Abstract
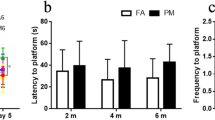
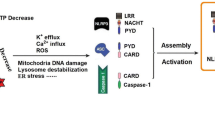
Ambient particulate matter (PM) is a global public and environmental problem. PM is closely associated with several neurological disorders that typically involve neuroinflammation. There have been few studies on the effect of PM on neuroinflammation to date. In this study, we used a juvenile rat model (PM exposure was conducted at a dose of 10 mg/kg body weight per day for 4 weeks) and a BV-2 cell model (PM exposure was conducted at concentrations of 50, 100, 150, and 200 μg/ml for 24 h) to investigate PM-induced neuroinflammation mediated by NLRP3 inflammasome activation and the role of TREM2 in this process. Our findings revealed that PM exposure reduced TREM2 protein and mRNA levels in the rat hippocampus and BV-2 cells. TREM2 overexpression attenuated PM-induced spatial learning and memory deficits in rats. Moreover, we observed that TREM2 overexpression in vivo and in vitro effectively mitigated the increase in NLRP3 and pro-Caspase1 protein expression, as well as the secretion of IL-1β and IL-18. Exposure to PM increased the expression of NF-κB and decreased the phosphorylation of PI3k/Akt in vivo and in vitro, and this process was effectively reversed by overexpressing TREM2. Our results indicated that PM exposure could reduce TREM2 expression and induce NLRP3 inflammasome-mediated neuroinflammation and that TREM2 could mitigate NLRP3 inflammasome-mediated neuroinflammation by regulating the NF-κB and PI3k/Akt signaling pathways. These findings shed light on PM-induced neuroinflammation mechanisms and potential intervention targets.







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- PM:
-
Ambient particulate matter
- GBD:
-
Global burden of disease
- CNS:
-
Central nervous system
- AD:
-
Alzheimer’s disease
- PD:
-
Parkinson’s disease
- ASC:
-
Apoptosis-associated speck-like protein
- pro-IL-1β:
-
Pro-interleukin-1β
- TREM2:
-
Triggering receptor expressed on myeloid cells 2
- Aβ:
-
β-Amyloid protein
- AAV:
-
Adeno-associated virus
- CCK8:
-
Cell counting kit-8
- SD:
-
Sprague Dawley
- PND:
-
Postnatal day
- NOR:
-
Novel object recognition test
- MWM:
-
Morris water maze test
- RT-qPCR:
-
Quantitative real-time polymerase chain reaction
- Ct:
-
Cycle threshold
- ELISA:
-
Enzyme-Linked Immunosorbent Assay
- SEM:
-
Standard errors of the means
- BBB:
-
Blood-brain barrier
- MMC:
-
Metropolitan Mexico City
- ASD:
-
Autism spectrum disorder
- ADHD:
-
Attention deficit hyperactivity disorder
- TNF-α:
-
Tumor necrosis factor-α
- IκB:
-
NF-κB inhibitors
- EMSA:
-
Electrophoretic mobility shift assay
- ChIP:
-
Chromatin immunoprecipitation
References
Afonina IS, Zhong Z, Karin M, Beyaert R (2017) Limiting inflammation-the negative regulation of NF-kappaB and the NLRP3 inflammasome. Nat Immunol 18:861–869
Ahadullah YSY, Lu HX, Lee TMC, Guo H, Chan CCH (2021) PM2.5 as a potential risk factor for autism spectrum disorder: Its possible link to neuroinflammation, oxidative stress and changes in gene expression. Neurosci Biobehav Rev 128:534–548
Calderon-Garciduenas L, Torres-Jardon R, Kulesza RJ, Mansour Y, Gonzalez-Gonzalez LO, Gonzalez-Maciel A, Reynoso-Robles R, Mukherjee PS (2020) Alzheimer disease starts in childhood in polluted Metropolitan Mexico City. A major health crisis in progress. Environ Res 183:109137
Chen S, Peng J, Sherchan P, Ma Y, Xiang S, Yan F, Zhao H, Jiang Y, Wang N, Zhang JH, Zhang H (2020a) TREM2 activation attenuates neuroinflammation and neuronal apoptosis via PI3K/Akt pathway after intracerebral hemorrhage in mice. J Neuroinflammation 17:168
Chen X, Guo J, Huang Y, Liu S, Huang Y, Zhang Z, Zhang F, Lu Z, Li F, Zheng JC, Ding W (2020b) Urban airborne PM2.5-activated microglia mediate neurotoxicity through glutaminase-containing extracellular vesicles in olfactory bulb. Environ Pollut 264:114716
Chen YC, Chuang TY, Liu CW, Liu CW, Lee TL, Lai TC, Chen YL (2020c) Particulate matters increase epithelial-mesenchymal transition and lung fibrosis through the ETS-1/NF-kappaB-dependent pathway in lung epithelial cells. Part Fibre Toxicol 17:41
Cheng J, Su H, Song J, Wang X (2022) Short-term effect of air pollution on childhood epilepsy in eastern China: A space-time-stratified case-crossover and pooled analysis. Environ Int 170:107591
Chu C, Zhang H, Cui S, Han B, Zhou L, Zhang N, Su X, Niu Y, Chen W, Chen R, Zhang R, Zheng Y (2019) Ambient PM2.5 caused depressive-like responses through Nrf2/NLRP3 signaling pathway modulating inflammation. J Hazard Mater 369:180–190
Cohen AJ et al (2017) Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet 389:1907–1918
Collaborators GBDRF (2020) Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1223–1249
Costa LG, Cole TB, Dao K, Chang YC, Coburn J, Garrick JM (2020) Effects of air pollution on the nervous system and its possible role in neurodevelopmental and neurodegenerative disorders. Pharmacol Ther 210:107523
Ehsanifar M, Montazeri Z, Zavareh MS, Rafati M, Wang J (2023) Cognitive impairment, depressive-like behaviors and hippocampal microglia activation following exposure to air pollution nanoparticles. Environ Sci Pollut Res Int 30:23527–23537
Ford JW, McVicar DW (2009) TREM and TREM-like receptors in inflammation and disease. Curr Opin Immunol 21:38–46
Gui J, Liu J, Han Z, Yang X, Ding R, Yang J, Luo H, Huang D, Chen H, Cheng L, Jiang L (2023) The dysfunctionality of hippocampal synapses may be directly related to PM-induced impairments in spatial learning and memory in juvenile rats. Ecotoxicol Environ Saf 254:114729
Guo L, Zhu N, Guo Z, Li GK, Chen C, Sang N, Yao QC (2012) Particulate matter (PM10) exposure induces endothelial dysfunction and inflammation in rat brain. J Hazard Mater 213–214:28–37
Guo L, Li B, Miao JJ, Yun Y, Li GK, Sang N (2015) Seasonal variation in air particulate matter (PM10) exposure-induced ischemia-like injuries in the rat brain. Chem Res Toxicol 28:431–439
Guo Y, Wei X, Yan H, Qin Y, Yan S, Liu J, Zhao Y, Jiang F, Lou H (2019) TREM2 deficiency aggravates alpha-synuclein-induced neurodegeneration and neuroinflammation in Parkinson’s disease models. FASEB J 33:12164–12174
Han X, Cheng X, Xu J, Liu Y, Zhou J, Jiang L, Gu X, Xia T (2022) Activation of TREM2 attenuates neuroinflammation via PI3K/Akt signaling pathway to improve postoperative cognitive dysfunction in mice. Neuropharmacology 219:109231
Hill W et al (2023) Lung adenocarcinoma promotion by air pollutants. Nature 616:159–167
Hu T, Lu XY, Shi JJ, Liu XQ, Chen QB, Wang Q, Chen YB, Zhang SJ (2020) Quercetin protects against diabetic encephalopathy via SIRT1/NLRP3 pathway in db/db mice. J Cell Mol Med 24:3449–3459
Jay TR et al (2015) TREM2 deficiency eliminates TREM2+ inflammatory macrophages and ameliorates pathology in Alzheimer’s disease mouse models. J Exp Med 212:287–295
Jiang W, Liu F, Li H, Wang K, Cao X, Xu X, Zhou Y, Zou J, Zhang X, Cui X (2022) TREM2 ameliorates anesthesia and surgery-induced cognitive impairment by regulating mitophagy and NLRP3 inflammasome in aged C57/BL6 mice. Neurotoxicology 90:216–227
Kang YJ, Tan HY, Lee CY, Cho H (2021) An Air Particulate Pollutant Induces Neuroinflammation and Neurodegeneration in Human Brain Models. Adv Sci (weinh) 8:e2101251
Kioumourtzoglou MA, Schwartz JD, Weisskopf MG, Melly SJ, Wang Y, Dominici F, Zanobetti A (2016) Long-term PM2.5 exposure and neurological hospital admissions in the northeastern united states. Environ Health Perspect 124:23–29
Ku T, Li B, Gao R, Zhang Y, Yan W, Ji X, Li G, Sang N (2017) NF-kappaB-regulated microRNA-574–5p underlies synaptic and cognitive impairment in response to atmospheric PM2.5 aspiration. Part Fibre Toxicol 14:34
Li C, Zhao B, Lin C, Gong Z, An X (2019) TREM2 inhibits inflammatory responses in mouse microglia by suppressing the PI3K/NF-kappaB signaling. Cell Biol Int 43:360–372
Li Y, Wang R, Xue L, Yang Y, Zhi F (2020) Astilbin protects against cerebral ischaemia/reperfusion injury by inhibiting cellular apoptosis and ROS-NLRP3 inflammasome axis activation. Int Immunopharmacol 84:106571
Li L, Xing C, Zhou J, Niu L, Luo B, Song M, Niu J, Ruan Y, Sun X, Lei Y (2021a) Airborne particulate matter (PM(2.5)) triggers ocular hypertension and glaucoma through pyroptosis. Part Fibre Toxicol 18:10
Li Y, Long W, Gao M, Jiao F, Chen Z, Liu M, Yu L (2021b) TREM2 Regulates High Glucose-Induced Microglial Inflammation via the NLRP3 Signaling Pathway. Brain Sci 11(7):896
Liu C, She Y, Huang J, Liu Y, Li W, Zhang C, Zhang T, Yu L (2022) HMGB1-NLRP3-P2X7R pathway participates in PM(2.5)-induced hippocampal neuron impairment by regulating microglia activation. Ecotoxicol Environ Saf 239:113664
Lv MR, Li B, Wang MG, Meng FG, Yu JJ, Guo F, Li Y (2017) Activation of the PI3K-Akt pathway promotes neuroprotection of the delta-opioid receptor agonist against cerebral ischemia-reperfusion injury in rat models. Biomed Pharmacother 93:230–237
Qiao Y, Wang P, Qi J, Zhang L, Gao C (2012) TLR-induced NF-kappaB activation regulates NLRP3 expression in murine macrophages. FEBS Lett 586:1022–1026
Shou Y, Huang Y, Zhu X, Liu C, Hu Y, Wang H (2019) A review of the possible associations between ambient PM2.5 exposures and the development of Alzheimer’s disease. Ecotoxicol Environ Saf 174:344–352
Sinharay R, Gong J, Barratt B, Ohman-Strickland P, Ernst S, Kelly FJ, Zhang JJ, Collins P, Cullinan P, Chung KF (2018) Respiratory and cardiovascular responses to walking down a traffic-polluted road compared with walking in a traffic-free area in participants aged 60 years and older with chronic lung or heart disease and age-matched healthy controls: a randomised, crossover study. Lancet 391:339–349
Song X, Yang S, Shao L, Fan J, Liu Y (2016) PM10 mass concentration, chemical composition, and sources in the typical coal-dominated industrial city of Pingdingshan, China. Sci Total Environ 571:1155–1163
Swanson KV, Deng M, Ting JP (2019) The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol 19:477–489
Takahashi K, Rochford CD, Neumann H (2005) Clearance of apoptotic neurons without inflammation by microglial triggering receptor expressed on myeloid cells-2. J Exp Med 201:647–657
Wang Y, Cella M, Mallinson K, Ulrich JD, Young KL, Robinette ML, Gilfillan S, Krishnan GM, Sudhakar S, Zinselmeyer BH, Holtzman DM, Cirrito JR, Colonna M (2015) TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell 160:1061–1071
Wang BR, Shi JQ, Ge NN, Ou Z, Tian YY, Jiang T, Zhou JS, Xu J, Zhang YD (2018) PM2.5 exposure aggravates oligomeric amyloid beta-induced neuronal injury and promotes NLRP3 inflammasome activation in an in vitro model of Alzheimer’s disease. J Neuroinflammation 15:132
Wang Y, Cao C, Zhu Y, Fan H, Liu Q, Liu Y, Chen K, Wu Y, Liang S, Li M, Li L, Liu X, Zhang Y, Wu C, Lu G, Wu M (2022) TREM2/beta-catenin attenuates NLRP3 inflammasome-mediated macrophage pyroptosis to promote bacterial clearance of pyogenic bacteria. Cell Death Dis 13:771
Xue Z, Zhang Z, Liu H, Li W, Guo X, Zhang Z, Liu Y, Jia L, Li Y, Ren Y, Yang H, Zhang L, Zhang Q, Da Y, Hao J, Yao Z, Zhang R (2019) lincRNA-Cox2 regulates NLRP3 inflammasome and autophagy mediated neuroinflammation. Cell Death Differ 26:130–145
Younan D et al (2020) Particulate matter and episodic memory decline mediated by early neuroanatomic biomarkers of Alzheimer’s disease. Brain 143:289–302
Zhang Y, Feng S, Nie K, Li Y, Gao Y, Gan R, Wang L, Li B, Sun X, Wang L, Zhang Y (2018) TREM2 modulates microglia phenotypes in the neuroinflammation of Parkinson’s disease. Biochem Biophys Res Commun 499:797–802
Zhang J, Xu D, Cui H, Zhao T, Chu C, Wang J (2021) Group-guided individual functional parcellation of the hippocampus and application to normal aging. Hum Brain Mapp 42:5973–5984
Zhao Y, Wu X, Li X, Jiang LL, Gui X, Liu Y, Sun Y, Zhu B, Pina-Crespo JC, Zhang M, Zhang N, Chen X, Bu G, An Z, Huang TY, Xu H (2018) TREM2 Is a Receptor for beta-Amyloid that Mediates Microglial Function. Neuron 97(1023–1031):e7
Zhong X, Xie L, Yang X, Liang F, Yang Y, Tong J, Zhong Y, Zhao K, Tang Y, Yuan C (2020) Ethyl pyruvate protects against sepsis-associated encephalopathy through inhibiting the NLRP3 inflammasome. Mol Med 26:55
Zhong W, Shi D, Zhou J, Yang Y, Wang B, Sun X, Shu Q, Li W, Xia Y, Ao L, Xiong K, Hao S, Xia H (2023) Role of trichocytic keratins in anti‐neuroinflammatory effects after spinal cord injury. Adv Funct Mater 33(23). https://doi.org/10.1002/adfm.202212870
Funding
This work was supported by the Talent Program of Chongqing, China (cstc2021ycjh-bgzxm0187) and the Chongqing Postdoctoral Special Foundation (CSTB2022NSCQ-BHX0708).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Jianxiong Gui, Li Jiang, Jie Liu, Lingman Wang, Hanyu Luo, and Ziyao Han. The first draft of the manuscript was written by Jianxiong Gui, Li Jiang, Dishu Huang, Xiaoyue Yang, Honghong Song, Ran Ding, and Jiaxin Yang and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
All animal experimental procedures conducted in our study adhered to the guidelines set forth by the National Research Council’s Guide for the Care and Use of Laboratory Animals. Furthermore, these procedures were thoroughly reviewed and approved by the Ethics Committee of the Children’s Hospital Affiliated with Chongqing Medical University, under Protocol No. CHCMU-IACUC20220629008.
Consent to participate
All authors agree to participate in the experiment and the writing of this paper.
Consent to publish
All authors agree that the Environmental Science and Pollution Research will publish this paper. All data in this paper can be published publicly.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Ludek Blaha
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gui, J., Liu, J., Wang, L. et al. TREM2 mitigates NLRP3-mediated neuroinflammation through the NF-κB and PI3k/Akt signaling pathways in juvenile rats exposed to ambient particulate matter. Environ Sci Pollut Res 30, 119863–119878 (2023). https://doi.org/10.1007/s11356-023-30764-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30764-6