Abstract
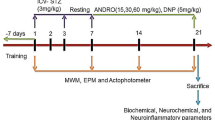
Despite the numerous treatment strategies used for Alzheimer’s disease (AD), only a few cholinesterase inhibitor drugs, such as memantine, are effective in symptomatically relieving the hallmarks of AD, providing momentary recovery of memory and cognitive decline. These available drugs do not treat the underlying causes of AD, and their chronic use is associated with serious adverse effects and disease progression. Berberine is an isoquinoline alkaloid that has been reported to possess therapeutic potential against AD. Therefore, its activity was evaluated against an aluminum chloride (AlCl3)-induced AD rat model, and a berberine-enriched extract (BEE) was used to determine if its activity is equivalent to pure berberine (PB). The rats were administered 300 mg/kg of oral AlCl3 to induce AD and were then treated with oral PB at a dosage of 50 mg/kg, BEE at a dosage of 50 mg/kg, and rivastigmine at a dosage of 1 mg/kg as a standard drug for 21 days. In this study, various parameters were assessed to evaluate cognitive functions, such as behavioral analysis, antioxidant enzyme levels, acetylcholinesterase (AChE) activity, proinflammatory cytokine levels, real-time polymerase chain reaction (RT-PCR) analysis of different biomarkers (AChE, IL-1α, IL-1β, BACE-1, TNF-α) linked to AD, and histopathological changes in the rats’ brains. After 21 days, the disease control group showed a significant decline in cognitive function, decreased levels of antioxidant enzymes, upregulated activity of the AChE enzyme, increased levels of proinflammatory cytokines, and marked elevation in mRNA expression of AD-associated biomarkers. On the other hand, the treatment groups showed significant improvements in memory deficits, elevated levels of antioxidant enzymes, reduced levels of proinflammatory cytokines, decreased AChE activity, and significant downregulation of the expression of predefined biomarkers. Histological examination of the treatment groups showed less neuroinflammation and fewer amyloid plaques compared to the disease control group. In conclusion, both PB and BEE have comparable neuroprotective potential to mitigate the pathological hallmarks of AD. However, controlled clinical trials are needed to assess their efficacy and safety.










Similar content being viewed by others
Data availability
All data generated and/or analyzed during this study are included in this published article.
Abbreviations
- AD:
-
Alzheimer’s disease
- AChE:
-
Acetylcholinesterase
- Amyloid plaques:
-
Aβ plaques
- AlCl3:
-
Aluminum chloride (disease control group)
- BEE:
-
Berberine-enriched extract
- BACE-1:
-
Beta-site amyloid precursor protein cleaving enzyme
- CNT:
-
Control group
- IL-1α:
-
Interleukin-1 alpha
- IL-1β:
-
Interleukin-1 beta
- NFTs:
-
Neurofibrillary tangles
- PB:
-
Pure berberine
- Riva:
-
Rivastigmine
- TNF-α:
-
Tumor necrosis factor alpha
References
Akbar M, Shabbir A, Rehman K, Akash MSH, Shah MA (2021) Neuroprotective potential of berberine in modulating Alzheimer’s disease via multiple signaling pathways. J Food Biochem 45:e13936. https://doi.org/10.1111/jfbc.13936
Arvanitakis Z, Wilson RS, Bienias JL, Evans DA, Bennett DA (2004) Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch Neurol 61:661–666
Bao X-Q, Li N, Wang T, Kong X-C, Tai W-J, Sun H, Zhang D (2013) FLZ Alleviates the Memory Deficits in Transgenic Mouse Model of Alzheimer’s Disease via Decreasing Beta-Amyloid Production and Tau Hyperphosphorylation. PloS One 8:e78033
Barrientos RM, Hein AM, Frank MG, Watkins LR, Maier SF (2012) Intracisternal interleukin-1 receptor antagonist prevents postoperative cognitive decline and neuroinflammatory response in aged rats. J Neurosci 32:14641–14648
Bartus RT, Dean R, Beer B, Lippa AS (1982) The cholinergic hypothesis of geriatric memory dysfunction. Science 217:408–414
Beel AJ, Sakakura M, Barrett PJ, Sanders CR (2010) Direct binding of cholesterol to the amyloid precursor protein: an important interaction in lipid–Alzheimers disease relationships? Biochim et Biophys Acta (BBA)-Mol Cell Biol Lipids 1801:975–982
BeigomHejaziyan L et al (2023) Effect of Rosa damascena Extract on Rat Model Alzheimer’s Disease: A Histopathological, Behavioral, Enzyme Activities, and Oxidative Stress Study. Evid-based Complement Alternat Med 2023:4926151. https://doi.org/10.1155/2023/4926151
Bhutada P et al (2011) Protection of cholinergic and antioxidant system contributes to the effect of berberine ameliorating memory dysfunction in rat model of streptozotocin-induced diabetes. Behav Brain Res 220:30–41
Biessels G-J, Kamal A, Ramakers GM, Urban IJ, Spruijt BM, Erkelens DW, Gispen WH (1996) Place learning and hippocampal synaptic plasticity in streptozotocin-induced diabetic rats. Diabetes 45:1259–1266
Bowen D et al (1983) Biochemical assessment of serotonergic and cholinergic dysfunction and cerebral atrophy in Alzheimer’s disease. J Neurochem 41:266–272
Chauhdary Z, Saleem U, Ahmad B, Shah S, Shah MA (2019) Neuroprotective evaluation of Tribulus terrestris L. in aluminum chloride induced Alzheimer’s disease. Pak J Pharm Sci 32:805–816
Coyle JT, Price DL, Delong MR (1983) Alzheimer’s disease: a disorder of cortical cholinergic innervation. Science 219:1184–1190
Cuello A et al (2010) Early-Stage Inflammation and Experimental Therapy in Transgenic Models of the Alzheimer-like Amyloid Pathology. Neurodegener Dis 7:96–98
de Oliveira JS et al (2016) Berberine protects against memory impairment and anxiogenic-like behavior in rats submitted to sporadic Alzheimer’s-like dementia: involvement of acetylcholinesterase and cell death. Neurotoxicology 57:241–250
Durairajan SSK et al (2012) Berberine ameliorates β-amyloid pathology, gliosis, and cognitive impairment in an Alzheimer’s disease transgenic mouse model. Neurobiol Aging 33:2903–2919
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Frantseva MV, Velazquez JLP, Hwang PA, Carlen PL (2000) Free radical production correlates with cell death in an in vitro model of epilepsy. Eur J Neurosci 12:1431–1439
Freitas RM, Vasconcelos SM, Souza FC, Viana GS, Fonteles MM (2005) Oxidative stress in the hippocampus after pilocarpine-induced status epilepticus in Wistar rats. FEBS J 272:1307–1312
Grothe MJ, Schuster C, Bauer F, Heinsen H, Prudlo J, Teipel SJ (2014) Atrophy of the cholinergic basal forebrain in dementia with Lewy bodies and Alzheimer’s disease dementia. J Neurol 261:1939–1948
He W, Wang C, Chen Y, He Y, Cai Z (2017) Berberine attenuates cognitive impairment and ameliorates tau hyperphosphorylation by limiting the self-perpetuating pathogenic cycle between NF-κB signaling, oxidative stress and neuroinflammation. Pharmacol Rep 69:1341–1348. https://doi.org/10.1016/j.pharep.2017.06.006
Huang M, Chen S, Liang Y, Guo Y (2016) The role of berberine in the multi-target treatment of senile dementia. Curr Top Med Chem 16:867–873
Jia L, Liu J, Song Z, Pan X, Chen L, Cui X, Wang M (2012) Berberine suppresses amyloid-beta-induced inflammatory response in microglia by inhibiting nuclear factor-kappaB and mitogen-activated protein kinase signalling pathways. J Pharm Pharmacol 64:1510–1521
Jiang W, Li S, Li X (2015) Therapeutic potential of berberine against neurodegenerative diseases. Sci China Life Sci 58:564–569
Kizilarslanoğlu MC et al (2015) Alzheimer disease, inflammation, and novel inflammatory marker: resistin. Turkish J Med Sci 45:1040–1046
Kulkarni S, Dhir A (2010) Berberine: a plant alkaloid with therapeutic potential for central nervous system disorders. Phytother Res 24:317–324
Li W, Yin N, Tao W, Wang Q, Fan H, Wang Z (2019) Berberine suppresses IL-33-induced inflammatory responses in mast cells by inactivating NF-κB and p38 signaling. Int immunopharmacol 66:82–90
Liu W et al (2008) Berberine inhibits aldose reductase and oxidative stress in rat mesangial cells cultured under high glucose. Arch Biochem Biophys 475:128–134
Ma X, Jiang Y, Wu A, Chen X, Pi R, Liu M, Liu Y (2010) Berberine attenuates experimental autoimmune encephalomyelitis in C57 BL/6 mice. PLoS One 5:13489
Mahdi O, Baharuldin MTH, Nor NHM, Chiroma SM, Jagadeesan S, Moklas MAM (2019) Chemicals Used for the Induction of Alzheimer’s Disease-like Cognitive Dysfunctions in Rodents Biomedical Research and Therapy 6:3460–3484
Matsumoto C, Miedema MD, Ofman P, Gaziano JM, Sesso HD (2014) An expanding knowledge of the mechanisms and effects of alcohol consumption on cardiovascular disease. J Cardiopulm Rehabil Prev 34:159–171
Mook-Jung I et al (1999) Protective effects of asiaticoside derivatives against beta-amyloid neurotoxicity. J Neurosci Res 58:417–425
Naz I, Masoud MS, Chauhdary Z, Shah MA, Panichayupakaranant P (2022) Anti-inflammatory potential of berberine-rich extract via modulation of inflammation biomarkers. J Food Biochem 46(12):e14389
Nichols E et al (2019) Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Neurol 18:88–106
Pamplona FA, Pandolfo P, Duarte FS, Takahashi RN, Prediger RD (2010) Altered emotionality leads to increased pain tolerance in amyloid β (Aβ1–40) peptide-treated mice. Behav Brain Res 212:96–102
Panahi N, Mahmoudian M, Mortazavi P, Hashjin GS (2013) Effects of Berberine on β-Secretase Activity in a Rabbit Model of Alzheimer’s Disease Archives of Medical Science: AMS 9:146
Parihar M, Hemnani T (2004) Alzheimer’s Disease Pathogenesis and Therapeutic Interventions Journal of Clinical Neuroscience 11:456–467
Pellow S, Chopin P, File SE, Briley M (1985) Validation of open: closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. J Neurosci Methods 14:149–167
Pinton S, da Rocha JT, Gai BM, Nogueira CW (2011) Sporadic dementia of Alzheimer’s type induced by streptozotocin promotes anxiogenic behavior in mice. Behav Brain Res 223:1–6
Popiołek I, Niziołek A, Kamiński K, Kwolek U, Nowakowska M, Szczubiałka K (2019) Cellular delivery and enhanced anticancer activity of berberine complexed with a cationic derivative of γ–cyclodextrin. Bioorg Med Chem 27:1414–1420
Prince MJ, Wimo A, Guerchet MM, Ali GC, Wu Y-T, Prina M (2015) World alzheimer report 2015-the global impact of dementia: an analysis of prevalence, incidence, cost and trends. Alzheimer’s disease international. http://www.alz.co.uk/research/world-report-2015
Ramakers I et al (2013) Anxiety is related to Alzheimer cerebrospinal fluid markers in subjects with mild cognitive impairment. Psychol Med 43:911
Sahibzada MUK, Sadiq A, Faidah HS, Khurram M, Amin MU, Haseeb A, Kakar M (2018) Berberine nanoparticles with enhanced in vitro bioavailability: characterization and antimicrobial activity. Drug Design, Dev Ther 12:303
Satija S, Bansal P, Dureja H, Garg M (2015) Microwave assisted extraction of Tinospora cordifolia and optimization through central composite design. J Biol Sci 15:106
Schelterns P, Feldman H (2003) Treatment of Alzheimer’s disease; current status and new perspectives. Lancet Neurol 2:539–547
Shoji M et al (1996) Systemic overexpression of a C-terminal fragment of human amyloid beta-protein precursor causes accumulation of Alzheimer beta-amyloid fibrils in pancreas of transgenic mice. Gerontology 42:48–56
Thenmozhi AJ, Raja TRW, Janakiraman U, Manivasagam T (2015) Neuroprotective effect of hesperidin on aluminium chloride induced Alzheimer’s disease in Wistar rats. Neurochem Res 40:767–776
Tillerson JL, Miller GW (2003) Grid performance test to measure behavioral impairment in the MPTP-treated-mouse model of parkinsonism. J Neurosci Methods 123:189–200
Vacas S, Degos V, Tracey KJ, Maze M (2014) High-mobility group box 1 protein initiates postoperative cognitive decline by engaging bone marrow–derived macrophages. Anesthesiology 120:1160–1167
Viegas Junior C, Bolzani VdS, Furlan M, Fraga CAM, Barreiro EJ (2004) natural products as candidates for useful drugs in the treatment of Alzheimer’s disease. Quim Nova 27:655–660
Vuddanda PR, Chakraborty S, Singh S (2010) Berberine: a potential phytochemical with multispectrum therapeutic activities. Expert Opin Investig Drugs 19:1297–1307
Walf AA, Frye CA (2007) The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat Protoc 2:322–328
Yamada A, Akimoto H, Kagawa S, Guillemin GJ, Takikawa O (2009) Proinflammatory cytokine interferon-gamma increases induction of indoleamine 2,3-dioxygenase in monocytic cells primed with amyloid beta peptide 1–42: implications for the pathogenesis of Alzheimer’s disease. J Neurochem 110:791–800. https://doi.org/10.1111/j.1471-4159.2009.06175.x
Yankner BA, Lu T (2009) Amyloid beta-protein toxicity and the pathogenesis of Alzheimer disease. J Biol Chem 284:4755–4759. https://doi.org/10.1074/jbc.R800018200
Zanin M, Takahashi RN (1994) Sex difference in sensitization to the locomotor effects of mazindol in rats. Brain Res Bull 34:385–387
Zerrouki K, Djebli N, Ozkan EE, Ozsoy N, Gul O, Mat A (2016) Hypericum perforatum improve memory and learning in Alzheimer’s model:(experimental study in mice). Int J Pharm Pharm Sci 8:49–57
Zhang J, Yang J-Q, He B-C, Zhou Q-X, Yu H-R, Tang Y, Liu B-Z (2009) Berberine and total base from rhizoma coptis chinensis attenuate brain injury in an aluminum-induced rat model of neurodegenerative disease. Saudi Med J 30:760–766
Acknowledgements
The authors would like to express their appreciation to the Deanship of Scientific Research at King Khalid University, Saudi Arabia, for providing funding for this research through the research group program under grant number RGP-2/370/44.
Author information
Authors and Affiliations
Contributions
MSHA, MAS, PP, and KR designed the research. MA and MSHA performed the experiment. KR, MI, MAS, and MAA analyzed the findings and prepared manuscript. All the authors read the final draft approved for publication.
Corresponding author
Ethics declarations
Ethical approval
This study was ethically approved by the Institutional Review Board (GCUF/ERC/2192) of Government College University Faisalabad (GCUF).
Consent to participate
N/A.
Consent for publication
N/A.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akash, M.S.H., Akbar, M., Rehman, K. et al. Biochemical profiling of berberine-enriched extract in aluminum chloride induced oxidative damage and neuroinflammation. Environ Sci Pollut Res 30, 85263–85275 (2023). https://doi.org/10.1007/s11356-023-28392-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-28392-1