox cycling of iron. Appl Environ Microbiol 77:6036–6042. https://doi.org/10.1128/AEM.00276-11
Dhakal P, Matocha CJ, Huggins FE, Vandiviere MM (2013) Nitrite Reactivity with Magnetite. Environ Sci Technol 47:6206–6213. https://doi.org/10.1021/es304011w
Jamieson J, Prommer H, Kaksonen AH, Sun J, Siade AJ, Yusov A, Bostick B (2018) Identifying and quantifying the intermediate processes during nitrate-dependent iron(II) oxidation. Environ Sci Technol 52:5771–5781. https://doi.org/10.1021/acs.est.8b01122
Kappler A, Straub K (2005) Geomicrobiological cycling of iron. Rev Mineral Geochem 59:85–108. https://doi.org/10.2138/rmg.2005.59.5
Klueglein N, Kappler A (2013) Abiotic oxidation of Fe(II) by reactive nitrogen species in cultures of the nitrate-reducing Fe(II) oxidizer Acidovorax sp. BoFeN1 – questioning the existence of enzymatic Fe(II) oxidation. Geobiology 11:180–190. https://doi.org/10.1111/gbi.12019
Kögel-Knabner I, Amelung W, Cao Z, Fiedler S, Frenzel P, Jahn R, Kalbitz K, Kölbl A, Schloter M (2010) Biogeochemistry of paddy soils. Geoderma 157:1–14. https://doi.org/10.1016/j.geoderma.2010.03.009
Kuenen JG (2008) Anammox bacteria: from discovery to application. Nat Rev Microbiol 6:320–326. https://doi.org/10.1038/nrmicro1857
Li B, Pan X, Zhang D, Lee DJ, Al-Misned FA, Mortuza MG (2015) Anaerobic nitrate reduction with oxidation of Fe(II) by Citrobacter freundii strain PXL1 – a potential candidate for simultaneous removal of As and nitrate from groundwater. Ecol Eng 77:196–201. https://doi.org/10.1016/j.ecoleng.2015.01.027
Liu P, Bai J, Ding Q, Shao H, Gao H, Xiao R (2012) Effects of water level and salinity on TN and TP contents in marsh soils of the Yellow River Delta, China. CLEAN - Soil Air Water 40:1118–1124. https://doi.org/10.1002/clen.201200029
Liu T, Chen D, Li X, Li F (2019)Microbially mediated coupling of nitrate reduction and Fe(II) oxidation under anoxic conditions. FEMS Microbiol Ecol 95.https://doi.org/10.1093/femsec/fiz030
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27:2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Malhotra J, Aparna K, Dua A, Sangwan N, Trimurtulu N (2014) Microbial and genetic ecology of tropical Vertisols under intensive chemical farming. Environ Monit Assess. https://doi.org/10.1007/s10661-014-4081-2
Melton ED, Rudolph A, Behrens S, Schmidt C, Kappler A (2014) Influence of nutrient concentrations on MPN quantification and enrichment of nitrate-reducing Fe(II)-oxidizing and Fe(III)-reducing bacteria from littoral freshwater lake sediments. Geomicrobiol J 31:788–801. https://doi.org/10.1080/01490451.2014.892765
Miller MN, Dandie CE, Zebarth BJ, Burton DL, Goyer C, Trevors JT (2012) Influence of carbon amendments on soil denitrifier abundance in soil microcosms. Geoderma 170:48–55. https://doi.org/10.1016/j.geoderma.2011.11.022
Miller MN, Zebarth BJ, Dandie CE, Burton DL, Goyer C, Trevors JT (2008) Crop residue influence on denitrification, N2O emissions and denitrifier community abundance in soil. Soil Biol Biochem 40:2553–2562. https://doi.org/10.1016/j.soilbio.2008.06.024
Muehe EM, Gerhardt S, Schink B, Kappler A (2009) Ecophysiology and the energetic benefit of mixotrophic Fe(II) oxidation by various strains of nitrate reducing bacteria. FEMS Microbiol Ecol 70:335–343. https://doi.org/10.1111/j.1574-6941.2009.00755.x
Ratering S, Schnell S (2001) Nitrate-dependent iron(II) oxidation in paddy soil. Environ Microbiol 3:100–109. https://doi.org/10.1046/j.1462-2920.2001.00163.x
Reddy KR, Patrick WH (1986) Denitrification losses in flooded rice fields. In: De Datta S.K., Patrick W.H. (eds) Nitrogen economy of flooded rice soils. Developments in Plant and Soil Sciences, vol 26. Springer, Dordrecht. https://doi.org/10.1007/978-94-009-4428-2_5.
Roden E, McBeth J, Blothe M, Percak-Dennett E, Fleming E, Holyoke R, Luther G, Emerson D (2012) The microbial ferrous wheel in a neutral pH groundwater seep. Front Microbiol 3.https://doi.org/10.3389/fmicb.2012.00172
Siemens J, Haas M, Kaupenjohann M (2003) Dissolved organic matter induced denitrification in subsoils and aquifers? Geoderma 113:253–271. https://doi.org/10.1016/S0016-7061(02)00364-6
Straub KL, Schönhuber WA, Buchholz-Cleven BEE, Schink B (2004) Diversity of ferrous iron-oxidizing, nitrate-reducing bacteria and their involvement in oxygen-independent iron cycling. Geomicrobiol J 21:371–378. https://doi.org/10.1080/01490450490485854
Su Jf, Shao Sc, Huang Tl, Ma F, Yang Sf Zhou Zm, Zheng Sc (2015) Anaerobic nitrate-dependent iron(II) oxidation by a novel autotrophic bacterium, Pseudomonas sp. SZF15. J Environ Chem Eng 3:2187–2193. https://doi.org/10.1016/j.jece.2015.07.030
Wang H, Hu C, Han L, Yang M (2015) Effects of microbial cycling of Fe(II)/Fe(III) and Fe/N on cast iron corrosion in simulated drinking water distribution systems. Corros Sci 100:599–606. https://doi.org/10.1016/j.corsci.2015.08.037
Wang M, Hu R, Zhao J, Kuzyakov Y, Liu S (2016) Iron oxidation affects nitrous oxide emissions via donating electrons to denitrification in paddy soils. Geoderma 271:173–180. https://doi.org/10.1016/j.geoderma.2016.02.022
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267. https://doi.org/10.1128/aem.00062-07
Weber KA, Achenbach LA, Coates JD (2006) Microorganisms pumping iron: anaerobic microbial iron oxidation and reduction. Nat Rev Microbiol 4:752–764. https://doi.org/10.1038/nrmicro1490
Wu L, Wei C, Zhang S, Wang Y, Kuzyakov Y, Ding X (2019) MgO-modified biochar increases phosphate retention and rice yields in saline-alkaline soil. J Clean Prod 235:901–909. https://doi.org/10.1016/j.jclepro.2019.07.043
Wu L, Zhang S, Wang J, Ding X (2020) Phosphorus retention using iron (II/III) modified biochar in saline-alkaline soils: adsorption, column and field tests. Environ Pollut 261:114223. https://doi.org/10.1016/j.envpol.2020.114223
Zhao L, Dong H, Edelmann RE, Zeng Q, Agrawal A (2017) Coupling of Fe(II) oxidation in illite with nitrate reduction and its role in clay mineral transformation. Geochim Cosmochim Acta 200:353–366. https://doi.org/10.1016/j.gca.2017.01.004
Zhao L, Dong H, Kukkadapu RK, Zeng Q, Edelmann RE, Pentrak M, Agrawal A (2015) Biological redox cycling of iron in nontronite and its potential application in nitrate removal. Environ Sci Technol 49:5493–5501. https://doi.org/10.1021/acs.est.5b00131
Funding
This work was supported in part by the National Key Research and Development Projects, China (2017YFD0200200/2017YFD0200208-3), the Soil and Cultivation Position of Modern Agricultural Technology System Innovation Team in Shandong Province, China (SDAIT-17–05), and the Natural Science Fund of Shandong Province, China (ZR2020MC154).
Author information
Authors and Affiliations
Contributions
Conceptualization: Xiaodong Ding, Yinghui Jiang. Data curation: Yinghui Jiang, Xiaodong Ding. Formal analysis: Yinghui Jiang, Xiaodong Ding. Funding acquisition: Xiaodong Ding, Shirong Zhang. Investigation: Yinghui Jiang, Shirong Zhang, Ruxue Wei. Methodology: Yinghui Jiang, Xiaodong Ding. Software: Yinghui Jiang, Ruxue Wei. Writing-original draft: Yinghui Jiang, Xiaodong Ding, Shirong Zhang. Writing-review and editing: Yinghui Jiang, Shirong Zhang, Xiaodong Ding, Ruxue Wei.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Jizheng He
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiang, Y., Zhang, S., Wei, R. et al. Microbial community changes during anaerobic nitrate reduction and Fe(II) oxidation of a coastal saline paddy soil under alkaline pH. J Soils Sediments 22, 2720–2730 (2022). https://doi.org/10.1007/s11368-022-03247-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-022-03247-3