Abstract
Purpose
In the soil–water system, adsorption–desorption characteristics of pesticides significantly influence their environmental fate (mobility, degradation, and transformation). However, bioavailability of pesticides can be modified by the addition of organic amendments in soils. Flupyradifurone is a neonicotinoid insecticide widely used to control sucking insect pests of vegetables in India. In this experiment, effect of organic amendment was investigated on the adsorption–desorption behavior of flupyradifurone in two Indian soils.
Methods
Soils used in this experiment were collected from the plough layer (0–15 cm) of two different agroclimatic zones of India; Terai soil of West Bengal (Entisol) and Alluvial soil of Delhi (Inceptisol). The batch equilibrium technique was used to evaluate the kinetics and sorption behavior of flupyradifurone in test soils. Residues of flupyradifurone were quantified by HPLC (High-Performance Liquid Chromatography).
Results
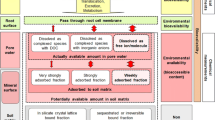
Sorption kinetics of flupyradifurone indicated that maximum adsorption was achieved at 24 h in both soils, and sorption kinetics was best explained by pseudo second-order model. High clay and organic carbon content of Entisol could be attributed to higher adsorption (37.92 ± 0.19) of flupyradifurone in Entisol as compared to Inceptisol (22.37 ± 0.19). Furthermore, addition of farmyard manure (FYM) markedly increased the adsorption capacity of both soils (49.98 ± 0.19 for Entisol and 22.37 ± 0.19 for Inceptisol). Adsorption data were best fitted with Freundlich adsorption isotherm, and slope of the Freundlich adsorption isotherm (1/n) in all soils were < 1 indicating that sorption was non-linear. Calculation of thermodynamic parameters pointed out that sorption was spontaneous and more favored at high temperatures. Flupyradifurone’s desorption rate is slower than the adsorption rate for two test soils, indicating that positive hysteresis occurs. The primary mechanism involved in flupyradifurone sorption for both amended and unamended soils was physical adsorption (surface adsorption and partition).
Conclusion
This experiment has relevance in assessing the fate of flupyradifurone in amended field conditions. Additionally, dose optimization can also be done in an attempt to reduce soil/groundwater contamination.




Similar content being viewed by others
Data availability
NA.
Code availability
NA.
References
Ahmed MB, Zhou JL, Ngo HH, Guo W, Johir MAH, Belhaj D (2017) Competitive sorption affinity of sulfonamides and chloramphenicol antibiotics toward functionalized biochar for water and wastewater treatment. Bioresour Technol 238:306–312. https://doi.org/10.1016/j.biortech.2017.04.042
Al-Aoh HA, Jamil Maah M, Ahmad AA, Radzi Bin Abas M (2012) Adsorption of 4-nitrophenol on palm oil fuel ash activated by amino silane coupling agent. Desalin Water Treat 40:159–167. https://doi.org/10.1080/19443994.2012.671162
Báez ME, Espinoza J, Silva R, Fuentes E (2015) Sorption-desorption behavior of pesticides and their degradation products in volcanic and nonvolcanic soils: interpretation of interactions through two-way principal component analysis. Environ Sci Pollut Res 22:8576–8585. https://doi.org/10.1007/s11356-014-4036-8
Black CA (1965) Methods of soil analysis (parts 1 and 2). American Society of Agronomy, Madison
Carpio MJ, Marín-Benito JM, García-Delgado C, Sánchez-Martín MJ, Rodríguez-Cruz MS (2020) Soil microbial community changes in a field treatment with chlorotoluron, flufenacet and diflufenican and two organic amendments. Agronomy 10:8–10. https://doi.org/10.3390/agronomy10081166
Carter MC, Kilduff JE, Weber WJ (1995) Site energy distribution analysis of preloaded adsorbents. Environmental Science & Technology 29:1773–1780. https://doi.org/10.1021/es00007a013
Chen X, Zhou Q, Liu F, Peng Q, Teng P (2019) Removal of nine pesticide residues from water and soil by biosorption coupled with degradation on biosorbent immobilized laccase. Chemosphere 233:49–56. https://doi.org/10.1016/j.chemosphere.2019.05.144
Cheng H, Hu E, Hu Y (2012) Impact of mineral micropores on transport and fate of organic contaminants: a review. J Contam Hydrol 129–130:80–90. https://doi.org/10.1016/j.jconhyd.2011.09.008
Cheung WH, Szeto YS, McKay G (2007) Intraparticle diffusion processes during acid dye adsorption onto chitosan. Bioresour Technol 98:2897–2904. https://doi.org/10.1016/j.biortech.2006.09.045
Das SK, Mukherjee I, Kumar A (2015) Effect of soil type and organic manure on adsorption–desorption of flubendiamide. Environ Monit Assess 187(7). https://doi.org/10.1007/s10661-015-4623-2
Das SK, Mukherjee I (2011) Effect of light and pH on persistence of flubendiamide. Bull Environ Contam Toxicol 87:292–296. https://doi.org/10.1007/s00128-011-0333-9
Das SK, Mukherjee I (2012) Effect of moisture and organic manure on persistence of flubendiamide in soil. Bull Environ Contam Toxicol 88:515–520. https://doi.org/10.1007/s00128-012-0551-9
Datta SC, Ghosh SK, Das D (2020) Soil Mineralogy and Clay Minerals. The Soils of India. Springer, Cham, pp 109–127
European Food Safety Authority (2015) Conclusion on the peer review of the pesticide risk assessment of the active substance flupyradifurone. EFSA J. https://doi.org/10.2903/j.efsa.2015.4020
Gajbhiye VT, Gupta S (2001) Adsorption-desorption behaviour of flufenacet in five different soils of India. Pest Manag Sci 57:633–639. https://doi.org/10.1002/ps.340
Gaonkar OD, Nambi IM, Govindarajan SK (2019) Soil organic amendments: impacts on sorption of organophosphate pesticides on an alluvial soil. J Soils Sediments 19:566–578. https://doi.org/10.1007/s11368-018-2080-6
González-Márquez LC, Hansen AM, González-Farias FA (2018) Effect of mono and divalent salts on the conformation and composition of a humic acid and on atrazine adsorption. Environ Sci Pollut Res 25:17509–17518. https://doi.org/10.1007/s11356-018-1939-9
Gustafon D (1989) Groundwater ubiquity score: a simple method for assessing pesticide leachability. Environ Toxicol Chem 8:339–357. https://doi.org/10.1002/etc.5620080411
Hu E, Zhao X, Pan S, Ye Z, He F (2019) Sorption of non-ionic aromatic organics to mineral micropores: interactive effect of cation hydration and mineral charge density. Environ Sci Technol 53:3067–3077. https://doi.org/10.1021/acs.est.9b00145s
Ihara M, Buckingham SD, Matsuda K, Sattelle DB (2017) Modes of action, resistance and toxicity of insecticides targeting nicotinic acetylcholine receptors. Curr Med Chem 24:1–8. https://doi.org/10.2174/0929867324666170206142019
Jackson ML (1967) Soil Chemical Analysis. Prentice-Hall of India Pvt Ltd, New Delhi.
Kara M, Yuzer H, Sabah E, Celik MS (2003) Adsorption of cobalt from aqueous solutions onto sepiolite. Water Res 37:224–232. https://doi.org/10.1016/S0043-1354(02)00265-8
Kumar A, Singh N (2020) Effect of crop residue ashes on sorption behavior of herbicides used in the succeeding crop in Indian soils. J Environ Sci Heal - Part B Pestic Food Contam Agric Wastes 55:630–645. https://doi.org/10.1080/03601234.2020.1757980
Kumar N, Mukherjee I, Varghese E (2015) Adsorption–desorption of tricyclazole: effect of soil types and organic matter. Environ Monit Assess 87(3). https://doi.org/10.1007/s10661-015-4280-5
Mandal A, Singh N (2016) Kinetic and isotherm error optimization studies for adsorption of atrazine and imidacloprid on bark of Eucalyptus tereticornis L. J Environ Sci Heal - Part B 51:192–203. https://doi.org/10.1080/03601234.2015.1108817
Moreau-Kervévan C, Mouvet C (1998) Adsorption and desorption of atrazine, deethylatrazine, and hydroxyatrazine by soil components. J Environ Qual 27:46–53. https://doi.org/10.2134/jeq1998.00472425002700010008x
Motoki Y, Iwafune T, Seike N, Otani T, Asano M (2014) Effects of organic carbon quality on the sorption behavior of pesticides in Japanese soils. J Pestic Sci 39:105–114. https://doi.org/10.1584/jpestics.D13-067
Ngah WSW, Fatinathan S, Yosop NA (2011) Isotherm and kinetic studies on the adsorption of humic acid onto chitosan-H2SO4 beads. Desalination 272:293–300. https://doi.org/10.1016/j.desal.2011.01.024
Oepen B Von, Kordel W, Klein W (1991) Sorption of nonpolar and polar compounds to soils: processes, measurements and experience with the applicability of the modified OECD-Guideline 106. Chemosphere 22:285–304. https://doi.org/10.1016/0045-6535(91)90318-8
Ou L, Gannon TW, Arellano C, Polizzotto ML (2018) A global meta-analysis to predict atrazine sorption from soil properties. J Environ Qual 47:1389–1399. https://doi.org/10.2134/jeq2017.11.0429
Parolo ME, Savini MC, Loewy RM (2017) Characterization of soil organic matter by FT-IR spectroscopy and its relationship with chlorpyrifos sorption. J Environ Manage 196:316–322. https://doi.org/10.1016/j.jenvman.2017.03.018
Pérez-Lucas G, El Aatik A, Vela N, Fenoll J, Navarro S (2021) Exogenous organic matter as strategy to reduce pesticide leaching through the soil. Arch Agron Soil Sci 67:934–945. https://doi.org/10.1080/03650340.2020.1768531
Pignatello JJ, Nason SL (2020) Importance of Soil Properties and Processes on Bioavailability of Organic Compounds. In: Ortega-Calvo JJ, Parsons JR (eds) Bioavailab org Chem Soils Sediments, 100th edn. Springer, Switzerland, pp 7–41
Rani S, Sud D (2015) Effect of temperature on adsorption-desorption behaviour of triazophos in Indian soils. Plant Soil Environ 61:36–42. https://doi.org/10.17221/704/2014-PSE
Ren X, Zeng G, Tang L, Wang J, Wan J, Liu Y et al (2018) Sorption, transport and biodegradation – an insight into bioavailability of persistent organic pollutants in soil. Sci Total Environ 610–611:1154–1163. https://doi.org/10.1016/j.scitotenv.2017.08.089
Rojas R, Morillo J, Usero J, Delgado-Moreno L, Gan J (2013) Enhancing soil sorption capacity of an agricultural soil by addition of three different organic wastes. Sci Total Environ 458–460:614–623. https://doi.org/10.1016/j.scitotenv.2013.04.032
Roman M, Van Dijk LH, Gutierrez L, Vanoppen M, Post JW, Wols BA et al (2019) Key physicochemical characteristics governing organic micropollutant adsorption and transport in ion-exchange membranes during reverse electrodialysis. Desalination 468:114084. https://doi.org/10.1016/j.desal.2019.114084
Sadegh-Zadeh F, Abd Wahid S, Jalili B (2017) Sorption, degradation and leaching of pesticides in soils amended with organic matter: a review. Adv Environ Technol 3:119–132. https://doi.org/10.22104/AET.2017.1740.1100
Saha P, Chowdhury S (2011) Insight into Adsorption Thermodynamics. In: Tadashi M (ed) Thermodynamics. InTech Open, Europe, pp 349–364.
Salman JM, Njoku VO, Hameed BH (2011) Bentazon and carbofuran adsorption onto date seed activated carbon: Kinetics and equilibrium. Chem Eng J 173:361–368. https://doi.org/10.1016/j.cej.2011.07.066
Sarkar S, Mukherjee I (2021) Effect of organic amendment on mobility behavior of flupyradifurone in two different Indian soils. Bull Environ Contam Toxicol 107:160–166. https://doi.org/10.1007/s00128-021-03209-4
Singh NS, Mukherjee I, Das SK, Varghese E (2018) Leaching of clothianidin in two different Indian soils: effect of organic amendment. Bull Environ Contam Toxicol 100:553–559. https://doi.org/10.1007/s00128-018-2290-z
Undabeytia T, Nir S, Rytwo G, Serban C, Morillo E, Maqueda C (2005) Modeling adsorption-desorption processes of Cu on montmorillonite and the effect of competitive adsorption with a cationic pesticide. In: Nützmann G, Viotti P, Aagaard P (eds) Reactive Transport in Soil and Groundwater: Processes and Models. Springer, Berlin Heidelberg, pp 79–91
Wauchope RD, Yeh S, Linders JBHJ, Kloskowski R, Tanaka K, Rubin B et al (2002) Pesticide soil sorption parameters: theory, measurement, uses, limitations and reliability. Pest Manag Sci 58:419–445. https://doi.org/10.1002/ps.489
Xiang L, Wang XD, Chen XH, Mo CH, Li YW, Li H et al (2019) Sorption mechanism, kinetics, and isotherms of Di- n-butyl phthalate to different soil particle-size fractions. J Agric Food Chem 67:4734–4745. https://doi.org/10.1021/acs.jafc.8b06357
Yadav S, Singh N (2021) Increased sorption of atrazine and fipronil in the sugarcane trash ash–mixed soils of Northern India. J Soil Sci Plant Nutr 21:1263–1276. https://doi.org/10.1007/s42729-021-00438-8
Yan XM, Shi BY, Lu JJ, Feng CH, Wang DS, Tang HX (2008) Adsorption and desorption of atrazine on carbon nanotubes. J Colloid Interface Sci 321:30–38. https://doi.org/10.1016/j.jcis.2008.01.047
Yang F, Wang M, Wang Z (2013) Sorption behavior of 17 phthalic acid esters on three soils: effects of pH and dissolved organic matter, sorption coefficient measurement and QSPR study. Chemosphere 93:82–89. https://doi.org/10.1016/j.chemosphere.2013.04.081
Yue L, Ge CJ, Feng D, Yu H, Deng H, Fu B (2017) Adsorption–desorption behavior of atrazine on agricultural soils in China. J Environ Sci (china) 57:180–189. https://doi.org/10.1016/j.jes.2016.11.002
Zhang P, Min L, Tang J, Rafiq MK, Sun H (2020) Sorption and degradation of imidacloprid and clothianidin in Chinese paddy soil and red soil amended with biochars. Biochar 2:329–341. https://doi.org/10.1007/s42773-020-00060-4
Zhu L, Chen B (2000) Sorption behavior of p-nitrophenol on the interface between anion−cation organobentonite and water. Environ Sci Technol 34:2997–3002. https://doi.org/10.1021/es991460z
Acknowledgements
The first author acknowledges ICAR-IARI for extending financial support extended in the form of fellowship. The author expresses his gratitude to Director, ICAR-IARI; Head of the Division of Agricultural Chemicals, ICAR-IARI for providing the facilities for research work and Dr. Abhishek Mandal, Scientist, Division of Agricultural Chemicals, ICAR-IARI for helping in manuscript preparation.
Funding
This work is financially supported by ICAR-Indian Agricultural Research Institute, New Delhi, India.
Author information
Authors and Affiliations
Contributions
SS performed the research work in wet lab, analyzed the data and prepared the first draft of the manuscript. AM helped in the improvement of the manuscript. IM conceptualized the research work and corrected the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Claudio Bini
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sarkar, S., Mukhopadhyay, A. & Mukherjee, I. A laboratory study on adsorption–desorption behavior of flupyradifurone in two Indian soils: effect of soil properties and organic amendment. J Soils Sediments 22, 2022–2035 (2022). https://doi.org/10.1007/s11368-022-03199-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-022-03199-8



