Abstract
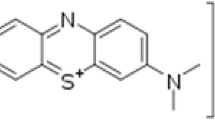
In the present studies, the synthesis of pure ZnO nanoparticles and Mg and S-doped ZnO particles were carried out using a non-aqueous sol–gel method. The synthesized nanoparticles (NPs) are characterized using XRD, FESEM, EDX, FTIR, UV–Vis-DRS, XPS, PL, and BET surface area analysis. X-ray diffraction (XRD) techniques were used to examine the crystallization of ZnO, Mg-ZnO, and S-ZnO samples. The Mg-ZnO and S-ZnO samples exhibit significant c-axis compression and smaller crystallite sizes as compared to undoped ZnO. The optical band gap of Mg-ZnO and S-ZnO NPs were found to be 2.93 eV and 2.32 eV, respectively, which are lower than that of ZnO NPs (3.05 eV). The S-doped ZnO resulted in the homogenous distribution of sulfur ions in the ZnO lattice crystal. XPS analysis revealed that the doped S element was mostly S4+ and S6+. A systematic evaluation has been conducted to assess the influence of several operational parameters, including doped/undoped stoichiometry, solution pH, catalyst dosage, and radical trapping experiment, on the photocatalytic degradation of Rhodamine 6G (Rh 6G) dye. Furthermore, we investigated the photocatalytic degradation activity of ZnO, Mg-ZnO, and S-ZnO samples with aquoues solution of 5 ppm Rhodamine 6G (Rh 6G) at room temperature. Results indicated that pure ZnO nanoparticles have the highest photocatalytic degradation rate constant (0.00344 min−1), compared to the samples Mg-ZnO (0.00104 min−1) and S-ZnO (0.00108 min−1) with Rh 6G dye in presence of visible light emitting diode (Vis-LED) source at room temperature. The enhanced visible light photocatalytic activities of pure ZnO NPs were attributed to their superior surface properties (18.30 m2/g) and effective electron–hole separation.
Graphical Abstract












Similar content being viewed by others
References
Abed C, Bouzidi C, Elhouichet H, Gelloz B, Ferid M (2015) Mg doping induced high structural quality of sol–gel ZnO nanocrystals: application in photocatalysis. Appl Surf Sci 349:855–863. https://doi.org/10.1016/j.apsusc.2015.05.078
Aga KW, Efa MT, Beyene TT (2022) Effects of sulfur doping and temperature on the energy bandgap of ZnO nanoparticles and their antibacterial activities. ACS Omega 7(12):10796–10803. https://doi.org/10.1021/acsomega.2c00647
Agrawal B, Patel J, Dave P, Thakarda J, Anand TS, Maity P (2021) Efficient synthesis of a new heterogeneous gold nanocatalyst stabilized by di-alkyne ligand and its applications for photocatalytic dye degradation and transfer hydrogenation reaction. J Nanopart Res 23:1–12. https://doi.org/10.1007/s11051-021-05182-9
Al-Hardan NH, Abdullah MJ, Aziz AA (2013) Performance of Cr-doped ZnO for acetone sensing. Appl Surf Sci 270:480–485. https://doi.org/10.1016/j.apsusc.2013.01.064
Aksoy S, Caglar Y, Ilican S, Caglar M (2012) Sol–gel derived Li–Mg co-doped ZnO films: preparation and characterization via XRD XPS FESEM. J Alloys Compd 512(1):171–178. https://doi.org/10.1016/j.jallcom.2011.09.058
Ardakani AG, Rafieipour P (2018) Using ZnO nanosheets grown by electrodeposition in random lasers as scattering centers: the effects of sheet size and presence of mode competition. JOSA B 35(7):1708–1716. https://doi.org/10.1364/JOSAB.35.001708
Bai H, Liu Z, Sun DD (2012) Hierarchical nitrogen-doped flowerlike ZnO nanostructure and its multifunctional environmental applications. Chem Asian J 7(8):1772–1780. https://doi.org/10.1002/asia.201200260
Balarabe BY, Maity P (2022) Visible light-driven complete photocatalytic oxidation of organic dye by plasmonic Au-TiO2 nanocatalyst under batch and continuous flow condition. Colloids Surf, A 655:130247. https://doi.org/10.1016/j.colsurfa.2022.130247
Balarabe BY, Paria S, Keita DS, Baraze ARI, Kalugendo E, Tetteh GNT, Oumarou NI (2022a) Enhanced UV-light active α-Bi2O3 nanoparticles for the removal of methyl orange and ciprofloxacin. Inorg Chem Commun 146:110204. https://doi.org/10.1016/j.inoche.2022.110204
Balarabe BY, Bowmik S, Ghosh A, Maity P (2022b) Photocatalytic dye degradation by magnetic XFe2O3 (X: Co, Zn, Cr, Sr, Ni, Cu, Ba, Bi, and Mn) nanocomposites under visible light: a cost efficiency comparison. J Magn Magn Mater 562:169823. https://doi.org/10.1016/j.jmmm.2022.169823
Bokov D, Turki Jalil A, Chupradi S, Suksatan W, Javed Ansari M, Shewael IH, Kianfar E (2021) Nanomaterial by sol-gel method: synthesis and application. Adv Mater Sci Eng 2021:1–21. https://doi.org/10.1155/2021/5102014
Capper P, Kasap SO, Willoughby A (2011) Zinc oxide materials for electronic and optoelectronic device applications. John Wiley & Sons
Coppa BJ, Davis RF, Nemanich RJ (2003) Gold Schottky contacts on oxygen plasma-treated, n-type ZnO (0001). Appl Phys Lett 82(3):400–402. https://doi.org/10.1063/1.1536264
Chen M, Wang X, Yu YH, Pei ZL, Bai XD, Sun C, Huang RF, Wen LS (2000) X-ray photoelectron spectroscopy and auger electron spectroscopy studies of Al-doped ZnO films. Appl Surf Sci 158:134–140. https://doi.org/10.1016/S0169-4332(99)00601-7
Debbichi M, Sakhraoui T, Debbichi L, Said M (2013) Hybrid functional study of structural electronic and magnetic properties of S-doped ZnO with and without neutral vacancy. J Alloys Compd 578:602–608. https://doi.org/10.1016/j.jallcom.2013.06.121
Ebrahimi S, Yarmand B (2019) Morphology engineering and growth mechanism of ZnS nanostructures synthesized by solvothermal process. J Nanoparticle Res 21:1–12. https://doi.org/10.1007/s11051-019-4714-z
Fujita S, Tanaka H, Fujita S (2005) MBE growth of wide band gap wurtzite MgZnO quasi-alloys with MgO/ZnO superlattices for deep ultraviolet optical functions. J Cryst Growth 278(1–4):264–267. https://doi.org/10.1016/j.jcrysgro.2005.01.029
Garcia-Martinez O, Rojas RM, Vila E, De VJM (1993) Microstructural characterization of nanocrystals of ZnO and CuO obtained from basic salts. Solid State Ion 63:442–449. https://doi.org/10.1016/0167-2738(93)90142-P
Giri P, Chakrabarti P (2016) Effect of Mg doping in ZnO buffer layer on ZnO thin film devices for electronic applications. Superlattices Microstruct 93:248–260. https://doi.org/10.1016/j.spmi.2016.03.024
Goncalves MS, Oliveira-Campos AM, Pinto EM, Plasencia PM, Queiroz MJR (1999) Photochemical treatment of solutions of azo dyes containing TiO2. Chemosphere 39(5):781–786
Hoggas K, Nouveau C, Djelloul A, Bououdina M (2015) Structural, microstructural, and optical properties of Zn1-xMgxO thin films grown onto glass substrate by ultrasonic spray pyrolysis. Appl Phys A 120:745–755. https://doi.org/10.1007/s00339-015-9252-7
Ip K, Heo YW, Norton DP, Pearton SJ, LaRoche JR, Ren F (2004) Zn 0.9 Mg 0.1 O∕ ZnO p-n junctions grown by pulsed-laser deposition. Appl Phys Lett 85(7):1169–1171. https://doi.org/10.1063/1.1783015
Jang M, Lee J, Park SY, Lee J, Lee KM, Song W, An KS (2021) Rational surface modification of ZnO with siloxane polymers for room-temperature-operated thin-film transistor-based gas sensors. Appl Surf Sci 542:148704. https://doi.org/10.1016/j.apsusc.2020.148704
Jayaraj SK, Sadishkumar V, Arun T, Thangadurai P (2018) Enhanced photocatalytic activity of V2O5 nanorods for the photodegradation of organic dyes: a detailed understanding of the mechanism and their antibacterial activity. Mater Sci Semicond 85:122–133. https://doi.org/10.1016/j.mssp.2018.06.006
Khan H, Habib M, Khan A, Boffito DC (2020) A modified sol-gel synthesis to yield a stable Fe3+/ZnO photocatalyst: degradation of water pollutants and mechanistic insights under UV and visible light. J Environ Chem Eng 8(5):104282. https://doi.org/10.1016/j.jece.2020.104282
Kumari V, Mittal A, Jindal J, Yadav S, Kumar N (2019) S-, N-and C-doped ZnO as semiconductor photocatalysts: a review. Front Mater Sci 13:1–22. https://doi.org/10.1007/s11706-019-0453-4
Labhane PK, Huse VR, Patle LB, Chaudhari AL, Sonawane GH (2015) Synthesis of Cu doped ZnO nanoparticles: crystallographic optical FTIR morphological and photocatalytic study. J Mater Sci Chem Eng 3(07):39. https://doi.org/10.4236/msce.2015.37005
Lai LW, Lee T (2008) Investigation of optical and electrical properties of ZnO thin films. Mater Chem Phy 110(2–3):393–396. https://doi.org/10.1016/j.matchemphys.2008.02.029
Lau GE, Che Abdullah CA, Wan Ahmad WN, Assaw S, Zheng ALT (2020) Eco-friendly photocatalysts for degradation of dyes. Catalysts 10(10):1129. https://doi.org/10.3390/catal10101129
Lin YY, Hsu CC, Tseng MH, Shyue JJ, Tsai FY (2015) Stable and high-performance flexible ZnO thin-film transistors by atomic layer deposition. ACS Appl Mater Interfaces 7(40):22610–22617. https://doi.org/10.1021/acsami.5b07278
Lee JY, Choi YS, Kim JH, Park MO, Im S (2002) Optimizing n-ZnO/p-Si heterojunctions for photodiode applications. Thin Solid Films 403:553–557. https://doi.org/10.1016/S0040-6090(01)01550-4
Lin C, Zhang H, Zhang J, Chen C (2019) Enhancement of the humidity sensing performance in Mg-doped hexagonal ZnO microspheres at room temperature. Sensors 19(3):519. https://doi.org/10.3390/s19030519
Liu T, Wang L, Lu X, Fan J, Cai X, Gao B, Miao R, Wang J, Lv Y (2017) Comparative study of the photocatalytic performance for the degradation of different dyes by ZnIn2S4: adsorption, active species and pathways. RSC Adv 7:12292–12300. https://doi.org/10.1039/C7RA00199A
Minemoto T, Negami T, Nishiwaki S, Takakura H, Hamakawa Y (2000) Preparation of Zn1- xMgxO films by radio frequency magnetron sputtering. Thin Solid Films 372(1–2):173–176. https://doi.org/10.1016/S0040-6090(00)01009-9
Mekasuwandumrong O, Pawinrat P, Praserthdam P, Panpranot J (2010) Effects of synthesis conditions and annealing post-treatment on the photocatalytic activities of ZnO nanoparticles in the degradation of methylene blue dye. Chem Eng 164(1):77–84. https://doi.org/10.1016/j.cej.2010.08.027
Maeda K, Domen K (2010) Solid solution of GaN and ZnO as a stable photocatalyst for overall water splitting under visible light. Chem Mater 22(3):612–623. https://doi.org/10.1021/cm901917a
Manikandan B, Endo T, Kaneko S, Murali KR, John R (2018) Properties of sol gel synthesized ZnO nanoparticles. J Mater Sci Mater Electron 29:9474–9485. https://doi.org/10.1007/s10854-018-8981-8
Mustafa SM, Barzinjy AA, Hamad AH (2023) An environmentally friendly green synthesis of Co2+ and Mn2+ ion doped ZnO nanoparticles to improve solar cell efficiency. J Environ Chem Eng 11(2):109514. https://doi.org/10.1016/j.jece.2023.109514
Mote VD, Purushotham Y, Dole BN (2012) Williamson-Hall analysis in estimation of lattice strain in nanometer-sized ZnO particles. J Theor Appl Phys 6:1–8. https://doi.org/10.1186/2251-7235-6-6
Mirzaeifard Z, Shariatinia Z, Jourshabani M, Rezaei Darvishi SM (2020) ZnO photocatalyst revisited: effective photocatalytic degradation of emerging contaminants using S-doped ZnO nanoparticles under visible light radiation. Ind Eng Chem Res 59(36):15894–15911. https://doi.org/10.1021/acs.iecr.0c03192
Neena D, Kondamareddy KK, Bin H, Lu D, Kumar P, Dwivedi RK, Fu D (2018) Enhanced visible light photodegradation activity of RhB/MB from aqueous solution using nanosized novel Fe-Cd co-modified ZnO. Sci Rep 8:10691. https://doi.org/10.1038/s41598-018-29025-1
Patterson AL (1939) The Scherrer formula for X-ray particle size determination. Phys Rev 56:978–982. https://doi.org/10.1103/PhysRev.56.978
Perumal SL, Hemalatha P, Alagara M, Pandiyaraj KN (2015) Investigation of structural, optical and photocatalytic properties of Sr doped Zno nanoparticles. Int J Phys Sci 4:1–13
Perween S, Ranjan A (2017) Improved visible-light photocatalytic activity in ZnTiO3 nanopowder prepared by sol-electrospinning. Sol Energy Mater Sol Cells 163:148–156. https://doi.org/10.1016/j.solmat.2017.01.020
Pradeev Raj K, Sadaiyandi K, Kennedy A, Sagadevan S, Chowdhury ZZ, Johan MRB, Rathina Bala R (2018) Influence of Mg doping on ZnO nanoparticles for enhanced photocatalytic evaluation and antibacterial analysis. Nanoscale Res Lett 13:1–13. https://doi.org/10.1186/s11671-018-2643-x
Prajapati AK, Mondal MK (2021) Novel green strategy for CuO–ZnO–C nanocomposites fabrication using marigold (Tagetes spp.) flower petals extract with and without CTAB treatment for adsorption of Cr (VI) and Congo red dye. J Environ Manage 290:112615. https://doi.org/10.1016/j.jenvman.2021.112615
Priscilla SJ, Daniel R, Dhakshayani Y, Caroline SC, Sivaji K (2021) Effect of magnesium dopant on the structural, morphological and electrical properties of ZnO nanoparticles by sol–gel method. Mater Today: Proc 36:793–796. https://doi.org/10.1016/j.matpr.2020.07.005
Rajesh UC, Wang J, Prescott S, Tsuzuki T, Rawat DS (2015) RGO/ZnO nanocomposite: an efficient sustainable heterogeneous amphiphilic catalyst for synthesis of 3-substituted indoles in water. ACS Sustain Chem Eng 3(1):9–18. https://doi.org/10.1021/sc500594w
Rana N, Chand S, Gathania AK (2015) Band gap engineering of ZnO by doping with Mg. Phys Scr 90(8):085502. https://doi.org/10.1088/0031-8949/90/8/085502
Rashid M, Al-Zaqri N, Guerrero-Barajas C, Hussain F, Ibrahim MNM (2022) Waste derived graphene oxide-ZnO: an efficient photocatalyst for Rhodamine 6G. Processes 10(11):2266. https://doi.org/10.3390/pr10112266
Ravirajan P, Peiró AM, Nazeeruddin MK, Graetzel M, Bradley DD, Durrant JR, Nelson J (2006) Hybrid polymer/zinc oxide photovoltaic devices with vertically oriented ZnO nanorods and an amphiphilic molecular interface layer. J Phys Chem B 110(15):7635–7639. https://doi.org/10.1021/jp0571372
Rodriguez JA, Jirsak T, Chaturvedi S, Kuhn M (1999) Reaction of SO2 with ZnO (0001̄)–O and ZnO powders: photoemission and XANES studies on the formation of SO3 and SO4. Surf Sci 442(3):400–412. https://doi.org/10.1016/S0039-6028(99)00952-8
Roxy MS, Ananthu A, Sumithranand VB (2021) Synthesis and characterization of undoped and magnesium doped zinc oxide nanoparticles. Int J Sci Res Sci Eng Technol 8:134–139. https://doi.org/10.32628/IJSRSET218225
Sivakami R, Dhanuskodi S, Karvembu R (2016) Estimation of lattice strain in nanocrystalline RuO2 by Williamson-Hall and size–strain plot methods. Spectrochim Acta A Mol Biomol Spectrosc 152:43–50. https://doi.org/10.1016/j.saa.2015.07.008
Sing KS (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl Chem 57(4):603–619. https://doi.org/10.1351/pac198557040603
Singh S, Perween S, Ranjan A (2021) Dramatic enhancement in adsorption of congo red dye in polymer-nanoparticle composite of polyaniline-zinc titanate. J Enviro Chem Eng 9(3):105149. https://doi.org/10.1016/j.jece.2021.105149
Singh S, Yadawa Y, Ranjan A (2023) Enhanced adsorption of methylene blue by mixed-phase bismuth ferrite prepared by non-aqueous sol-gel route. J Environ Chem Eng 11(1):109229. https://doi.org/10.1016/j.jece.2022.109229
Talam S, Karumuri SR, Gunnam N (2012) Synthesis, characterization, and spectroscopic properties of ZnO nanoparticles. Int Sch Res Notices. https://doi.org/10.5402/2012/372505
Thejaswini TVL, Prabhakaran D, Maheswari MA (2016) Soft synthesis of potassium co-doped Al-ZnO nanocomposites: a comprehensive study on their visible-light driven photocatalytic activity on dye degradation. J Mater Sci 51:8187–8208. https://doi.org/10.1007/s10853-016-0095-1
Tian C, Jiang D, Li B, Lin J, Zhao Y, Yuan W, Qin J (2014) Performance enhancement of ZnO UV photodetectors by surface plasmons. ACS Appl Mater Interfaces 6(3):2162–2166. https://doi.org/10.1021/am405292p
Umaralikhan L, Jaffar MJM (2017) Green synthesis of ZnO and Mg doped ZnO nanoparticles and its optical properties. J Mater Sci Mater Electron 28:7677–7685. https://doi.org/10.1007/s10854-017-6461-1
Vargas MA, Rivera-Muñoz EM, Diosa JE, Mosquera EE, Rodríguez-Páez JE (2021) Nanoparticles of ZnO and Mg-doped ZnO: synthesis, characterization and efficient removal of methyl orange (MO) from aqueous solution. Ceram Int 47(11):15668–15681. https://doi.org/10.1016/j.ceramint.2021.02.137
Vasantharaj S, Sathiyavimal S, Senthilkumar P, Kalpana VN, Rajalakshmi G, Alsehli M, Pugazhendhi A (2021) Enhanced photocatalytic degradation of water pollutants using bio-green synthesis of zinc oxide nanoparticles (ZnO NPs). J Environ Chem Eng 9(4):105772. https://doi.org/10.1016/j.jece.2021.105772
Vinh THT, Thuy NTB, Thi CM, Van Viet P (2021) Visible-light-driven photocatalysis of anisotropic silver nanoparticles decorated on ZnO nanorods: synthesis and characterizations. J Environ Chem Eng 9(2):105103. https://doi.org/10.1016/j.jece.2021.105103
Vishwakarma A, Singh SP (2020) Synthesis of zinc oxide nanoparticle by sol-gel method and study its characterization. Int J Res Appl Sci Eng Technol 8(4):1625–1627. https://doi.org/10.22214/ijraset.2020.4265
Wang CT, Lin JC (2008) Surface nature of nanoparticle zinc-titanium oxide aerogel catalysts. Appl Surf Sci 254(15):4500–4507. https://doi.org/10.1016/j.apsusc.2008.01.024
Wang X, Zhou J, Song J, Liu J, Xu N, Wang ZL (2006) Piezoelectric field effect transistor and nanoforce sensor based on a single ZnO nanowire. Nano Lett 6(12):2768–2772. https://doi.org/10.1021/nl061802g
Wong KK, Ng A, Chen XY, Ng YH, Leung YH, Ho KH, Phillips DL (2012) Effect of ZnO nanoparticle properties on dye-sensitized solar cell performance. ACS Appl Mater Interfaces 4(3):1254–1261. https://doi.org/10.1021/am201424d
Wróbel J, Piechota J (2008) On the structural stability of ZnO phases. Solid State Commun 146(7–8):324–329. https://doi.org/10.1016/j.ssc.2008.03.001
Wu S, Chen Z, Wang T, Ji X (2017) A facile approach for the fabrication of Au/ZnO-hollow-sphere-monolayer thin films and their photocatalytic properties. Appl Surf Sci 412:69–76. https://doi.org/10.1016/j.apsusc.2017.03.166
Yadawa Y, Singh S, Ranjan A (2023) Processing induced morphology change in ZnO-TiO2 multilayer thin films and its effect on their photocatalytic activity under visible light irradiation. Mater Sci Eng B 288:116164. https://doi.org/10.1016/j.mseb.2022.116164
Yang J, Xu C, Ye T, Wang Y, Meng D (2017a) Synthesis of S-doped hierarchical ZnO nanostructures via hydrothermal method and their optical properties. J Mater Sci: Mater Electron 28:1785–1792. https://doi.org/10.1007/s10854-016-5726-4
Yang J, Wang Y, Kong J, Yu M, Jin H (2016) Synthesis of Mg-doped hierarchical ZnO nanostructures via hydrothermal method and their optical properties. J Alloys Compd 657:261–267. https://doi.org/10.1016/j.jallcom.2015.10.117
Yang M, Zhang Z, Ma J, Liu L, Wang M, Pan B, Tong Z (2017b) Fabrication of intercalation hybrid of Ni-Al layered double hydroxide with Cu (II) phthalocyanine via exfoliation/restacking route and photocatalytic activity on elimination of Rhodamine 6G. J Incl Phenom Macrocycl Chem 87:117–125. https://doi.org/10.1007/s10847-016-0684-2
Yaou Balarabe B, Illiassou Oumarou MN, Korone AS, Adjama I, Ibrahim Baraze R (2023) Photo-oxidation of organic dye by F2O3 nanoparticles: catalyst, electron acceptor, and polyurethane membrane (PU-Fe2O3) effects. J Nanotechnol 2023. https://doi.org/10.1155/2023/1292762
Zayed M, Ahmed AM, Shaban M (2019) Synthesis and characterization of nanoporous ZnO and Pt/ZnO thin films for dye degradation and water splitting applications. Int J Hydrogen Energy 44(33):17630–17648. https://doi.org/10.1016/j.ijhydene.2019.05.117
Zhan P, Xie Z, Li Z, Wang W, Zhang Z, Li Z, Cao X (2013) Origin of the defects-induced ferromagnetism in un-doped ZnO single crystals. Appl Phys Lett 102(7):071914. https://doi.org/10.1063/1.4793574
Zhang H, Tao Z, Xu W, Lu S, Yuan F (2012) First-principles study of dopants and defects in S-doped ZnO and its effect on photocatalytic activity. Comput Mater Sci 58:119–124. https://doi.org/10.1016/j.commatsci.2012.01.016
Zhao Z, Lei W, Zhang X, Wang B, Jiang H (2010) ZnO-based amperometric enzyme biosensors. Sensors 10(2):1216–1231. https://doi.org/10.3390/s100201216
Zheng ALT, Sabidi S, Ohno T, Maeda T, Andou Y (2022) Cu2O/TiO2 decorated on cellulose nanofiber/reduced graphene hydrogel for enhanced photocatalytic activity and its antibacterial applications. Chemosphere 286:131731. https://doi.org/10.1016/j.chemosphere.2021.131731
Zheng ALT, Abdullah CAC, Chung ELT (2023) Andou Y (2023) Recent progress in visible light-doped ZnO photocatalyst for pollution control. Int J Environ Sci Technol 20:5753–5772. https://doi.org/10.1007/s13762-022-04354-x
Zheng Y, Chen C, Zhan Y, Lin X, Zheng Q, Wei K, Zhu Y (2007) Luminescence and photocatalytic activity of ZnO nanocrystals: correlation between structure and property. Inorg Chem 46(16):6675–6682. https://doi.org/10.1021/ic062394m
Zhuang H, Wang HJ, Li LJ, Xu P (2011) Structural and optical properties of ZnO nanowires doped with magnesium. Acta Phys Pol A 119(6):819–823
Acknowledgements
Authors gratefully acknowledge the CRF facility of RGIPT, Jais, Amethi, India for allowing us to use the powder XRD, FESEM, FTIR, XPS, and UV-Vis characterization facilities and Dr. Shikha Singh and Prof. M S Balathanigaimani for using the PL and BET surface area characterization.
Funding
RGIPT institute departmental fund.
Author information
Authors and Affiliations
Contributions
YY: conceptualization, validation, data curation, supervision, writing—original draft with review and editing.
DJ: perform experiment, collecting data and characterization.
NJ: perform experiment, collecting data and characterization.
Corresponding author
Ethics declarations
Ethical approval
On the behalf of me (Mr. Yogendra Yadawa) and all co-authors have allowed for ethical approval.
Consent to participate
Yes, allowed for consent to participate.
Consent for publication
Yes, allowed for consent to publish.
Competing interests
The authors declare no competing interests.
Ethical clearance
Mr. Yogendra Yadawa (on behalf of all co-authors).
Department of Chemical Engineering and Biochemical Engineering, Rajiv Gandhi Institute of Petroleum Technology, Jais, Amethi, Uttar Pradesh-229304, India.
Additional information
Responsible Editor: Sami Rtimi
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Mg and S-ZnO nanoparticles have easily synthesized by non-aqueous sol-gel route.
• Crystallite size of Mg and S-ZnO particles have reduced due to lattice strain.
• Flower-like nanosheet structure of S-ZnO samples has reduces the energy band gap.
• The high surface area enhanced the photocatalytic activity of Rh 6G dye.
• PVP reduces the photocatalytic activity of Rh 6G under visible light irradiation.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yadawa, Y., Jha, D. & Joshi, N. Cost-effective efficient materials for dye degradation using non-aqueous sol–gel route. Environ Sci Pollut Res 31, 740–756 (2024). https://doi.org/10.1007/s11356-023-31036-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-31036-z