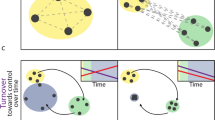
Abstract
Purpose
Given the vast microorganisms and infertile resources in the soil, nutritional resources would be a focal point of bacterial competition. Microbes could quickly capture the nutrition to outcompete their competitors or efficiently cross-feed to cooperate with other bacterial species. However, how interspecies trophic patterns shape the bacterial interactions in the co-occurrence network remains unclear. This study investigates the role of trophic patterns, containing niche overlap and nestedness, on the bacterial relationships in the co-occurrence network structure.
Materials and methods
We assessed the correlation between bacterial interactions and trophic patterns according to high-throughput techniques and culturomics. A co-occurrence network was constructed to obtain the relationship of OTUs. The substrate consumption patterns of culturable bacteria were used to calculate the interspecies trophic patterns.
Results and discussion
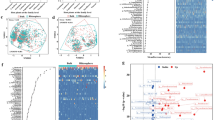
The co-occurrence network was constructed by the thirty-three culturable bacteria that matched 33 OTUs with 112 links in total. The negative correlation pairs possessed a higher niche overlap than the positive correlations pairs. However, negative and positive correlation pairs displayed no significant differences in the niche nestedness. Interestingly, we found substrate types could also influence microbial interactions. Elucidating the association between the trophic patterns and interspecies interaction is crucial for comprehending the resource’s role and contribution to the microbial community.
Conclusion
Together, these findings indicate that interspecies trophic patterns, such as niche overlap, could be a predictor to understand the bacterial relationships in the co-occurrence network.





Similar content being viewed by others
Data availability
Raw sequence files for each sample can be accessed through the NCBI SRA BioProject accession number PRJNA655807.
References
Aguiar MAMD, Newman EA, Pires MM, Yeakel JD, Boettiger C, Burkle LA, Gravel D, Guimarães PR, O’Donnell JL, Poisot T, Fortin MJ, Hembry DH (2019) Revealing biases in the sampling of ecological interaction networks. PeerJ 7:e7566. https://doi.org/10.7717/peerj.7566
Borthagaray AI, Arim M, Marquet PA (2014) Inferring species roles in metacommunity structure from species co-occurrence networks. Proc Biol Sci 281:20141425. https://doi.org/10.1098/rspb.2014.1425
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6:1621–1624. https://doi.org/10.1038/ismej.2012.8
Caporaso JG, Lauber CL, Walter WA, Berglyons D, Lozupone CA, Turnbaugh PJ, Fierer N, Knight R (2011) Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc Natl Acad Sci U S A 108:4516–4522. https://doi.org/10.1073/pnas.1000080107
Carr A, Diener C, Baliga NS, Gibbons SM (2019) Use and abuse of correlation analyses in microbial ecology. ISME J 13:2647–2655. https://doi.org/10.1038/s41396-019-0459-z
Dai LY, Zhang YH, Lin Y, Xie LM, Zhao SQ, Zhang ZM (2021) Thick roots and less microaggregates improve hydrological connectivity. Chemosphere 266:129008. https://doi.org/10.1016/j.chemosphere.2020.129008
Dal Bello M, Lee H, Goyal A, Gore J (2021) Resource-diversity relationships in bacterial communities reflect the network structure of microbial metabolism. Nat Ecol Evol 5:1424–1434. https://doi.org/10.1038/s41559-021-01535-8
Dong M, Zhao M, Shen Z, Deng X, Ou Y, Tao C, Liu H, Li R, Shen Q (2020) Biofertilizer application triggered microbial assembly in micro-aggregates associated with tomato bacterial wilt suppression. Biol Fert Soils 56:551–563. https://doi.org/10.1007/s00374-020-01459-8
Feng Y, Chen R, Hu JL, Zhao F, Wang JH, Chu HY, Zhang JB, Dolfing J, Lin XG (2015) Bacillus asahii comes to the fore in organic manure fertilized alkaline soils. Soil Biol Biochem 81:186–194. https://doi.org/10.1016/j.soilbio.2014.11.021
Finn DR, Yu J, Ilhan ZE, Fernandes VMC, Penton CR, Krajmalnik-Brown R, Garcia-Pichel F, Vogel TM (2020) MicroNiche: an R package for assessing microbial niche breadth and overlap from amplicon sequencing data. FEMS Microbiol Ecol 96.https://doi.org/10.1093/femsec/fiaa131
Fu W (2015) Niche dynamics of species in succession process in the Wetland of Yangtze Rivers Lower Reach, China. Plant Ecol Evol 148:43–51. https://doi.org/10.5091/plecevo.2015.956
Gralka M, Szabo R, Stocker R, Cordero OX (2020) Trophic interactions and the drivers of microbial community assembly. Curr Biol 30:1176–1188. https://doi.org/10.1016/j.cub.2020.08.007
Großkopf T, Soyer OS (2014) Synthetic microbial communities. Curr Opin Microbiol 18:72–77. https://doi.org/10.1016/j.mib.2014.02.002
Han S, Huang Q, Chen W (2020) Partitioning Nitrospira community structure and co-occurrence patterns in a long-term inorganic and organic fertilization soil. J Soil Sediment 21:1099–1108. https://doi.org/10.1007/s11368-020-02813-x
Hernandez-Soriano MC, Dalal RC, Warren FJ, Wang P, Green K, Tobin MJ, Menzies NW, Kopittke PM (2018) Soil Organic carbon stabilization: mapping carbon speciation from intact microaggregates. Environ Sci Technol 52(21):12275–12284. https://doi.org/10.1021/acs.est.8b03095
Hester ER, Jetten MSM, Welte CU, Lücker S (2019) Metabolic overlap in environmentally diverse microbial communities. Front Genet 10:989. https://doi.org/10.3389/fgene.2019.00989
Ji P, Wilson M (2002) Assessment of the importance of similarity in carbon source utilization profiles between the biological control agent and the pathogen in biological control of bacterial speck of tomato. Appl Environ Microbiol 68:4383–4389. https://doi.org/10.1128/AEM.68.9.4383-4389.2002
Levi SA (1998) Ecosystems and the biosphere as complex adaptive systems. Ecosystems 1:431–436. https://doi.org/10.1007/s100219900037
Liao H, Zhang Y, Zuo Q, Du B, Chen W, Wei D, Huang Q (2018) Contrasting responses of bacterial and fungal communities to aggregate-size fractions and long-term fertilizations in soils of northeastern China. Sci Total Environ 635:784–792. https://doi.org/10.1016/j.scitotenv.2018.04.168
Manlick PJ, Pauli JN (2020) Human disturbance increases trophic niche overlap in terrestrial carnivore communities. Proc Natl Acad Sci U S A 117:26842–26848. https://doi.org/10.1073/pnas.2012774117
Pandit SN, Kolasa J, Cottenie K (2009) Contrasts between habitat generalists and specialists: an empirical extension to the basic metacommunity framework. Ecology 90:2253–2262. https://doi.org/10.1890/08-0851.1
Pickett STA, Cadenasso ML (1995) Landscape ecology: spatial heterogeneity in ecological systems. Science 269:331–334. https://doi.org/10.1126/science.269.5222.331
Rabbi S, Daniel H, Lockwood P, Macdonald C, Pereg L, Tighe M, Wilson BR, Young IM (2016) Physical soil architectural traits are functionally linked to carbon decomposition and bacterial diversity. Sci Rep 6:33012. https://doi.org/10.1038/srep33012
Rodríguez-Gironés MA, Santamaría L (2006) A new algorithm to calculate the nestedness temperature of presence-absence matrices. J Biogeogr 33:924–935. https://doi.org/10.1111/j.1365-2699.2006.01444.x
Ranjard L, Poly F, Combrisson J, Richaume A, Gourbiere F, Thioulouse J, Nazaret S (2000) Heterogeneous cell density and genetic structure of bacterial pools associated with various soil microenvironments as determined by enumeration and DNA fingerprinting approach (RISA). Microb Ecol 39:263–272. https://doi.org/10.1007/s002480000032
Röttjers L, Faust K (2018) From hairballs to hypotheses-biological insights from microbial networks. FEMS Microbiol Rev 42:761–780. https://doi.org/10.1093/femsre/fuy030
Russel J, Røder HL, Madsen JS, Burmølle M, Sørensen SJ (2017) Antagonism correlates with metabolic similarity in diverse bacteria. Proc Natl Acad Sci U S A 114:10684–10688. https://doi.org/10.1073/pnas
Schnider-Keel U, Seematter A, Maurhofer M, Blumer C, Duffy B, Gigot-Bonnefoy C, Reimmann C, Notz R, Défago G, Haas D, Keel C (2000) Autoinduction of 2,4-diacetylphloroglucinol biosynthesis in the biocontrol agent Pseudomonas fluorescens CHA0 and repression by the bacterial metabolites salicylate and pyoluteorin. J Bacteriol 182:1215–1225. https://doi.org/10.1128/JB.182.5.1215-1225.2000
Schweizer SA, Hoeschen C, Schlüter S, Kögel-Knabner I, Mueller CW (2018) Rapid soil formation after glacial retreat shaped by spatial patterns of organic matter accrual in microaggregates. Glob Chang Biol 24(4):1637–1650. https://doi.org/10.1111/gcb.14014
Six J, Bossuyt H, Degryze S, Denef K (2004) A history of research on the link between (micro)aggregates, soil biota, and soil organic matter dynamics. Soil Tillage Res 79:7–31. https://doi.org/10.1016/j.still.2004.03.008
Thebault E, Fontaine C (2010) Stability of ecological communities and the architecture of mutualistic and trophic networks. Science 329:853–856. https://doi.org/10.1126/science.1188321
Tilman D (2004) Niche tradeoffs, neutrality, and community structure: a stochastic theory of resource competition, invasion, and community assembly. Proc Natl Acad Sci U S A 101:10854–10861. https://doi.org/10.1073/pnas.0403458101
Tisdall JM, Oades JM (1982) Organic-matter and water-stable aggregates in soils. J Soil Sci 33:141–163. https://doi.org/10.1111/j.1365-2389.1982.tb01755.x
Totsche KU, Amelung W, Gerzabek MH, Guggenberger G, Klumpp E, Knief C, Lehndorff E, Mikutta R, Peth S, Prechtel A, Ray N, Kögel-Knabner I (2018) Microaggregates in soils. J Plant Nutr Soil Sci 181:104–136. https://doi.org/10.1002/jpln.201600451
Wang L, Luo X, Liao H, Chen W, Wei D, Cai P, Huang Q (2018) Ureolytic microbial community is modulated by fertilization regimes and particle-size fractions in a Black soil of Northeastern China. Soil Biol Biochem 116:171–178. https://doi.org/10.1016/j.soilbio.2017.10.012
Wei Z, Yang T, Friman VP, Xu Y, Shen Q, Jousset A (2015) Trophic network architecture of root-associated bacterial communities determines pathogen invasion and plant health. Nat Commun 6:8413. https://doi.org/10.1038/ncomms9413
Wilpiszeski RL, Aufrecht JA, Retterer ST, Sullivan MB, Graham DE, Pierce EM, Zablocki OD, Palumbo AV, Elias DA (2019) Soil aggregate microbial communities: towards understanding microbiome interactions at biologically relevant scales. Appl Environ Microbiol 85:e00324-e419. https://doi.org/10.1128/AEM.00324-19
Xiong X, Xing Y, He J, Wang L, Shen Z, Chen W, Huang Q (2021) Keystone species determine the “selection mechanism” of multispecies biofilms for bacteria from soil aggregates. Sci Total Environ 773:145069. https://doi.org/10.1016/j.scitotenv.2021.145069
Funding
This work was supported by National Natural Science Foundation of China (42020104003 and 41830756) and National Key Research and Development Program of China (2018YFE0105600).
Author information
Authors and Affiliations
Contributions
XX, WC, and QH designed experiments and initiated research projects. WC and QH provided supervision, funding acquisition, conceptualization, and writing-review and editing. XX and WW performed experiments and analyzed data. All authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: Bin Ma
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiong, X., Wang, W., Xing, Y. et al. Niche overlap is a predictor of the interspecies correlations detected by microbial network analysis in soil micro-aggregates. J Soils Sediments 22, 1521–1529 (2022). https://doi.org/10.1007/s11368-022-03165-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-022-03165-4