Abstract
Purpose
Anaeromyxobacter is a typical representative genus of dissimilatory metal-reducing microbes. However, the community structure and metabolic function of Anaeromyxobacter have rarely been reported because of the limited number of Anaeromyxobacter isolations. Therefore, this study aimed to investigate the community structure and succession of Anaeromyxobacter in a Fe(III)-reducing enriched culture of paddy soils.
Materials and methods
A 40-day anaerobic incubation of paddy soils enriched with ferrihydrite and goethite was conducted to investigate the response of the community structure and succession of Anaeromyxobacter to iron oxide addition.
Results and discussion
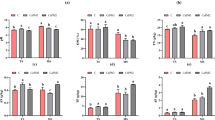
The dominant Anaeromyxobacter in paddy soils were potentially capable of Fe(III) reduction. Ferrihydrite enrichment increased the absolute abundance of Anaeromyxobacter by 0.01 × 108 to 3.2 × 108 copies g−1 soil, while goethite enrichment increased the absolute abundance of Anaeromyxobacter by 0.004 × 108 to 1.8 × 108 copies g−1 soil. Iron oxide enrichment significantly influenced the richness of Anaeromyxobacter during the later stages of incubation but had a negligible influence on the evenness. Nonetheless, Fe(II) accumulation was stimulated by ferrihydrite enrichment after paddy soil was incubated for 5 days, whereas goethite had a negligible effect on Fe(II) accumulation. Redundancy analysis revealed that Anaeromyxobacter community succession was closely correlated with the processes of Fe(III) reduction.
Conclusions
Exogenous ferrihydrite addition showed a greater influence than goethite on the Anaeromyxobacter community during anaerobic incubation of paddy soils. The difference in inherent amorphous iron oxide content in paddy soils was also decisive in the distinct community structure and succession of Anaeromyxobacter in paddy soils.







Similar content being viewed by others
References
Bonneville S, Cappellen PV, Behrends T (2004) Microbial reduction of iron (III) oxyhydroxides: effects of mineral solubility and availability. Chem Geol 212:255–268
Bonneville S, Behrends T, Van Cappellen P (2009) Solubility and dissimilatory reduction kinetics of iron (III) oxyhydroxides: a linear free energy relationship. Geochim Cosmochim Acta 73:5273–5282
Bosch J, Heister K, Hofmann T, Meckenstock RU (2010) Nanosized iron oxide colloids strongly enhance microbial iron reduction. Appl Environ Microbiol 76:184–189
Burgin AJ, Yang WH, Hamilton SK, Silver WL (2011) Beyond carbon and nitrogen: how the microbial energy economy couples elemental cycles in diverse ecosystems. Front Ecol Environ 9:44–52
Cornell RM, Schwertmann U (2003) The iron oxides: structure, properties, reactions, occurrences and uses. Wiley, Hoboken
Ding L, Su J, Xu H, Jia Z, Zhu Y (2015) Long-term nitrogen fertilization of paddy soil shifts iron-reducing microbial community revealed by RNA-13C-acetate probing coupled with pyrosequencing. ISME J 9:721–734
He J, Qu D (2008) Dissimilatory Fe (III) reduction characteristics of paddy soil extract cultures treated with glucose or fatty acids. J Environ Sci (China) 20:1103–1108
He Q, Sanford RA (2003) Characterization of Fe(III) reduction by chlororespiring Anaeromxyobacter dehalogenans. Appl Environ Microbiol 69:2712–2718
He Q, Yao K (2011) Impact of alternative electron acceptors on selenium (IV) reduction by Anaeromyxobacter dehalogenans. Bioresour Technol 102:3578–3580
He Z, Zhang Q, Feng Y, Luo H, Pan X, Gadd GM (2018) Microbiological and environmental significance of metal-dependent anaerobic oxidation of methane. Sci Total Environ 610:759–768
Hori T, Noll M, Igarashi Y, Friedrich MW, Conrad R (2007) Identification of acetate-assimilating microorganisms under methanogenic conditions in anoxic rice field soil by comparative stable isotope probing of RNA. Appl Environ Microbiol 73(1):101–109
Hori T, Müller A, Igarashi Y, Conrad R, Friedrich MW (2010) Identification of iron-reducing microorganisms in anoxic rice paddy soil by 13C-acetate probing. ISME J 4:267–278
Hori T, Aoyagi T, Itoh H, Narihiro T, Oikawa A, Suzuki K, Ogata A, Friedrich MW, Conrad R, Kamagata Y (2015) Isolation of microorganisms involved in reduction of crystalline iron(III) oxides in natural environments. Front Microbiol 6:1–16
Johnston SG, Burton ED, Aaso T, Tuckerman G (2014) Sulfur, iron and carbon cycling following hydrological restoration of acidic freshwater wetlands. Chem Geol 371:9–26
Li X, Liu T, Zhang N, Ren G, Li F, Li Y (2012a) Effect of Cr(VI) on Fe (III) reduction in three paddy soils from the Hani terrace field at high altitude. Appl Clay Sci 64:53–60
Li HJ, Peng JJ, Li HB (2012b) Diversity and characterization of potential H2-dependent Fe (III)-reducing bacteria in paddy soils. Pedosphere 22:673–680
Lin B, Hyacinthe C, Bonneville S, Braster M, Van Cappellen P, Röling WF (2007) Phylogenetic and physiological diversity of dissimilatory ferric iron reducers in sediments of the polluted Scheldt estuary, Northwest Europe. Environ Microbiol 9:1956–1968
Lovley DR (2006) Dissimilatory Fe(III)- and Mn(IV)-reducing prokaryotes. In: Dworkin M, Falkow S, Rosenberg E, Schleifer K-H, Stackebrandt E (eds) The Prokaryotes. Springer, New York, pp 635–658
Page AL, Miller RH, Keeny OR (1982) Chemical and microbiological properties. In: Buxton DR (ed) Methods of soil analysis, 2nd edn. American Society of Agronomy Press, Madison, pp 199–776
Qu D, Ratering S, Schnell S (2004) Microbial reduction of weakly crystalline iron(III) oxides and suppression of methanogenesis in paddy soil. Bull Environ Contam Toxicol 72:1172–1181
Roden EE (2003) Fe (III) oxide reactivity toward biological versus chemical reduction. Environ Sci Technol 37:1319–1324
Roden EE, Urrutia MM (2002) Influence of biogenic Fe(II) on bacterial crystalline Fe (III) oxide reduction. Geomicrobiol J 19:209–251
Sanford RA, Wu Q, Sung Y, Thomas SH, Amos BK, Prince EK, Loffler FE (2007) Hexavalent uranium supports growth of anaeromyxobacter dehalogenans and geobacter spp. with lower than predicted biomass yields. Environ Microbiol 9:2885–2893
Sanford RA, Wagner DD, Qingzhong W, Chee-Sanford JC, Thomas SH, Claribel CG, Gina R, Arturo MD, Krishnani KK, Ritalahti KM (2012) Unexpected nondenitrifier nitrous oxide reductase gene diversity and abundance in soils. Proc Natl Acad Sci U S A 109:19709–19714
Schwertmann U, Cornell RM (1991) Iron oxides in the laboratory: preparation and characterization, 2nd edn. WILEY-VCH: Verlagsgesellschaft, Weinheim
Strycharz SM, Gannon SM, Boles AR, Franks AE, Nevin KP, Lovley DR (2010) Reductive dechlorination of 2-chlorophenol by Anaeromyxobacter dehalogenans with an electrode serving as the electron donor. Environ MicrobioI Rep 2:289–294
Thomas SH, Padilla-Crespo E, Jardine PM, Sanford RA, Löffler FE (2009) Diversity and distribution of Anaeromyxobacter strains in a uranium-contaminated subsurface environment with a nonuniform groundwater flow. Appl Environ Microbiol 75:3679–3687
Tonomura M, Ehara A, Suzuki H, Amachi S (2015) Draft genome sequence of Anaeromyxobacter sp. strain PSR-1, an arsenate-respiring bacterium isolated from arsenic-contaminated soil. Genome Announc 3:e00472–e00415
Treude N, Rosencrantz D, Liesack W, Schnell S (2003) Strain FAc12, a dissimilatory iron-reducing member of the Anaeromyxobacter subgroup of Myxococcales. FEMS Microbiol Ecol 44:261–269
Unger IM, Kennedy AC, Muzika R (2009) Flooding effects on soil microbial communities. Appl Soil Ecol 42:1–8
Weber KA, Achenbach LA, Coates JD (2006) Microorganisms pumping iron: anaerobic microbial iron oxidation and reduction. Nat Rev Microbiol 4:752–764
Wu Q, Sanford RA, Löffler FE (2006) Uranium(VI) reduction by Anaeromyxobacter dehalogenans strain 2CP-C. Appl Environ Microbiol 72:3608–3614
Xu Y, He Y, Feng X, Liang L, Xu J, Brookes PC, Wu J (2014) Enhanced abiotic and biotic contributions to dechlorination of pentachlorophenol during Fe (III) reduction by an iron-reducing bacterium Clostridium beijerinckii Z. Sci Total Environ 473:215–223
You J, Xia S, Wang B, Qu D (2011) Effect of flooding time on community structure and abundance of Geobacteraceae in paddy soil. Wei Sheng Wu Xue Bao 51:796–804
Yuan HY, Ding LJ, Wang N, Chen SC, Deng Y, Li XM, Zhu YG (2016) Geographic distance and amorphous iron affect the abundance and distribution of Geobacteraceae in paddy soils in China. J Soils Sediments 16:2657–2665
Zhang J, Zhang Y, Chang J, Quan X, Li Q (2013) Biological sulfate reduction in the acidogenic phase of anaerobic digestion under dissimilatory Fe (III)–reducing conditions. Water Res 47:2033–2040
Zhu C, Xia SH, Wang BL, Qu D (2011) Variation of Anaeromyxobacter community structure and abundance in paddy soil slurry over flooding time. Afr J Agric Res 6:6107–6118
Acknowledgments
The authors are also grateful for the anonymous reviewers for their quality comments. We thank Alex Boon, PhD, from Liwen Bianji, Edanz Editing China (www.liwenbianji.cn/ac), for editing the English text of a draft of this manuscript.
Funding
Support for this paper was provided by the National Natural Science Foundation of China (Grant No: 41571239).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Jizheng He
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 166 kb).
Rights and permissions
About this article
Cite this article
Wang, K., Jia, R., Li, L. et al. Community structure of Anaeromyxobacter in Fe(III) reducing enriched cultures of paddy soils. J Soils Sediments 20, 1621–1631 (2020). https://doi.org/10.1007/s11368-019-02529-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-019-02529-7