Abstract
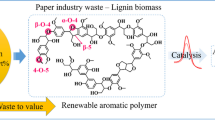
In recent decades, due to abundance (second most abundant natural polymer after cellulose) and sustainability, lignin has attracted much interest from different researchers to use as a raw material for producing various value-added materials such as polymer and fuel. In addition to that, the aromatic structure of lignin makes it a suitable candidate for producing platform chemicals with aromatic rings. As a result, lignin depolymerization has become an interesting process to derive different phenolic monomers like vanillin, acetosyringone, and guaiacol. Among them, due to the bioactive characteristics and efficiency of acetosyringone in plant regulatory systems, the production of acetosyringone from lignin has been presented in this work. A green and cost-effective method was developed for the selective formation of acetosyringone via depolymerization of isolated rice straw lignin (RSL) by using metal catalyst-free conditions in the biphasic medium and described. The RSL was characterized with various spectroscopic techniques such as FT-IR, solid-state 13C NMR, XPS, and TGA. The selectivity of synthesized acetosyringone during depolymerization of RSL was checked from GC-MS analysis. The molecular structure and purity of acetosyringone isolated from preparative thin layer chromatography (TLC) were confirmed with the help of 1H NMR and HRMS, respectively.












Similar content being viewed by others
Data Availability
The experimental and instrumental raw data can be provided upon request.
References
Ahmad Z, Paleologou M, Xu CC (2021) Oxidative depolymerization of lignin using nitric acid under ambient conditions. Ind Crops Prod 170:113757. https://doi.org/10.1016/j.indcrop.2021.113757
Arefmanesh M, Vuong TV, Mobley JK, Alinejad M, Master ER, Nejad M (2020) Bromide-based ionic liquid treatment of hardwood organosolv lignin yielded a more reactive biobased polyol. Ind Eng Chem Res 59:18740–18747. https://doi.org/10.1021/acs.iecr.0c03718
Cao C, Xie Y, Li L, Wei W, Jin H, Wang S, Li W (2021) Supercritical water gasification of lignin and cellulose catalyzed with co-precipitated CeO2-ZrO2. Energy and Fuels 35:6030–6039. https://doi.org/10.1021/acs.energyfuels.1c00242
Chaudhary R, Dhepe PL (2019) Depolymerization of lignin using a solid base catalyst. Energy and Fuels 33:4369–4377. https://doi.org/10.1021/acs.energyfuels.9b00621
Collard FX, Blin J (2014) A review on pyrolysis of biomass constituents: mechanisms and composition of the products obtained from the conversion of cellulose, hemicelluloses and lignin. Renew Sustain Energy Rev 38:594–608. https://doi.org/10.1016/j.rser.2014.06.013
Deepa AK, Dhepe PL (2014) Solid acid catalyzed depolymerization of lignin into value added aromatic monomers. RSC Adv 4:12625–12629. https://doi.org/10.1039/c3ra47818a
Dutta K, Saikia A, Saikia BK, Singh A (2023) Functionalization and thereafter grafting with lactic acid to synthesize lignin-polylactic acid copolymer for thin film preparation. J Polym Environ 31:3393–3403. https://doi.org/10.1007/s10924-023-02825-x
Dutta K, Singh A (2022) Chemical modification of lignin and thereafter grafting with lactic acid for flexible polymer film preparation. J Appl Polym Sci 139:52320. https://doi.org/10.1002/app.52320
Feng Y, Lan J, Ma P, Dong X, Qu J, He H (2017) Chemical structure and thermal properties of lignin modified with polyethylene glycol during steam explosion. Wood Sci Technol 51:135–150. https://doi.org/10.1007/s00226-016-0870-9
Gosselink RJA, Abächerli A, Semke H, Malherbe R, Käuper P, Nadif A, Van Dam JEG (2004) Analytical protocols for characterisation of sulphur-free lignin. Ind Crops Prod 19:271–281. https://doi.org/10.1016/j.indcrop.2003.10.008
Guo X, Zhang S, Shan X (2008) Adsorption of metal ions on lignin. J Hazard Mater 151:134–142. https://doi.org/10.1016/j.jhazmat.2007.05.065
Hasegawa I, Inoue Y, Muranaka Y, Yasukawa T, Mae K (2011) Selective production of organic acids and depolymerization of lignin by hydrothermal oxidation with diluted hydrogen peroxide. Energy Fuels 25:791–796. https://doi.org/10.1021/ef101477d
Isikgor FH, Becer CR (2015) Lignocellulosic biomass: a sustainable platform for the production of bio-based chemicals and polymers. Polym Chem 6:4497–4559. https://doi.org/10.1039/C5PY00263J
Kang K, Azargohar R, Dalai AK, Wang H (2015) Noncatalytic gasification of lignin in supercritical water using a batch reactor for hydrogen production: an experimental and modeling study. Energy Fuels 29:1776–1784. https://doi.org/10.1021/ef5027345
Kumar A, Biswas B, Bhaskar T (2020) Effect of cobalt on titania, ceria and zirconia oxide supported catalysts on the oxidative depolymerization of prot and alkali lignin. Bioresour Technol 299:122589. https://doi.org/10.1016/j.biortech.2019.122589
Kumar A, Biswas B, Saini K, Kumar A, Kumar J, Krishna BB, Bhaskar T (2021) Py-GC/MS study of prot lignin with cobalt impregnated titania, ceria and zirconia catalysts. Renew Energy 172:121–129. https://doi.org/10.1016/j.renene.2021.03.011
Li J, Henriksson G, Gellerstedt G (2007) Lignin depolymerization/repolymerization and its critical role for delignification of aspen wood by steam explosion. Bioresour Technol 98:3061–3068. https://doi.org/10.1016/j.biortech.2006.10.018
Li W, Zhang Y, Das L, Wang Y, Li M, Wanninayake N, Pu Y, Kim DY, Cheng YT, Ragauskas AJ, Shi J (2018) Linking lignin source with structural and electrochemical properties of lignin-derived carbon materials. RSC Adv 8:38721–38732. https://doi.org/10.1039/c8ra08539k
Lu YC, Lu Y, Fan X (2020) Structure and characteristics of lignin. Lignin: Biosynthesis Transformation Ind Appl:17–75. https://doi.org/10.1007/978-3-030-40663-9_2
Mahmood Z, Yameen M, Jahangeer M, Riaz M, Ghaffar A, Javid I (2018) Lignin as natural antioxidant capacity. Lignin - Trends Appl 10:181–205. https://doi.org/10.5772/intechopen.73284
Malins K (2017) Production of bio-oil via hydrothermal liquefaction of birch sawdust. Energy Convers Manag 144:243–251. https://doi.org/10.1016/j.enconman.2017.04.053
Mathew AK, Abraham A, Mallapureddy KK, Sukumaran RK (2018) Lignocellulosic biorefinery wastes, or resources? In: Waste biorefinery: potential and perspectives. Elsevier, pp 267–297. https://doi.org/10.1016/B978-0-444-63992-9.00009-4
Meier D, Ante R, Faix O (1992) Catalytic hydropyrolysis of lignin: influence of reaction conditions on the formation and composition of liquid products. Bioresour Technol 40:171–177. https://doi.org/10.1016/0960-8524(92)90205-C
Nandiwale KY, Danby AM, Ramanathan A, Chaudhari RV, Motagamwala AH, Dumesic JA, Subramaniam B (2020) Enhanced acid-catalyzed lignin depolymerization in a continuous reactor with stable activity. ACS Sustain Chem Eng 8:4096–4106. https://doi.org/10.1021/acssuschemeng.9b06556
Panyadee R, Saengsrichan A, Posoknistakul P, Laosiripojana N, Ratchahat S, Matsagar BM, Wu KCW, Sakdaronnarong C (2021) Lignin-derived syringol and acetosyringone from palm bunch using heterogeneous oxidative depolymerization over mixed metal oxide catalysts under microwave heating. Molecules 26:7444. https://doi.org/10.3390/molecules26247444
Park JH, Jin MH, Lee YJ, Song GS, Choi JW, Lee DW, Choi YC, Park SJ, Song KH, Kim JG (2019) Two-in-one fuel synthetic bioethanol-lignin from lignocellulose with sewage sludge and its air pollutants reduction effects. Energies (Basel) 12:3072. https://doi.org/10.3390/en12163072
Questell-Santiago YM, Galkin MV, Barta K, Luterbacher JS (2020) Stabilization strategies in biomass depolymerization using chemical functionalization. Nat Rev Chem 4:311–330. https://doi.org/10.1038/s41570-020-0187-y
Rawat S, Gupta P, Singh B, Bhaskar T, Natte K, Narani A (2020) Molybdenum-catalyzed oxidative depolymerization of alkali lignin: selective production of Vanillin. Appl Catal A Gen 598:117567. https://doi.org/10.1016/j.apcata.2020.117567
Resende FLP, Fraley SA, Berger MJ, Savage PE (2008) Noncatalytic gasification of lignin in supercritical water. Energy Fuels 22:1328–1334. https://doi.org/10.1021/ef700574k
Roldan ML, Centeno SA, Rizzo A, Van Dyke Y (2014) Characterization of bistre pigment samples by FTIR, SERS, Py-GC/MS and XRF. Mater Res Soc Symp Proc 1656. https://doi.org/10.1557/opl.2014.823
Rozite L, Varna J, Joffe R, Pupurs A (2013) Nonlinear behavior of PLA and lignin-based flax composites subjected to tensile loading. J Thermoplast Compos Mater 26:476–496. https://doi.org/10.1177/0892705711425846
Schwanninger M, Rodrigues JC, Pereira H, Hinterstoisser B (2004) Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib Spectrosc 36:23–40. https://doi.org/10.1016/j.vibspec.2004.02.003
Song Q, Wang F, Cai J, Wang Y, Zhang J, Yu W, Xu J (2013) Lignin depolymerization (LDP) in alcohol over nickel-based catalysts via a fragmentation-hydrogenolysis process. Energy Environ Sci 6:994–1007. https://doi.org/10.1039/c2ee23741e
Terrett OM, Lyczakowski JJ, Yu L, Iuga D, Franks WT, Brown SP, Dupree R, Dupree P (2019) Molecular architecture of softwood revealed by solid-state NMR. Nat Commun 10:4978. https://doi.org/10.1038/s41467-019-12979-9
Tran VT, Le TM, Vu PV, Nguyen HM, Duong YHP, Le PK (2022) Depolymerization of rice straw lignin into value-added chemicals in sub-supercritical ethanol. Sci World J 2022. https://doi.org/10.1155/2022/7872307
Walch F, Abdelaziz OY, Meier S, Bjelić S, Hulteberg CP, Riisager A (2021) Oxidative depolymerization of Kraft lignin to high-value aromatics using a homogeneous vanadium-copper catalyst. Catal Sci Technol 11:1843–1853. https://doi.org/10.1039/d0cy02158j
Watkins D, Nuruddin M, Hosur M, Tcherbi-Narteh A, Jeelani S (2015) Extraction and characterization of lignin from different biomass resources. J Mater Res Technol 4:26–32. https://doi.org/10.1016/j.jmrt.2014.10.009
Xu W, Miller SJ, Agrawal PK, Jones CW (2012) Depolymerization and hydrodeoxygenation of switchgrass lignin with formic acid. ChemSusChem 5:667–675. https://doi.org/10.1002/cssc.201100695
Yadav R, Mehrotra M, Singh AK, Niranjan A, Singh R, Sanyal I, Lehri A, Pande V, Amla DV (2017) Improvement in Agrobacterium-mediated transformation of chickpea (Cicer arietinum L.) by the inhibition of polyphenolics released during wounding of cotyledonary node explants. Protoplasma 254:253–269. https://doi.org/10.1007/s00709-015-0940-0
Yu J, Wang D, Sun L (2021) The pyrolysis of lignin: pathway and interaction studies. Fuel 290:120078. https://doi.org/10.1016/j.fuel.2020.120078
Zhang M, Resende FLP, Moutsoglou A, Raynie DE (2012) Pyrolysis of lignin extracted from prairie cordgrass, aspen, and Kraft lignin by Py-GC/MS and TGA/FTIR. J Anal Appl Pyrolysis 98:65–71. https://doi.org/10.1016/j.jaap.2012.05.009
Zhao W, Simmons B, Singh S, Ragauskas A, Cheng G (2016) From lignin association to nano-/micro-particle preparation: extracting higher value of lignin. Green Chem 18:5693–5700. https://doi.org/10.1039/C6GC01813K
Zhou G, Taylor G, Polle A (2011) FTIR-ATR-based prediction and modelling of lignin and energy contents reveals independent intra-specific variation of these traits in bioenergy poplars. Plant Methods 7:9. https://doi.org/10.1186/1746-4811-7-9
Acknowledgements
The authors would like to thank the Director, CSIR-NEIST, Jorhat, for his support and encouragement in carrying out the research work. Further, the authors also acknowledge AcSIR-Ghaziabad, India 201002, for permitting Ms. Ankumoni Saikia to register under PhD program.
Funding
This work was funded by the CSIR-North East Institute of Science and Technology, Jorhat, India, under In-house project OLP-2059.
Author information
Authors and Affiliations
Contributions
Ankumoni Saikia: material preparation, data collection, formal analysis, and first draft of the manuscript. Koushik Dutta: data collection, formal analysis, and first draft of the manuscript. Debashree Bora: material preparation. Biswajit Saha: formal analysis, review, and editing of the first draft. Ajit Singh: supervision, review, and editing of the manuscript. All authors contributed and approved the final manuscript.
Corresponding author
Ethics declarations
Consent to participate
All authors have made substantial contributions to this research work. The contribution of all the authors has been clearly mentioned in the author contribution section. All authors agree to be accountable and approve the manuscript for further processing.
Consent for publication
All authors agree to publish this manuscript in this journal.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The submitted work is original. The manuscript is neither submitted nor published to anywhere else.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saikia, A., Dutta, K., Bora, D. et al. Metal catalyst-free selective acetosyringone synthesis from rice straw lignin. Environ Sci Pollut Res 30, 100203–100214 (2023). https://doi.org/10.1007/s11356-023-29349-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-29349-0