Abstract
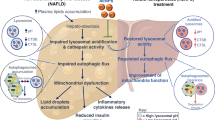
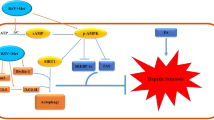
Cellular senescence is an important risk factor in the development of hepatic steatosis. Senolytics present therapeutic effects on age-related hepatic steatosis without eliminating senescent hepatocytes directly. Therefore, it highlights the need to find senolytics’ therapeutic targets. Dysfunction of adipose tissue underlies the critical pathogenesis of lipotoxicity in the liver. However, the correlation between adipose tissue and hepatic steatosis during aging and its underlying molecular mechanism remains poorly understood. We explored the correlation between white adipose tissue (WAT) and the liver during aging and evaluated the effect of lipolysis of aged WAT on hepatic steatosis and hepatocyte senescence. We screened out the ideal senolytics for WAT and developed a WAT-targeted delivery system for senotherapy. We assessed senescence and lipolysis of WAT and hepatic lipid accumulation after treatment. The results displayed that aging accelerated cellular senescence and facilitated lipolysis of WAT. Free fatty acids (FFAs) generated by WAT during aging enhanced hepatic steatosis and induced hepatocyte senescence. The combined usage of dasatinib and quercetin was screened out as the ideal senolytics to eliminate senescent cells in WAT. To minimize non-specific distribution and enhance the effectiveness of senolytics, liposomes decorated with WAT affinity peptide P3 were constructed for senotherapy in vivo. In vivo study, WAT-targeted treatment eliminated senescent cells in WAT and reduced lipolysis, resulting in the alleviation of hepatic lipid accumulation and hepatocyte senescence when compared to non-targeted treatment, providing a novel tissue-targeted, effective and safe senotherapy for age-related hepatic steatosis.







Similar content being viewed by others
Abbreviations
- WAT:
-
White adipose tissue
- SCs:
-
Senescent cells
- SASP:
-
Senescence-associated secretory phenotype
- D:
-
Dasatinib
- Q:
-
Quercetin
- FFA:
-
Free fatty acid
- ALT:
-
Alanine aminotransamine
- TG:
-
Triglyceride
- TCH:
-
Total cholesterol
- H2O2 :
-
Hydrogen peroxide
- BSA:
-
Bovine serum albumin
- SA-β-gal:
-
Senescence-associated β-galactosidase
- HPLC:
-
High-performance liquid chromatography
- LE:
-
Loading efficiency
- EE:
-
Entrapment efficiency
- Cpt1α:
-
Carnitine palmitoyltransferase 1α
- Ucp2:
-
Uncoupling protein 2
- Accα:
-
Activated acetyl-CoA carboxylase
- Acox1:
-
Acyl-CoA oxidase type 1
- Scd1:
-
Stearoyl-CoA desaturase 1
References
da Costa JP, et al. A synopsis on aging-theories, mechanisms and future prospects. Ageing Res Rev. 2016;29:90–112.
Palmer AK, Kirkland JL. Aging and adipose tissue: potential interventions for diabetes and regenerative medicine. Exp Gerontol. 2016;86:97–105.
Childs BG, et al. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015;21(12):1424–35.
van Deursen JM. The role of senescent cells in ageing. Nature. 2014;509(7501):439–46.
He S, Sharpless NE. Senescence in health and disease. Cell. 2017;169(6):1000–11.
Sharpless NE, Sherr CJ. Forging a signature of in vivo senescence. Nat Rev Cancer. 2015;15(7):397–408.
Coppe JP, et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6(12):2853–68.
Collado M, Blasco MA, Serrano M. Cellular senescence in cancer and aging. Cell. 2007;130(2):223–33.
Kim IH, Kisseleva T, Brenner DA. Aging and liver disease. Curr Opin Gastroenterol. 2015;31(3):184–91.
Ramirez T, et al. Aging aggravates alcoholic liver injury and fibrosis in mice by downregulating sirtuin 1 expression. J Hepatol. 2017;66(3):601–9.
Alqahtani SA, Schattenberg JM. NAFLD in the elderly. Clin Interv Aging. 2021;16:1633–49.
Cree MG, et al. Intramuscular and liver triglycerides are increased in the elderly. J Clin Endocrinol Metab. 2004;89(8):3864–71.
Fan JG, et al. Prevalence of and risk factors for fatty liver in a general population of Shanghai, China. J Hepatol. 2005;43(3):508–14.
Gong Z, et al. Hepatic lipid metabolism and non-alcoholic fatty liver disease in aging. Mol Cell Endocrinol. 2017;455:115–30.
Raza S, et al. Current treatment paradigms and emerging therapies for NAFLD/NASH. Front Biosci (Landmark Ed). 2021;26(2):206–37.
Triana-Martinez F, et al. Identification and characterization of cardiac glycosides as senolytic compounds. Nat Commun. 2019;10(1):4731.
Roos CM, et al. Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell. 2016;15(5):973–7.
Ogrodnik M, et al. Obesity-Induced cellular senescence drives anxiety and impairs neurogenesis. Cell Metab. 2019;29(5):1233.
Ogrodnik M, et al. Cellular senescence drives age-dependent hepatic steatosis. Nat Commun. 2017;8:15691.
Raffaele M, et al. Mild exacerbation of obesity- and age-dependent liver disease progression by senolytic cocktail dasatinib + quercetin. Cell Commun Signal. 2021;19(1):44.
Bedossa P, et al. Systematic review of bariatric surgery liver biopsies clarifies the natural history of liver disease in patients with severe obesity. Gut. 2017;66(9):1688–96.
Roden M, Shulman GI. The integrative biology of type 2 diabetes. Nature. 2019;576(7785):51–60.
Tchkonia T, et al. Fat tissue, aging, and cellular senescence. Aging Cell. 2010;9(5):667–84.
Frasca D, Blomberg BB. Adipose tissue, immune aging, and cellular senescence. Semin Immunopathol. 2020;42(5):573–87.
Lutz W, Sanderson W, Scherbov S. The coming acceleration of global population ageing. Nature. 2008;451(7179):716–9.
Green CJ, Hodson L. The influence of dietary fat on liver fat accumulation. Nutrients. 2014;6(11):5018–33.
Li X, Wang H. Multiple organs involved in the pathogenesis of non-alcoholic fatty liver disease. Cell Biosci. 2020;10(1):140.
Liu YY, et al. Hydrogen sulfide protects SH-SY5Y neuronal cells against d-galactose induced cell injury by suppression of advanced glycation end products formation and oxidative stress. Neurochem Int. 2013;62(5):603–9.
Duan J, et al. Irreversible cellular senescence induced by prolonged exposure to H2O2 involves DNA-damage-and-repair genes and telomere shortening. Int J Biochem Cell Biol. 2005;37(7):1407–20.
Zhang H. Thin-film hydration followed by extrusion method for liposome preparation. Methods Mol Biol. 2017;1522:17–22.
Huang X, et al. ICAM-1-targeted liposomes loaded with liver X receptor agonists suppress PDGF-induced proliferation of vascular smooth muscle cells. Nanoscale Res Lett. 2017;12(1):322.
Tang X, et al. Improved antifungal activity of amphotericin B-loaded TPGS-b-(PCL-ran-PGA) nanoparticles. Int J Clin Exp Med. 2015;8(4):5150–62.
Azman KF, Zakaria R. D-Galactose-induced accelerated aging model: an overview. Biogerontology. 2019;20(6):763–82.
Lee GH, et al. Anthocyanins attenuate endothelial dysfunction through regulation of uncoupling of nitric oxide synthase in aged rats. Aging Cell. 2020;19(12):e13279.
Toussaint O, Medrano EE, von Zglinicki T. Cellular and molecular mechanisms of stress-induced premature senescence (SIPS) of human diploid fibroblasts and melanocytes. Exp Gerontol. 2000;35(8):927–45.
Yang X, et al. The G(0)/G(1) switch gene 2 regulates adipose lipolysis through association with adipose triglyceride lipase. Cell Metab. 2010;11(3):194–205.
Kirkland JL, et al. The clinical potential of senolytic drugs. J Am Geriatr Soc. 2017;65(10):2297–301.
Zhang L, et al. Recent advances in the discovery of senolytics. Mech Ageing Dev. 2021;200:111587.
Xu M, et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018;24(8):1246–56.
Kolonin MG, et al. Reversal of obesity by targeted ablation of adipose tissue. Nat Med. 2004;10(6):625–32.
Xue Y, et al. Preventing diet-induced obesity in mice by adipose tissue transformation and angiogenesis using targeted nanoparticles. Proc Natl Acad Sci U S A. 2016;113(20):5552–7.
Medina J, Moreno-Otero R. Pathophysiological basis for antioxidant therapy in chronic liver disease. Drugs. 2005;65(17):2445–61.
Mari M, et al. Redox control of liver function in health and disease. Antioxid Redox Signal. 2010;12(11):1295–331.
Rolo AP, Teodoro JS, Palmeira CM. Role of oxidative stress in the pathogenesis of nonalcoholic steatohepatitis. Free Radic Biol Med. 2012;52(1):59–69.
Papatheodoridi AM, et al. The role of senescence in the development of nonalcoholic fatty liver disease and progression to nonalcoholic steatohepatitis. Hepatology. 2020;71(1):363–74.
Fernandez-Real JM, et al. Circulating interleukin 6 levels, blood pressure, and insulin sensitivity in apparently healthy men and women. J Clin Endocrinol Metab. 2001;86(3):1154–9.
du Plessis J, et al. Association of adipose tissue inflammation with histologic severity of nonalcoholic fatty liver disease. Gastroenterology. 2015;149(3):635–48 e14.
Baker DJ, et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479(7372):232–6.
Xu M, et al. Targeting senescent cells enhances adipogenesis and metabolic function in old age. Elife. 2015;4:e12997.
Farr JN, et al. Targeting cellular senescence prevents age-related bone loss in mice. Nat Med. 2017;23(9):1072–9.
Kirkland JL, Tchkonia T. Cellular senescence: a translational perspective. EBioMedicine. 2017;21:21–8.
Day CP, James OF. Steatohepatitis: a tale of two "hits"? Gastroenterology. 1998;114(4):842–5.
Liu J, et al. The role of fibroblast growth factor 21 in the pathogenesis of non-alcoholic fatty liver disease and implications for therapy. Metabolism. 2015;64(3):380–90.
Zhu Y, et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015;14(4):644–58.
Garcia-Gomez A, et al. Dasatinib as a bone-modifying agent: anabolic and anti-resorptive effects. PLoS One. 2012;7(4):e34914.
Vandyke K, et al. The tyrosine kinase inhibitor dasatinib dysregulates bone remodeling through inhibition of osteoclasts in vivo. J Bone Miner Res. 2010;25(8):1759–70.
Zoico E, et al. Senolytic effects of quercetin in an in vitro model of pre-adipocytes and adipocytes induced senescence. Sci Rep. 2021;11(1):23237.
Hartmann JT, et al. Tyrosine kinase inhibitors - a review on pharmacology, metabolism and side effects. Curr Drug Metab. 2009;10(5):470–81.
Munoz-Espin D, et al. Programmed cell senescence during mammalian embryonic development. Cell. 2013;155(5):1104–18.
Demaria M, et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell. 2014;31(6):722–33.
Cox LS, Redman C. The role of cellular senescence in ageing of the placenta. Placenta. 2017;52:139–45.
Allen TM. Ligand-targeted therapeutics in anticancer therapy. Nat Rev Cancer. 2002;2(10):750–63.
Garcia-Pinel B, Porras-Alcalá C, Ortega-Rodríguez A, Sarabia F, Prados J, Melguizo C, et al. Lipid-based nanoparticles: application and recent advances in cancer treatment. Nanomaterials. 2019;9(4):638. https://doi.org/10.3390/nano9040638.
Tenchov R, et al. Lipid nanoparticles horizontal line from liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano. 2021;15(11):16982–7015.
Funding
This work was supported by grants from the National Key Research and Development Program of China (2021YFA1100603), the National Natural Science Foundation of China (82071092, U21A20369), the Sichuan Science and Technology program (2023YFS0056, 2022YFS0126), and the Fundamental Research Funds for the Central Universities (2021SCU12140).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval statement
All animal procedures were performed under the relevant guidelines and approved by the Institutional Animal Care and Use Committee (IACUC) at West China Hospital of Stomatology (WCHSIRB-D-2019-034).
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 10.2 mb)
About this article
Cite this article
Tang, Q., Xing, X., Huang, H. et al. Eliminating senescent cells by white adipose tissue–targeted senotherapy alleviates age-related hepatic steatosis through decreasing lipolysis. GeroScience 46, 3149–3167 (2024). https://doi.org/10.1007/s11357-024-01068-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-024-01068-5