Abstract
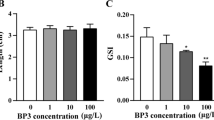
Increased availability of cannabis and cannabinoid-containing products necessitates the need for an understanding of how these substances influence aging. In this study, zebrafish (Danio rerio) were exposed to different concentrations of THC (0.08, 0.4, 2 μM) during embryonic-larval development and the effects on aging were measured 30 months later and in the offspring of the exposed fish (F1 generation). Exposure to 0.08 μM THC resulted in increased male survival at 30 months of age. As the concentration of THC increased, this protective effect was lost. Treatment with the lowest concentration of THC also significantly increased egg production, while higher concentrations resulted in impaired fecundity. Treatment with the lowest dose of THC significantly reduced wet weight, the incidence of kyphosis, and the expression of several senescence and inflammatory markers (p16ink4ab, tnfα, il-1β, il-6, pparα and pparγ) in the liver, but not at higher doses indicating a biphasic or hormetic effect. Exposure to THC did not affect the age-related reductions in locomotor behavior. Within the F1 generation, many of these changes were not observed. However, the reduction in fecundity due to THC exposure was worse in the F1 generation because offspring whose parents received high dose of THC were completely unable to reproduce. Together, our results demonstrate that a developmental exposure to THC can cause significant effects on longevity and healthspan of zebrafish in a biphasic manner.




Similar content being viewed by others
References
Ahrnsbrak R, Bose J, Hedden SL, Lipari RN, Park-Lee E. Key substance use and mental health indicators results from the 2016 national survey in the United States: on drug use and health; 2017. https://www.samhsa.gov/data/sites/default/files/NSDUH-FFR1-2016/NSDUH-FFR1-2016.htm.
Assaf F, Fishbein M, Gafni M, Keren O, Sarne Y. Pre- and post-conditioning treatment with an ultra-low dose of Δ9-tetrahydrocannabinol (THC) protects against pentylenetetrazole (PTZ)-induced cognitive damage. Behav Brain Res. 2011;220:194–201. https://doi.org/10.1016/j.bbr.2011.02.005.
Austad SN. Sex differences in health and aging: a dialog between the brain and gonad? GeroScience. 2019;41:267–73. https://doi.org/10.1007/s11357-019-00081-3.
Bada HS, Das A, Bauer CR, Shankaran S, Lester BM, Gard CC, et al. Low birth weight and preterm births: etiologic fraction attributable to prenatal drug exposure. J Perinatol. 2005;25:631–7. https://doi.org/10.1038/sj.jp.7211378.
Bara A, Manduca A, Bernabeu A, Borsoi M, Serviado M, Lassalle O, et al. Sex-dependent effects of in utero cannabinoid exposure on cortical function. Elife. 2018;7:1–31. https://doi.org/10.7554/eLife.36234.
Barker DJP. The origins of the developmental origins theory. J Intern Med. 2007;261:412–7. https://doi.org/10.1111/j.1365-2796.2007.01809.x.
Berger J, Moller DE. The mechanisms of action of PPARs. Annu Rev Med. 2002;53:409–35. https://doi.org/10.1146/annurev.med.53.082901.104018.
Bilkei-Gorzo A, Albayram O, Draffehn A, Michel K, Piyanova A, Oppenheimer H, et al. A chronic low dose of Δ9-tetrahydrocannabinol (THC) restores cognitive function in old mice. Nat Med. 2017;23:782–7. https://doi.org/10.1038/nm.4311.
Bollaerts I, Van Houcke J, Andries L, De Groef L, Moons L. Neuroinflammation as fuel for axonal regeneration in the injured vertebrate central nervous system. Mediat Inflamm. 2017;2017:1–14. https://doi.org/10.1155/2017/9478542.
Bouaboula M, Hilairet S, Marchand J, Fajas L, Le Fur G, Casellas P. Anandamide induced PPARγ transcriptional activation and 3T3-L1 preadipocyte differentiation. Eur J Pharmacol. 2005;517:174–81. https://doi.org/10.1016/j.ejphar.2005.05.032.
Brents LK. Marijuana, the endocannabinoid system and the female reproductive system. Yale J Biol Med. 2016;89:175–91.
Calabrese EJ, Mattson MP. How does hormesis impact biology, toxicology, and medicine? Npj Aging Mech Dis. 2017;3:1–8. https://doi.org/10.1038/s41514-017-0013-z.
Cao C, Li Y, Liu H, Bai G, Mayl J, Lin X, et al. The potential therapeutic effects of THC on Alzheimer’s disease. J Alzheimers Dis. 2014;42:973–84. https://doi.org/10.3233/JAD-140093.
Carty DR, Thornton C, Gledhill JH, Willett KL. Developmental effects of cannabidiol and Δ9-tetrahydrocannabinol in zebrafish. Toxicol Sci. 2018;162:137–45. https://doi.org/10.1093/toxsci/kfx232.
Carty DR, Miller ZS, Thornton C, Pandelides Z, Kutchma ML, Willett KL. Multigenerational consequences of early-life cannabinoid exposure in zebrafish. Toxicol Appl Pharmacol. 2019;364:133–43. https://doi.org/10.1016/j.taap.2018.12.021.
Corrales J, Fang X, Thornton C, Mei W, Barbazuk WB, Duke M, et al. Effects on specific promoter DNA methylation in zebrafish embryos and larvae following benzo[a]pyrene exposure. Comp Biochem Physiol C Toxicol Pharmacol. 2014;163:37–46. https://doi.org/10.1016/j.cbpc.2014.02.005.
Dalterio SL, DeRooij DG. Maternal cannabinoid exposure effects on spermatogenesis in male offspring. Int J Androl. 1986;9:250–8. https://doi.org/10.1111/j.1365-2605.1986.tb00888.x.
Dalterio S, Steger R, Mayfield D, Bartke A. Early cannabinoid exposure influences neuroendocrine and reproductive functions in male mice: I prenatal exposure. Pharmacol Biochem Behav. 1984;20:107–13. https://doi.org/10.1016/0091-3057(84)90110-2.
Desai A, Mark K, Terplan M. Marijuana use and pregnancy prevalence, associated behaviors, and birth outcomes. Obstet Gynecol. 2014;123:46S–111. https://doi.org/10.1007/s00737-015-0529-9.
El-Mohandes A, Herman AA, El-Khorazaty MN, Katta PS, White D, Grylack L. Prenatal care reduces the impact of illicit drug use on perinatal outcomes. J Perinatol. 2003;23:354–60. https://doi.org/10.1038/sj.jp.7210933.
Feltes BC, de Faria Poloni J, Bonatto D. Development and aging: two opposite but complementary phenomena. Interdiscip Top Gerontol. 2015;40:74–84. https://doi.org/10.1159/000364932.
Fergusson DM, Horwood LJ, Northstone K. Maternal use of cannabis and pregnancy outcome. BJOG Int J Obstet Gynaecol. 2002;109:21–7. https://doi.org/10.1111/j.1471-0528.2002.01020.x.
Fernández-Ruiz J, Gómez-Ruiz M, García C, Hernández M, Ramos JA. Modeling neurodegenerative disorders for developing cannabinoid-based neuroprotective therapies. Methods Enzymol. 2017;593:175–98. https://doi.org/10.1016/bs.mie.2017.06.021.
Fish EW, Murdaugh LB, Zhang C, Boschen KE, Boa-Amponsem O, Mendoza-Romero HN, et al. Cannabinoids exacerbate alcohol teratogenesis by a CB1-hedgehog interaction. Sci Rep. 2019;9:1–16. https://doi.org/10.1038/s41598-019-52336-w.
Fishbein M, Gov S, Assaf F, Gafni M, Keren O, Sarne Y. Long-term behavioral and biochemical effects of an ultra-low dose of Δ9-tetrahydrocannabinol (THC): neuroprotection and ERK signaling. Exp Brain Res. 2012;221:437–48. https://doi.org/10.1007/s00221-012-3186-5.
Fishbein-Kaminietsky M, Gafni M, Sarne Y. Ultralow doses of cannabinoid drugs protect the mouse brain from inflammation-induced cognitive damage. J Neurosci Res. 2014;92:1669–77. https://doi.org/10.1002/jnr.23452.
Gerhold DL, Franklin L, Jiang G, Zhihua LI, Jian XU, Meiqing LU, et al. Gene expression profile of adipocyte differentiation and its regulation by peroxisome proliferator-activated receptor-γ agonists. Endocrinology. 2002;143:2106–18. https://doi.org/10.1210/endo.143.6.8842.
Gidday JM. Extending injury- and disease-resistant CNS phenotypes by repetitive epigenetic conditioning. Front Neurol. 2015;6:1–7. https://doi.org/10.3389/fneur.2015.00042.
Haugen AC, Schug TT, Collman G, Heindel JJ. Evolution of DOHaD: the impact of environmental health sciences. J Dev Orig Health Dis. 2015;6:55–64. https://doi.org/10.1017/S2040174414000580.
Hayatbakhsh MR, Flenady VJ, Gibbons KS, Kingsbury AM, Hurrion E, Mamun AA, et al. Birth outcomes associated with cannabis use before and during pregnancy. Pediatr Res. 2012;71:215–9. https://doi.org/10.1038/pr.2011.25.
Hegde VL, Hegde S, Cravatt BF, Hofseth LJ, Nagarkatti M, Nagarkatti PS. Attenuation of experimental autoimmune hepatitis by exogenous and endogenous cannabinoids: involvement of regulatory T cells. Mol Pharmacol. 2008;74:20–33. https://doi.org/10.1124/mol.108.047035.
Hodges EL, Ashpole NM. Aging circadian rhythms and cannabinoids. Neurobiol Aging. 2019;79:110–8. https://doi.org/10.1016/j.neurobiolaging.2019.03.008.
Hurd YL, Wang X, Anderson V, Beck O, Minkoff H, Dow-Edwards D. Marijuana impairs growth in mid-gestation fetuses. Neurotoxicol Teratol. 2005;27:221–9. https://doi.org/10.1016/j.ntt.2004.11.002.
Hutchings DE, Martin BR, Gamagaris Z, Miller N, Fico T. Plasma concentrations of delta-9-tetrahydrocannabinol in dams and fetuses following acute or multiple prenatal dosing in rats. Life Sci. 1989;44:697–701. https://doi.org/10.1016/0024-3205(89)90380-9.
Jura M, Kozak LP. Obesity and related consequences to ageing. Age (Omaha). 2016. https://doi.org/10.1007/s11357-016-9884-3.
Klein TW, Friedman H, Specter S. Marijuana, immunity and infection. J Neuroimmunol. 1998;83:102–15. https://doi.org/10.1016/S0165-5728(97)00226-9.
Ko JY, Farr SL, Tong VT, Creanga AA, Callaghan WM. Prevalence and patterns of marijuana use among pregnant and nonpregnant women of reproductive age. Am J Obstet Gynecol. 2015;213:201.e1–201.e10. https://doi.org/10.1016/j.ajog.2015.03.021.
Lewis SEM, Rapino C, Di Tommaso M, Pucci M, Battista N, Paro R, et al. Differences in the endocannabinoid system of sperm from fertile and infertile men. PLoS One. 2012;7:1–8. https://doi.org/10.1371/journal.pone.0047704.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods. 2001;25:402–8. https://doi.org/10.1006/meth.2001.1262.
Marusich JA, Craft RM, Lefever TW, Wiley JL. The impact of gonadal hormones on cannabinoid dependence. Exp Clin Psychopharmacol. 2015;23:206–16. https://doi.org/10.1037/pha0000027.
Morgan DJ, Muller CH, Murataeva NA, Davis BJ, Mackie K. Δ 9-tetrahydrocannabinol (Δ 9-THC) attenuates mouse sperm motility and male fecundity. Br J Pharmacol. 2012;165:2575–83. https://doi.org/10.1111/j.1476-5381.2011.01506.x.
Nagarkatti P, Pandey R, Rieder SA, Hegde VL, Nagarkatti M. Cannabinoids as novel anti-inflammatory drugs. Future Med Chem. 2009;1:1333–49. https://doi.org/10.4155/fmc.09.93.
Pandelides Z, Thornton C, Faruque AS, Whitehead AP, Willett KL, Ashpole NM. Developmental exposure to cannabidiol (CBD) alters longevity and health span of zebrafish (Danio rerio). GeroScience. 2020. https://doi.org/10.1007/s11357-020-00182-4.
Parichy DM, Elizondo MR, Mills MG, Gordon TN, Engeszer RE. Normal table of post-embryonic zebrafish development: staging by externally visible anatomy of the living fish. Dev Dyn. 2009;238:9–12. https://doi.org/10.1002/dvdy.22113.
Payne KS, Mazur DJ, Hotaling JM, Pastuszak AW. Cannabis and male fertility: a systematic review. J Urol. 2019;202:674–81. https://doi.org/10.1097/JU.0000000000000248.
Pazhakh V, Lieschke GJ. Hematopoietic growth factors: the scenario in zebrafish. Growth Factors. 2018;36:196–212. https://doi.org/10.1080/08977194.2019.1567506.
Ramírez BG, Blázquez C, Gómez Del Pulgar T, Guzmán M, De Ceballos ML. Prevention of Alzheimer’s disease pathology by cannabinoids: neuroprotection mediated by blockade of microglial activation. J Neurosci. 2005;25:1904–13. https://doi.org/10.1523/JNEUROSCI.4540-04.2005.
Reed, B., Jennings, M., 2011. Guidance on the housing and care of zebrafish (Danio rerio), Research Animals Department, Science Group, RSPCA https://doi.org/10.1111/evo.12990.
Richardson JR, Caudle WM, Wang M, Dean ED, Pennell KD, Miller GW. Developmental exposure to the pesticide dieldrin alters the dopamine system and increases neurotoxicity in an animal model of Parkinson’s disease. FASEB J. 2006;20. https://doi.org/10.1096/fj.06-5864fje.
Royce GH, Brown-Borg HM, Deepa SS. The potential role of necroptosis in inflammaging and aging. GeroScience. 2019;41:795–811. https://doi.org/10.1007/s11357-019-00131-w.
Schneider CA, Rasband WS, Eliceiri KW. NIH image to imageJ: 25 years of image analysis. Nat Methods. 2012;9:671–5. https://doi.org/10.1038/nmeth.2089.
van Gelder MMHJ, Reefhuis J, Caton AR, Werler MM, Druschel CM, Roeleveld N. Characteristics of pregnant illicit drug users and associations between cannabis use and perinatal outcome in a population-based study. Drug Alcohol Depend. 2010;109:243–7. https://doi.org/10.1016/j.drugalcdep.2010.01.007.
Van Houcke J, De Groef L, Dekeyster E, Moons L. The zebrafish as a gerontology model in nervous system aging, disease, and repair. Ageing Res Rev. 2015;24:358–68. https://doi.org/10.1016/j.arr.2015.10.004.
Varvel SA, Hamm RJ, Martin BR, Lichtman AH. Differential effects of Δ9-THC on spatial reference and working memory in mice. Psychopharmacology. 2001;157:142–50. https://doi.org/10.1007/s002130100780.
Volkow ND, Han B, Compton WM, McCance-Katz EF. Self-reported medical and nonmedical cannabis use among pregnant women in the United States. JAMA - J Am Med Assoc. 2019;322:167–9. https://doi.org/10.1001/jama.2019.7982.
Wakley AA, Wiley JL, Craft RM. Sex differences in antinociceptive tolerance to delta-9-tetrahydrocannabinol in the rat. Drug Alcohol Depend. 2014;143:22–8. https://doi.org/10.1016/j.drugalcdep.2014.07.029.
Zawia NH, Basha MR. Environmental risk factors and the developmental basis for Alzheimer’s disease. Rev Neurosci. 2005;16:325–37. https://doi.org/10.1515/REVNEURO.2005.16.4.325.
Acknowledgments
The authors would like to thank Dennis R. Carty for conducting the initial exposure, as well as Kennedy E. Dickson, Mary-Beth Gillespie, James H. Gledhill, Haley Watts, and Bailey Westling for their help during sampling.
Funding
This work was supported by the National Institute on Drug Abuse R21DA044473-01 grant awarded to KW and P30GM122733 to NMA and KW.
Author information
Authors and Affiliations
Contributions
KW, NA, and ZP conceived and designed the experiments; NA and ZP analyzed the data; AF, AW, CT, KL, NA, and ZP performed the experiments; KW, NA and ZP prepared the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 418 kb)
About this article
Cite this article
Pandelides, Z., Thornton, C., Lovitt, K.G. et al. Developmental exposure to Δ9-tetrahydrocannabinol (THC) causes biphasic effects on longevity, inflammation, and reproduction in aged zebrafish (Danio rerio). GeroScience 42, 923–936 (2020). https://doi.org/10.1007/s11357-020-00175-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-020-00175-3