Abstract
Purpose
Ammonia-oxidizing archaea (AOA) are ubiquitous in aquatic and terrestrial environments, and the copper-containing nitrite reductase (NirK) plays a pivotal role in AOA metabolism. However, distribution and function of nirK gene-containing thaumarchaea (AnirK) have rarely been estimated, especially in terrestrial ecosystems.
Methods
Soil and sediment samples were taken from 20 sites including coastal mud flat, freshwater wetland, paddy soil, agricultural upland, and forest soil. Potential N2O production rates, community composition, and transcriptional activity of nirK gene-containing thaumarchaea in different habitats were investigated by combining field surveys with microcosm incubations.
Results
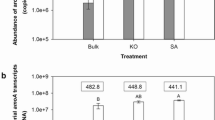
AnirK gene abundance and N2O production rates were significantly higher in flooded habitats like freshwater wetland and paddy soil than in agricultural upland and forest soils. In comparison to the AOA amoA gene, AnirK gene was less diverse and mainly affiliated within Candidatus Nitrosoarchaeum, Nitrosopumilus, and Candidatus Nitrosotenuis (Group I.1a) and Candidatus Nitrosotalea (Group I.1a-associated). Microcosm incubations further demonstrated that AnirK gene transcription activity was significantly higher than bacterial nirK and nirS genes, corresponding to the high nitrate reduction and N2O emissions in wetland and paddy soil. Nitrosopumilus in coastal sediment and Candidatus Nitrosoarchaeum in paddy soil and wetland were identified as the active AnirK gene carriers.
Conclusion
Our study demonstrated that the AnirK gene was widely present in coastal sediment and terrestrial ecosystem like wetland and paddy soils, and that AnirK gene-containing Nitrosopumilus and Candidatus Nitrosoarchaeum are potential active contributors to nitrite reduction and N2O production in these ecosystems.






Similar content being viewed by others
References
Abell GCJ, Revill AT, Smith C, Bissett AP, Volkman JK, Robert SS (2010) Archaeal ammonia oxidizers and nirS-type denitrifiers dominate sediment nitrifying and denitrifying populations in a subtropical macrotidal estuary. ISME J 4:286–300
Alves RJE, Minh BQ, Urich T, von Haeseler A, Schleper C (2018) Unifying the global phylogeny and environmental distribution of ammonia-oxidising archaea based on amoA genes. Nat Commun 9:1517
Arévalo-Martínez DL, Kock A, Löscher CR, Schmitz RA, Bange HW (2015) Massive nitrous oxide emissions from the tropical South Pacific Ocean. Nat Geosci 8:530–533
Assémien FL, Cantarel AAM, Florio A, Lerondelle C, Pommier T, Gonnety JT, Le Roux X (2019) Different groups of nitrite-reducers and N2O-reducers have distinct ecological niches and functional roles in West African cultivated soils. Soil Biol Biochem 129:39–47
Attard E, Recous S, Chabbi A, De Berranger C, Guillaumaud N, Labreuche J, Philippot L, Schmid B, Le Roux X (2011) Soil environmental conditions rather than denitrifier abundance and diversity drive potential denitrification after changes in land uses. Glob Chang Biol 17:1975–1989
Bai R, Fang YT, Mo LY, Shen JP, Song LL, Wang YQ, Zhang LM, He JZ (2020) Greater promotion of DNRA rates and nrfA gene transcriptional activity by straw incorporation in alkaline than in acidic paddy soils. Soil Ecol Lett 2:255–267
Bartossek R, Nicol GW, Lanzen A, Klenk H, Schleper C (2010) Homologues of nitrite reductases in ammonia-oxidizing archaea: diversity and genomic context. Environ Microbiol 12:1075–1088
Blainey PC, Mosier AC, Potanina A, Francis CA, Quake SR (2011) Genome of a low-salinity ammonia-oxidizing archaeon determined by single-cell and metagenomic analysis. PLoS ONE 6:e16626
Bowen H, Maul JE, Cavigelli MA, Yarwood S (2020) Denitrifier abundance and community composition linked to denitrification activity in an agricultural and wetland soil. Appl Soil Ecol 151:103521
Chapuis-Lardy L, Wrage N, Metay A, Chotte JL, Bernoux M (2007) Soils, a sink for N2O? A review. Glob Chang Biol 13:1–17
Chen XP, Zhu YG, Xia Y, Shen JP, He JZ (2008) Ammonia-oxidizing archaea: important players in paddy rhizosphere soil? Environ Microbiol 10:1978–1987
Chen Z, Luo X, Hu R, Wu M, Wu J, Wei W (2010) Impact of long-term fertilization on the composition of denitrifier communities based on nitrite reductase analyses in a paddy soil. Microb Ecol 60:850–861
Dai WW, Bai E, Li W, Jiang P, Dai GH, Zheng XB (2020) Predicting plant–soil N cycling and soil N2O emissions in a Chinese old-growth temperate forest under global changes: uncertainty and implications. Soil Ecol Lett 2:73–82
DeLong EF (1998) Everything in moderation: Archaea as “non-extremophiles.” Curr Opin Genet Dev 8:649–654
Dick WA, Tabatabai MA (1977) An alkaline oxidation method for determination of total phosphorus in soils. Soil Sci Soc Am J 41:511–514
Ding LL, Zhou JY, Li QY, Tang JJ, Chen X (2022) Effects of land-use type and flooding on the soil microbial community and functional genes in reservoir riparian zones. Microb Ecol 83:393–407
Dörsch P, Braker G, Bakken LR (2012) Community-specific pH response of denitrification: experiments with cells extracted from organic soils. FEMS Microbiol Ecol 79:530–541
Duff AM, Zhang LM, Smith CJ (2017) Small-scale variation of ammonia oxidisers within intertidal sediments dominated by ammonia-oxidising bacteria Nitrosomonas sp. amoA genes and transcripts. Sci Rep 7:13200
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998
Francis CA, Roberts KJ, Beman JM, Santoro AE, Oakley BB (2005) Ubiquity and diversity of ammonia-oxidizing archaea in water columns and sediments of the ocean. P Natl Acad Sci USA 102:14683–14688
Frank JA, Reich CI, Sharma S, Weisbaum JS, Wilson BA, Olsen GJ (2008) Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl Environ Microb 74:2461–2470
Freing A, Wallace DWR, Bange HW (2012) Global oceanic production of nitrous oxide. Philos T R Soc B 367:1245–1255
French E, Kozlowski JA, Mukherjee M, Bullerjahn G, Bollmann A (2012) Ecophysiological characterization of ammonia-oxidizing archaea and bacteria from freshwater. Appl Environ Microb 78:5773–5780
Frostegård Å, Vick SH, Lim NY, Bakken LR, Shapleigh JP (2022) Linking meta-omics to the kinetics of denitrification intermediates reveals pH-dependent causes of N2O emissions and nitrite accumulation in soil. ISME J: 26–37
Grömping U (2006) Relative importance for linear regression in R: the package relaimpo. J Stat Softw 17:1–27
Gubry-Rangin C, Hai B, Quince C, Engel M, Thomson BC, James P, Schloter M, Griffiths RI, Prosser JI, Nicol GW (2011) Niche specialization of terrestrial archaeal ammonia oxidizers. P Natl Acad Sci USA 108:21206–21211
Gubry-Rangin C, Nicol GW, Prosser JI (2010) Archaea rather than bacteria control nitrification in two agricultural acidic soils. FEMS Microbiol Ecol 74:566–574
Hallin S, Jones CM, Schloter M, Philippot L (2009) Relationship between N-cycling communities and ecosystem functioning in a 50-year-old fertilization experiment. ISME J 3:597–605
Han B, Mo LY, Fang YT, Di HJ, Wang JT, Shen JP, Zhang LM (2021) Rates and microbial communities of denitrification and anammox across coastal tidal flat lands and inland paddy soils in East China. Appl Soil Ecol 157:103768
Herbold CW, Lebedeva E, Palatinszky M, Wagner M (2016) Candidatus Nitrosotenuis. In: Trujillo ME, Dedysh S, DeVos P, Hedlund B, Kämpfer P, Rainey FA, Whitman WB (eds) Bergey's Manual of Systematics of Archaea and Bacteria
Hink L, Gubry-Rangin C, Nicol GW, Prosser JI (2018) The consequences of niche and physiological differentiation of archaeal and bacterial ammonia oxidisers for nitrous oxide emissions. ISME J 12:1084–1093
Hink L, Nicol GW, Prosser JI (2017) Archaea produce lower yields of N2O than bacteria during aerobic ammonia oxidation in soil. Environ Microbiol 19:4829–4837
Hollibaugh JT, Gifford S, Sharma S, Bano N, Moran MA (2011) Metatranscriptomic analysis of ammonia-oxidizing organisms in an estuarine bacterioplankton assemblage. ISME J 5:866–878
Houlton BZ, Bai E (2009) Imprint of denitrifying bacteria on the global terrestrial biosphere. P Natl Acad Sci USA 106:21713–21716
Hu HW, Chen DL, He JZ (2015) Microbial regulation of terrestrial nitrous oxide formation: understanding the biological pathways for prediction of emission rates. FEMS Microbiol Rev 39:729–749
IPCC (2014) Climate change 2014: synthesis report. Contribution of Working Groups I, II and III to the fifth assessment report of the Intergovernmental Panel on Climate Change [Core Writing Team, Pachauri RK, Meyer LA (eds)]. IPCC, Geneva, Switzerland 151
Jung MY, Park SJ, Min D, Kim JS, Rijpstra WIC, Sinninghe Damsté JS, Kim GJ, Madsen EL, Rhee SK (2011) Enrichment and characterization of an autotrophic ammonia-oxidizing archaeon of mesophilic crenarchaeal group I.1a from an agricultural soil. Appl Environ Microb 77:8635–8647
Jung MY, Well R, Min D, Giesemann A, Park SJ, Kim JG, Kim SJ, Rhee SK (2014) Isotopic signatures of N2O produced by ammonia-oxidizing archaea from soils. ISME J 8:1115–1125
Kerou M, Alves RJE, Schleper C (2018) Nitrososphaerales. In: Trujillo ME, Dedysh S, DeVos P, Hedlund B, Kämpfer P, Rainey FA, Whitman WB (eds) Bergey's Manual of Systematics of Archaea and Bacteria
Kerou M, Offre P, Valledor L, Abby SS, Melcher M, Nagler M, Weckwerth W, Schleper C (2016) Proteomics and comparative genomics of Nitrososphaera viennensis reveal the core genome and adaptations of archaeal ammonia oxidizers. P Natl Acad Sci USA 113:E7937–E7946
Kim BK, Jung M, Yu DS, Park S, Oh TK, Rhee S, Kim JF (2011) Genome sequence of an ammonia-oxidizing soil archaeon, “Candidatus Nitrosoarchaeum koreensis” MY1. J Bacteriol 193:5539–5540
Kobayashi S, Hira D, Yoshida K, Toyofuku M, Shida Y, Ogasawara W, Yamaguchi T, Araki N, Oshiki M (2018) Nitric oxide production from nitrite reduction and hydroxylamine oxidation by copper-containing dissimilatory nitrite Reductase (NirK) from the aerobic ammonia-oxidizing archaeon, Nitrososphaera viennensis. Microbes Environ 33:428–434
Könneke M, Bernhard AE, de la Torre JR, Walker CB, Waterbury JB, Stahl DA (2005) Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 437:543–546
Kozlowski JA, Stieglmeier M, Schleper C, Klotz MG, Stein LY (2016) Pathways and key intermediates required for obligate aerobic ammonia-dependent chemolithotrophy in bacteria and Thaumarchaeota. ISME J 10:1836–1845
Kuypers MMM, Marchant HK, Kartal B (2018) The microbial nitrogen-cycling network. Nat Rev Microbiol 16: 263-276
Le Roux X, Schmid B, Poly F, Barnard RL, Niklaus PA, Guillaumaud N, Habekost M, Oelmann Y, Philippot L, Salles JF, Schloter M, Steinbeiss S, Weigelt A (2013) Soil environmental conditions and microbial build-up mediate the effect of plant diversity on soil nitrifying and denitrifying enzyme activities in temperate grasslands. PLoS ONE 8:e61069
Lehtovirta-Morley LE, Stoecker K, Vilcinskas A, Prosser JI, Nicol GW (2011) Cultivation of an obligate acidophilic ammonia oxidizer from a nitrifying acid soil. P Natl Acad Sci USA 108:15892–15897
Lennon EF, Houlton BZ (2017) Coupled molecular and isotopic evidence for denitrifier controls over terrestrial nitrogen availability. ISME J 11:727–740
Long A, Song B, Fridey K, Silva A (2015) Detection and diversity of copper containing nitrite reductase genes (nirK) in prokaryotic and fungal communities of agricultural soils. FEMS Microbiol Ecol 91:1–9
Long XE, Shen JP, Wang JT, Zhang LM, Di HJ, He JZ (2017) Contrasting response of two grassland soils to N addition and moisture levels: N2O emission and functional gene abundance. J Soils Sediments 17:384–392
Löscher CR, Kock A, Könneke M, LaRoche J, Bange HW, Schmitz RA (2012) Production of oceanic nitrous oxide by ammonia-oxidizing archaea. Biogeosciences 9:2419–2429
Lund MB, Smith JM, Francis CA (2012) Diversity, abundance and expression of nitrite reductase (nirK)-like genes in marine thaumarchaea. ISME J 6:1966–1977
Luo J, Tillman RW, Ball PR (2000) Nitrogen loss through denitrification in a soil under pasture in New Zealand. Soil Biol Biochem 32:497–509
Lycus P, Bøthun KL, Bergaust L, Shapleigh JP, Bakken LR, Frostegård Å (2017) Phenotypic and genotypic richness of denitrifiers revealed by a novel isolation strategy. ISME J 11: 2219-2232
Maag M, Vinther FP (1996) Nitrous oxide emission by nitrification and denitrification in different soil types and at different soil moisture contents and temperatures. Appl Soil Ecol 4:5–14
Muck S, De Corte D, Clifford EL, Bayer B, Herndl GJ, Sintes E (2019) Niche differentiation of aerobic and anaerobic ammonia oxidizers in a high latitude deep oxygen minimum zone. Front Microbiol 10:2141
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36
Pester M, Rattei T, Flechl S, Gröngröft A, Richter A, Overmann J, Reinhold Hurek B, Loy A, Wagner M (2012) amoA-based consensus phylogeny of ammonia-oxidizing archaea and deep sequencing of amoA genes from soils of four different geographic regions. Environ Microbiol 14:525–539
Philippot L (2002) Denitrifying genes in bacterial and archaeal genomes. BBA-Gene Struct Expr 1577:355–376
Philippot L, Čuhel J, Saby NPA, Chèneby D, Chroňáková A, Bru D, Arrouays D, Martin-Laurent F, Šimek M (2009) Mapping field-scale spatial patterns of size and activity of the denitrifier community. Environ Microbiol 11:1518–1526
Philippot L, Hallin S, Schloter M (2007) Ecology of denitrifying prokaryotes in agricultural soil. Adv Agron 96:249–305
Prosser JI, Hink L, Gubry Rangin C, Nicol GW (2019) Nitrous oxide production by ammonia oxidizers: Physiological diversity, niche differentiation and potential mitigation strategies. Glob Chang Biol 26:103–118
Qin W, Amin SA, Lundeen RA, Heal KR, Martens-Habbena W, Turkarslan S, Urakawa H, Costa KC, Hendrickson EL, Wang T, Beck DA, Tiquia-Arashiro SM, Taub F, Holmes AD, Vajrala N, Berube PM, Lowe TM, Moffett JW, Devol AH, Baliga NS, Arp DJ, Sayavedra-Soto LA, Hackett M, Armbrust EV, Ingalls AE, Stahl DA (2018) Stress response of a marine ammonia-oxidizing archaeon informs physiological status of environmental populations. ISME J 12:508–519
Qu Z, Wang JG, AlmØy T, Bakken LR (2014) Excessive use of nitrogen in Chinese agriculture results in high N2O/(N2O+N2) product ratio of denitrification, primarily due to acidification of the soils. Glob Chang Biol 20:1685–1698
Ravishankara AR, Da Niel JS, Portmann RW (2009) Nitrous Oxide (N2O): The dominant ozone-depleting dubstance emitted in the 21st century. Science 326:123–125
Reji L, Tolar BB, Smith JM, Chavez FP, Francis CA (2019) Depth distributions of nitrite reductase (nirK) gene variants reveal spatial dynamics of thaumarchaeal ecotype populations in coastal Monterey Bay. Environ Microbiol 21:4032–4045
Rich JJ, Myrold DD (2004) Community composition and activities of denitrifying bacteria from adjacent agricultural soil, riparian soil, and creek sediment in Oregon, USA. Soil Biol Biochem 36:1431–1441
Santoro AE, Buchwald C, McIlvin MR, Casciotti KL (2011) Isotopic signature of N2O produced by marine ammonia-oxidizing archaea. Science 333:1282–1285
Santoro AE, Dupont CL, Richter RA, Craig MT, Carini P, McIlvin MR, Yang Y, Orsi WD, Moran DM, Saito MA (2015) Genomic and proteomic characterization of “Candidatus Nitrosopelagicus brevis”: An ammonia-oxidizing archaeon from the open ocean. P Natl Acad Sci USA 112:1173–1178
Seitzinger S, Harrison JA, Böhlke JK, Bouwman AF, Lowrance R, Peterson B, Tobias C, Drecht GV (2006) Denitrification across landscapes and waterscapes: a synthesis. Ecol Appl 16:2064–2090
Shi YM, Tyson GW, Eppley JM, DeLong EF (2011) Integrated metatranscriptomic and metagenomic analyses of stratified microbial assemblages in the open ocean. ISME J 5:999–1013
Smith MS, Tiedje JM (1979) Phases of denitrification following oxygen depletion in soil. Soil Biol Biochem 11:261–267
Srikanthasamy T, Leloup J, N’Dri AB, Barot S, Gervaix J, Koné AW, Koffi KF, Le Roux X, Raynaud X, Lata J (2018) Contrasting effects of grasses and trees on microbial N-cycling in an African humid savanna. Soil Biol Biochem 117:153–163
Stieglmeier M, Mooshammer M, Kitzler B, Wanek W, Zechmeister-Boltenstern S, Richter A, Schleper C (2014) Aerobic nitrous oxide production through N-nitrosating hybrid formation in ammonia-oxidizing archaea. ISME J 8:1135–1146
Tolar BB, Reji L, Smith JM, Blum M, Pennington JT, Chavez FP, Francis CA (2020) Time series assessment of Thaumarchaeota ecotypes in Monterey Bay reveals the importance of water column position in predicting distribution-environment relationships. Limnol Oceanogr 65:2041–2055
Treusch AH, Leininger S, Kletzin A, Schuster SC, Klenk HP, Schleper C (2005) Novel genes for nitrite reductase and Amo-related proteins indicate a role of uncultivated mesophilic crenarchaeota in nitrogen cycling. Environ Microbiol 7:1985–1995
Trimmer M, Chronopoulou PM, Maanoja ST, Upstill-Goddard RC, Kitidis V, Purdy KJ (2016) Nitrous oxide as a function of oxygen and archaeal gene abundance in the North Pacific. Nat Commun 7:13451
Venter JC, Remington K, Heidelberg JF, Halpern AL, Rusch D, Eisen JA, Wu D, Paulsen I, Nelson KE, Nelson W (2004) Environmental genome shotgun sequencing of the Sargasso Sea. Science 304:66–74
Waldron PJ, Petsch ST, Martini AM, Nüsslein K, (2007) Salinity constraints on subsurface archaeal diversity and methanogenesis in sedimentary rock rich in organic matter. Appl Environ Microb 73:4171–4179
Walker CB, de la Torre JR, Klotz MG, Urakawa H, Pinel N, Arp DJ, Brochier-Armanet C, Chain PSG, Chan PP, Gollabgir A, Hemp J, Hugler M, Karr EA, Konneke M, Shin M, Lawton TJ, Lowe T, Martens-Habbena W, Sayavedra-Soto LA, Lang D, Sievert SM, Rosenzweig AC, Manning G, Stahl DA (2010) Nitrosopumilus maritimus genome reveals unique mechanisms for nitrification and autotrophy in globally distributed marine crenarchaea. P Natl Acad Sci USA 107:8818–8823
Ward BB, Devol AH, Rich JJ, Chang BX, Bulow SE, Naik H, Pratihary A, Jayakumar A (2009) Denitrification as the dominant nitrogen loss process in the Arabian Sea. Nature 461:78–81
Wei W, Isobe K, Nishizawa T, Zhu L, Shiratori Y, Ohte N, Koba K, Otsuka S, Senoo K (2015a) Higher diversity and abundance of denitrifying microorganisms in environments than considered previously. ISME J 9:1954–1965
Wei W, Isobe K, Shiratori Y, Nishizawa T, Ohte N, Ise Y, Otsuka S, Senoo K (2015b) Development of PCR primers targeting fungal nirK to study fungal denitrification in the environment. Soil Biol Biochem 81:282–286
Williams TJ, Long E, Evans F, DeMaere MZ, Lauro FM, Raftery MJ, Ducklow H, Grzymski JJ, Murray AE, Cavicchioli R (2012) A metaproteomic assessment of winter and summer bacterioplankton from Antarctic Peninsula coastal surface waters. ISME J 6:1883–1900
Wu L, Chen XM, Wei W, Liu YW, Wang DB, Ni BJ (2020) A critical review on nitrous oxide production by ammonia-oxidizing archaea. Environ Sci Technol 54:9175–9190
Xie W, Zhang CL, Zhou XD, Wang P (2014) Salinity-dominated change in community structure and ecological function of Archaea from the lower Pearl River to coastal South China Sea. Appl Microbiol Biot 98:7971–7982
Xu YB, Cai ZC (2007) Denitrification characteristics of subtropical soils in China affected by soil parent material and land use. Eur J Soil Sci 58:1293–1303
Yakimov MM, La Cono V, Smedile F, DeLuca TH, Juárez S, Ciordia S, Fernández M, Albar JP, Ferrer M, Golyshin PN (2011) Contribution of crenarchaeal autotrophic ammonia oxidizers to the dark primary production in Tyrrhenian deep waters (Central Mediterranean Sea). ISME J 5:945–961
Yoshida M, Ishii S, Otsuka S, Senoo K (2009) nirK-harboring denitrifiers are more responsive to denitrification-inducing conditions in rice paddy soil than nirS-harboring bacteria. Microbes Environ 25:45–48
Zhang LM, Duff AM, Smith CJ (2018) Community and functional shifts in ammonia oxidizers across terrestrial and marine (soil/sediment) boundaries in two coastal Bay ecosystems. Environ Microbiol 20:2834–2853
Zhang MM, Alves RJE, Zhang DD, Han LL, He JZ, Zhang LM (2017a) Time-dependent shifts in populations and activity of bacterial and archaeal ammonia oxidizers in response to liming in acidic soils. Soil Biol Biochem 112:77–89
Zhang Y, Zhao W, Zhang JB, Cai ZC (2017b) N2O production pathways relate to land use type in acidic soils in subtropical China. J Soils Sediments 17:306–314
Zhu X, Burger M, Doane TA, Horwath WR (2013) Ammonia oxidation pathways and nitrifier denitrification are significant sources of N2O and NO under low oxygen availability. P Natl Acad Sci USA 110:6328–6333
Acknowledgements
We would like to thank Anhui Ge, Yabo Zhang, Yaqi Wang, and Wei Shi for assistance in soil sampling, and Ren Bai for helps in laboratory analysis.
Funding
This study was financially supported by the National Natural Science Foundation of China (41771288).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Jizheng He
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11368_2022_3219_MOESM1_ESM.pdf
Supplementary file1 (PDF 1823 kb). Fig. S1 Location of sampling sites. Different colors represent different ecosystem types including coastal mud flat, freshwater wetland, paddy soil, agricultural upland, and forest soil. The sites indicated by stars are chosen for microcosm incubation.
11368_2022_3219_MOESM2_ESM.pdf
Supplementary file2 (PDF 444 kb). Fig. S2 The abundances of archaeal nirK gene estimated with the primer pair 58F/579R for AnirK and the primer pair qAnirK1F/qAnirK1R for qAnirK in each site. “N.” means below the detection limit. The asterisks above the bars indicate a significant difference (P < 0.05) between AnirK and qAnirK gene abundances. Vertical bars indicate standard errors of three replicates within each site.
11368_2022_3219_MOESM3_ESM.pdf
Supplementary file3 (PDF 234 kb). Fig. S3 Regression relationship between the NO3–-N reduction amount and rates of N2O production. Different colors refer to the different sites, and different shapes refer to different incubation time points.
Rights and permissions
About this article
Cite this article
Han, B., Liu, SY., Han, LL. et al. The contribution of nirK gene-containing thaumarchaea to denitrification and N2O production across coastal sediment and terrestrial ecosystems. J Soils Sediments 22, 2246–2261 (2022). https://doi.org/10.1007/s11368-022-03219-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-022-03219-7