Abstract
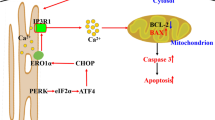
Copper nanoparticles (CuNPs) have been widely utilized in various applications. Due to its wider application, humans are at risk of its exposure. It has been reported that the exposure of CuNPs can lead to organ accumulation and affect organ toxicity. Recent study suggested that CuNPs can translocate into the uterus and affect uterine injury in rat, whereas uterine toxicity still remains unclear. The uterus is an important female organ which is required to sustain pregnancy. Thus, uterine structure and physiology are important. Therefore, this study hypothesized that CuNPs might have a toxic effect on the uterine features of mice. In this study, we have investigated the potential effects of CuNPs on the uterus of mice both in vivo and in vitro. In in vivo study, two groups of female mice were exposed to 5 and 50 mg/kg/day via oral exposure. In vivo results showed that CuNP treatment decreases the body weight and uterus weight and changes in antioxidant status with low estrogen and progesterone levels. Furthermore, CuNPs up-regulated the expression of caspase3 and down-regulated the expression of apelin receptor (APJ). Immunolocalization of apelin showed low abundance in the CuNP-treated uterus. These results suggest a poor apelin signaling in the uterus after CuNP treatment. The in vivo findings were further supported by the in vitro studies. Firstly, the uterus was cultured with 5 and 40 μg of CuNPs, and in the second in vitro experiment, the uterus was divided into 4 groups: control, 40 μg of CuNPs, 40 μg of CuNPs with apelin, and 40 μg of CuNPs with apelin receptor antagonist (ML221). In vitro study showed that CuNPs could directly induce the oxidative stress and apoptosis as well as changing antioxidant status in the uterus. The in vitro apelin 13 (APLN 13) treatments alleviated the expression of BCL2 and improved the antioxidant markers in CuNP-treated uterus. These results also provided an evidence of apelin-mediated signaling in the CuNP-treated uterus. In summary, our results present evidence that CuNPs can stimulate apoptotic pathways which may lead to uterine impairment due to weak apelin signaling.









Similar content being viewed by others
Data availability
All data used and/or analyzed during the experiments will be made available on reasonable request.
References
Ameh T, Sayes CM (2019) The potential exposure and hazards of copper nanoparticles: a review. Environ Toxicol Pharmacol 71:103220. https://doi.org/10.1016/j.etap.2019.103220
Anima B, Guruswami G, Roy VK (2023) Postnatal developmental expression and localization of apelin and apelin receptor protein in the ovary and uterus of mice. Mol Reprod Dev 90(1):42–52. https://doi.org/10.1002/mrd.23657
Annie L, Gurusubramanian G, Roy VK (2020) Changes in the localization of ovarian visfatin protein and its possible role during estrous cycle of mice. Acta Histochem 122(8):151630. https://doi.org/10.1016/j.acthis.2020.151630
Anreddy RNR (2018) Copper oxide nanoparticles induces oxidative stress and liver toxicity in rats following oral exposure. Toxicol Rep 5:903–904. https://doi.org/10.1016/j.toxrep.2018.08.022
Arulprakasajothi M, Elangovan K, Chandrasekhar U, Suresh S (2018) Performance study of conical strip inserts in tube heat exchanger using water based titanium oxide nanofluid. Therm Sci 22(1 Part B):477–485
Asalah AK, Attia KI, Mahdi SES (2020) Apelin induced modulation of uterine contractility in adult albino rats and its possible mechanism/s of action. Zagazig Univ Med J 26(1):174–185. https://doi.org/10.21608/zumj.2019.11148.1157
Bancroft JD, Gamble M (2008) Theory and practice of histological techniques. Elsevier health sciences
Bradford NJAB (1976) A rapid and sensitive method for the quantitation microgram quantities of a protein isolated from red cell membranes. Anal Biochem 72(248):e254. https://doi.org/10.1016/0003-2697(76)90527-3
Chen B, Chang CH (2003) Finite element simulation of incompressible fluid flow in an elastic vessel. Int J Numer Methods Fluids 42:131–146. https://doi.org/10.1016/j.toxlet.2005.10.003
Choobineh K, Zavareh S, Salehnia M, Ghorbanian MT (2016) Expression of pluripotent stem cell markers in mouse uterine tissue during estrous cycle. In Veterinary Research Forum (7, 3, 181). Faculty of Veterinary Medicine, Urmia University, Urmia, Iran.
Curtis J, Greenberg M, Kester J, Phillips S, Krieger G (2006) Nanotechnology and nanotoxicology. Toxicol Rev 25(4):245–260. https://doi.org/10.2165/00139709-200625040-00005
Das M, Annie L, Derkach KV, Shpakov AO, Gurusubramanian G, Roy VK (2021) Expression and localization of apelin and its receptor in the testes of diabetic mice and its possible role in steroidogenesis. Cytokine 144:155554. https://doi.org/10.1016/j.cyto.2021.155554
Din MI, Arshad F, Hussain Z, Mukhtar M (2017) Green adeptness in the synthesis and stabilization of copper nanoparticles: catalytic, antibacterial, cytotoxicity, and antioxidant activities. Nanoscale Res Lett 12(1):1–15. https://doi.org/10.1186/s11671-017-2399-8
El Bialy BE, Hamouda RA, AbdEldaim MA, El Ballal SS, Heikal HS, Khalifa HK, Hozzein WN (2020) Comparative toxicological effects of biologically and chemically synthesized copper oxide nanoparticles on mice. Int J Nanomedicine 15:3827. https://doi.org/10.2147/IJN.S241922
Ermini ML, Voliani V (2021) Antimicrobial nano-agents: the copper age. ACS Nano 15(4):6008–6029
Gao G, Ze Y, Li B, Zhao X, Zhang T, Sheng L, Hu R, Gui S, Sang X, Sun Q, Cheng J (2012) Ovarian dysfunction and gene-expressed characteristics of female mice caused by long-term exposure to titanium dioxide nanoparticles. J Hazard Mater 243:19–27. https://doi.org/10.1016/j.jhazmat.2012.08.049
Hehir MP, Morrison JJ (2012) The adipokine apelin and human uterine contractility. Am J Obstet Gynecol 206(4):359–3e1. https://doi.org/10.1016/j.ajog.2012.01.032
Hou CC, Zhu JQ (2017) Nanoparticles and female reproductive system: how do nanoparticles affect oogenesis and embryonic development. Oncotarget 8(65):109799. https://doi.org/10.18632/oncotarget.19087
Hou J, Wan X, Wang F, Xu G, Liu Z, Zhang T (2009) Effects of titanium dioxide nanoparticles on development and maturation of rat preantral follicle in vitro. Acad J Second Mil Univ 30(8):869–873
Hu LX, He J, Hou L, Wang H, Li J, Xie C et al (2013) Biological evaluation of the copper/low-density polyethylene nanocomposite intrauterine device. PLoS One 8(9):e74128. https://doi.org/10.1371/journal.pone.0074128
Hu S, Yang J, Rao M, Wang Y, Zhou F, Cheng G, Xia W, Zhu C (2019) Copper nanoparticle-induced uterine injury in female rats. Environ Toxicol 34(3):252–261. https://doi.org/10.1002/tox.22680
Hu S, He L, Chen B, You Y (2022) Apelin-13 attenuates depressive-like behaviors induced by chronic unpredictable mild stress via activating AMPK/PGC-1α/FNDC5/BDNF pathway. Pept 156:170847. https://doi.org/10.1016/j.peptides.2022.170847
Huet-Hudson YM, Andrews GK, Dey SK (1989) Cell type-specific localization of c-myc protein in the mouse uterus: modulation by steroid hormones and analysis of the periimplantation period. Endocr 125(3):1683–1690. https://doi.org/10.1210/endo-125-3-1683
Jamnongjit M, Hammes SR (2006) Ovarian steroids: the good, the bad, and the signals that raise them. Cell Cycle 5(11):1178–1183. https://doi.org/10.4161/cc.5.11.2803
Jamnongjit M, Gill A, Hammes SR (2005) Epidermal growth factor receptor signaling is required for normal ovarian steroidogenesis and oocyte maturation. Proc Natl Acad Sci 102(45):16257–16262. https://doi.org/10.1073/pnas.0508521102
Jeremy M, Gurusubramanian G, Roy VK (2019) Vitamin D3 regulates apoptosis and proliferation in the testis of D-galactose-induced aged rat model. Sci Rep 9(1):1–15
Joswig A, Gabriel HD, Kibschull M, Winterhager E (2003) Apoptosis in uterine epithelium and decidua in response to implantation: evidence for two different pathways. Reprod Biol Endocrinol 1(1):1–9. https://doi.org/10.1186/1477-7827-1-44
Kim J, Cha S, Lee MY, Hwang YJ, Yang E, Ryou C, Jung HI, Cheon YP (2018) Chronic low-dose nonylphenol or di-(2-ethylhexyl) phthalate has a different estrogen-like response in mouse uterus. Dev Reprod 22(4):379. https://doi.org/10.12717/DR.2018.22.4.379
Kurowska P, Barbe A, Różycka M, Chmielińska J, Dupont J, Rak A (2018) Apelin in reproductive physiology and pathology of different species: a critical review. Int J Endocrinol 2018:1–12
Li Y, Bai YJ, Jiang YR, Yu WZ, Shi X, Chen L, Feng J, Sun GB (2018) Apelin-13 is an early promoter of cytoskeleton and tight junction in diabetic macular edema via PI-3K/Akt and MAPK/Erksignaling pathways. Biomed Res Int 2018:3242574. https://doi.org/10.1155/2018/3242574
Liu T, Xiao B, Xiang F, Tan J, Chen Z, Zhang X, Wu C, Mao Z, Luo G, Chen X, Deng J (2020) Ultrasmall copper-based nanoparticles for reactive oxygen species scavenging and alleviation of inflammation related diseases. Nat Commun 11(1):1–16. https://doi.org/10.1038/s41467-020-16544-7
Lu J, Wang Z, Cao J, Chen Y, Dong Y (2018) A novel and compact review on the role of oxidative stress in female reproduction. Reprod Biol Endocrinol 16(1):1–18. https://doi.org/10.1186/s12958-018-0391-5
Lv S, Feng Y, Jiang Q, Lv X, Yang Y (2021) Relationship between apelin/APJ signaling, oxidative stress, and diseases. Oxidative Med Cell Longev 2021:8866725. https://doi.org/10.1155/2021/8866725
Maloney PR, Khan P, Hedrick M, Gosalia P, Milewski M, Li L et al (2012) Discovery of 4-oxo-6-((pyrimidin-2-ylthio) methyl)-4H-pyran-3-yl 4-nitrobenzoate (ML221) as a functional antagonist of the apelin (APJ) receptor. Bioorg Med Chem Lett 22(21):6656–6660. https://doi.org/10.1016/j.bmcl.2012.08.105
Melnik EA, Demin VF, Demin VA, Gmoshinski IV, Tyshko NV, Tutelyan VA (2013) Transfer of silver nanoparticles through the placenta and breast milk during in vivo experiments on rats. Acta Nat 5((3 (18))):107–115
Meng H, Chen Z, Xing G, Yuan H, Chen C, Zhao F, Zhang C, Zhao Y (2007) Ultrahigh reactivity provokes nanotoxicity: explanation of oral toxicity of nano-copper particles. Toxicol Lett 175(1-3):102–110. https://doi.org/10.1016/j.toxlet.2007.09.015
Mlyczyńska E, Myszka M, Kurowska P, Dawid M, Milewicz T, Bałajewicz-Nowak M, Kowalczyk P, Rak A (2021) Anti-apoptotic effect of apelin in human placenta: studies on BeWo cells and villous explants from third-trimester human pregnancy. Int J Mol Sci 22(5):2760. https://doi.org/10.3390/ijms22052760
Nel A, Xia T, Mädler L, Li N (2006) Toxic potential of materials at the nanolevel. Sci 311:5761–5622. https://doi.org/10.1126/science.1114397
Nita M, Grzybowski A (2016) The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxidative Med Cell Longev 2016:3164734. https://doi.org/10.1155/2016/3164734
Oberdörster G, Sharp Z, Atudorei V, Elder A, Gelein R, Kreyling W, Cox C (2004) Translocation of inhaled ultrafine particles to the brain. Inhal Toxicol 16(6-7):437–445. https://doi.org/10.1080/08958370490439597
O'Dowd BF, Heiber M, Chan A, Heng HH, Tsui LC, Kennedy JL, Shi X, Petronis A, George SR, Nguyen T (1993) A human gene that shows identity with the gene encoding the angiotensin receptor is located on chromosome 11. Gene 136(1-2):355–360. https://doi.org/10.1016/0378-1119(93)90495-O
Ohkawa H, Ohishi W, Yagi K (1979) Colorimetric method for determination of MDA activity. Biochem 95:351. https://doi.org/10.1016/0003-2697(79)90738-3
Ozkan ZS, Cilgin H, Simsek M, Cobanoglu B, Ilhan N (2013) Investigation of apelin expression in endometriosis. J Reprod Infertil 14(2):50–55
Pizzino G, Irrera N, Cucinotta M, Pallio G, Mannino F, Arcoraci V, Squadrito F, Altavilla D, Bitto A (2017) Oxidative stress: harms and benefits for human health. Oxidative Med Cell Longev 2017:8416763. https://doi.org/10.1155/2017/8416763
Poley M, Mora-Raimundo P, Shammai Y, Kaduri M, Koren L, Adir O, Shklover J, Shainsky-Roitman J, Ramishetti S, Man F, De Rosales RT (2022) Nanoparticles accumulate in the female reproductive system during ovulation affecting cancer treatment and fertility. ACS Nano 16(4):5246–5257. https://doi.org/10.1021/acsnano.1c07237
Rawle AF (2017) Characterization of nanomaterials. In: Metrology and Standardization of Nanotechnology: Protocols and Industrial Innovations, pp 129–150. https://doi.org/10.1002/9783527800308
Rempuia V, Anima B, Jeremy M, Gurusubramanian G, Pankaj PP, Kharwar RK, Roy VK (2022) Effects of metformin on the uterus of d-galactose-induced aging mice: histomorphometric, immunohistochemical localization (B-cell lymphoma 2, Bcl2-associated X protein, and active capase3), and oxidative stress study. J Exp Zool A:Ecol Integr Physiol 337(6):600–611. https://doi.org/10.1002/jez.2592
Sato T, Fukazawa Y, Kojima H, Enari M, Iguchi T, Ohta Y (1997) Apoptotic cell death during the estrous cycle in the rat uterus and vagina. Anat Rec 248(1):76–83. https://doi.org/10.1002/(SICI)1097-0185(199705)248:1<76::AID-AR9>3.0.CO;2-D
Singh A, Suragani M, Ehtesham NZ, Krishna A (2015) Localization of resistin and its possible roles in the ovary of a vespertilionid bat, Scotophilus heathi. Steroids 95:17–23. https://doi.org/10.1016/j.steroids.2014.12.018
Sizova E, Miroshnikov S, Polyakova V, Gluschenko N, Skalny A (2011) Copper nanoparticles as modulators of apoptosis and structural changes in tissues. J Biomater Nanobiotechnol 3:17006. https://doi.org/10.4236/jbnb.2012.31013
Sizochenko N, Mikolajczyk A, Syzochenko M, Puzyn T, Leszczynski J (2021) Zeta potentials (ζ) of metal oxide nanoparticles: A meta-analysis of experimental data and apredictive neural networks modeling. NanoImpact 22:100317. https://doi.org/10.1016/j.impact.2021.100317
Ssekatawa K, Byarugaba DK, Angwe MK, Wampande EM, Ejobi F, Nxumalo E et al (2022) Phyto-mediated copper oxide nanoparticles for antibacterial, antioxidant and photocatalytic performances. Front Bioeng Biotechnol 10:175. https://doi.org/10.3389/fbioe.2022.820218
Stelzer R, Hutz RJ (2009) Gold nanoparticles enter rat ovarian granulosa cells and subcellular organelles, and alter in-vitro estrogen accumulation. J Reprod Dev 55(6):685–690. https://doi.org/10.1262/jrd.20241
Than A, Zhang X, Leow MK, Poh CL, Chong SK, Chen P (2014) Apelin attenuates oxidative stress in human adipocytes. J Biol Chem 289(6):3763–3774. https://doi.org/10.1074/jbc.M113.526210
Thomas S, Sakthikumar D, Joy PA, Yoshida Y, Anantharaman MR (2006) Optically transparent magnetic nanocomposites based on encapsulated Fe3O4 nanoparticles in a sol–gel silica network. Nanotechnol 17(22):5565. https://doi.org/10.1088/0957-4484/17/22/007
Wang X, Liu X, Song Z, Shen X, Lu S, Ling Y, Kuang H (2020) Emerging roles of APLN and APELA in the physiology and pathology of the female reproductive system. PeerJ 8:e10245. https://doi.org/10.7717/peerj.10245
Wood GA, Fata JE, Watson KL, Khokha R (2007) Circulating hormones and estrous stage predict cellular and stromal remodeling in murine uterus. Reprod 133(5):1035–1044. https://doi.org/10.1530/REP-06-0302
Yang J, Hu S, Rao M, Hu L, Lei H, Wu Y, Wang Y, Ke D, Xia W, Zhu CH (2017) Copper nanoparticle-induced ovarian injury, follicular atresia, apoptosis, and geneexpression alterations in female rats. Int J Nanomedicine 18:5959–5971. https://doi.org/10.2147/IJN.S139215
Zhang Q, Paria BC (2006) Importance of uterine cell death, renewal, and their hormonal regulation in hamsters that show progesterone-dependent implantation. Endocrinol 147(5):2215–2227. https://doi.org/10.1210/en.2005-1555
Zhang CH, Wang Y, Sun QQ, Xia LL, Hu JJ, Cheng K, Wang X, Fu XX, Gu H (2018) Copper nanoparticles show obvious in vitro and in vivo reproductive toxicity via ERK mediated signaling pathway in female mice. Int J Biol Sci 14(13):1834. https://doi.org/10.7150/ijbs.27640
Zhou X, Zhao L, Luo J, Tang H, Xu M, Wang Y et al (2019) The toxic effects and mechanisms of nano-Cu on the spleen of rats. Int J Mol Sci 20(6):1469
Zou L, Cheng G, Xu C, Liu H, Wang Y, Li N, Fan X, Zhu C, Xia W (2021) Copper nanoparticles induce oxidative stress via the heme oxygenase 1 signaling pathway in vitro studies. Int J Nanomedicine 16:1565. https://doi.org/10.2147/IJN.S292319
Acknowledgements
The research infrastructure facility provided to Department of Zoology, Mizoram University, by DST-FIST program, DST, New Delhi, is greatly acknowledged.
Author information
Authors and Affiliations
Contributions
VKR, GG, BA: conceptualization, experiment design, resources generation of study. BA: performed the experiments. PM: performed TEM and selected area electron diffraction of nanoparticles. VKR, GG, BA: experimental section analysis, data analysis, writing of the manuscript
Corresponding author
Ethics declarations
Ethics approval
All experiments were performed according to the protocols approved by the Mizoram University institutional ethical committee (approval no. MZU/IAEC/2020/01), Mizoram University, Mizoram.
Consent to participate
Not applicable for this study.
Consent for publication
All authors agree to submit this manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 470 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anima, ., Mondal, P., Gurusubramanian, G. et al. Mechanistic study of copper nanoparticle (CuNP) toxicity on the mouse uterus via apelin signaling. Environ Sci Pollut Res 30, 88824–88841 (2023). https://doi.org/10.1007/s11356-023-28746-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-28746-9