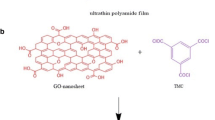
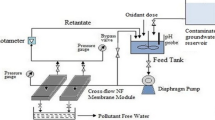
Abstract
Polyether sulfone (PES)-based thin-film nanofiltration (TFN) membranes embedded with ferric hydroxide (FeIII(OH)x) functionalized graphene oxide (GO) nanoparticles were fabricated through interfacial polymerization for a generalized application in removal of a plethora of anionic and toxic water contaminants. Following the most relevant characterization, the newly synthesized membranes were fitted in a novel flat sheet cross-flow module, for experimental investigation on purification of live contaminated groundwater collected from different affected areas. The separation performances of the membranes in the flat sheet cross-flow module demonstrated that GOF membranes had higher selectivity for monovalent and divalent salt rejections than pristine GO membranes. Furthermore, both membranes were tested for simultaneously removing widely occurring hazardous ions of heavy metals and metalloids in groundwater, such as arsenic, selenium, chromium, and fluoride. Compared to the pristine GO and the reported membranes in the literature, the GOF membrane exhibited remarkable performance in terms of rejection efficiency (Cr (VI): 97.2%, Se (IV): 96.6%, As(V): 96.3%, F− 88.4%) and sustained flux of 184 LMH (Lm−2 h−1) at an optimum transmembrane pressure of 16 bar. The investigated membrane module equipped with the GOF membrane proved to be a low-cost system with higher anionic rejection and sustained high flux at a comprehensive pH range, as evident over long hours of study vis-à-vis reported systems.
Graphical Abstract














Similar content being viewed by others
Data availability
All data generated during investigations are included in the manuscript. Any additional relevant data will be made available on request.
References
Abdi S, Nasiri M, Yuan S et al (2021) Fabrication of PES-based super-hydrophilic ultrafiltration membranes by combining hydrous ferric oxide particles and UV irradiation. Sep Purif Technol 259:118132. https://doi.org/10.1016/j.seppur.2020.118132
Association APH, Association AWW, Federation WPC (1989) Standard methods for the examination of water and wastewater. 17th edition. Am Public Heal Assoc
Bacquart T, Frisbie SH, Mitchell E et al (2015) Multiple inorganic toxic substances contaminating the groundwater of Myingyan Township, Myanmar: arsenic, manganese, fluoride, iron, and uranium. Sci Total Environ 517:232–245. https://doi.org/10.1016/j.scitotenv.2015.02.038
Bagheripour E, Moghadassi AR, Hosseini SM et al (2018) Novel composite graphene oxide/chitosan nanoplates incorporated into PES based nanofiltration membrane: chromium removal and antifouling enhancement. J Ind Eng Chem 62:311–320. https://doi.org/10.1016/J.JIEC.2018.01.009
BIS (2012) Indian standard drinking water specification (Second Revision). Bur Indian Stand IS 10500:1–11
Braghetta A, DiGiano FA, Ball WP (1997) Nanofiltration of natural organic matter: pH and ionic strength effects. J Environ Eng 123:628–641. https://doi.org/10.1061/(ASCE)0733-9372(1997)123:7(628)
Eklund L, Persson I (2014) Structure and hydrogen bonding of the hydrated selenite and selenate ions in aqueous solution. Dalt Trans 43:6315–6321. https://doi.org/10.1039/C3DT53468E
Giagnorio M, Steffenino S, Meucci L et al (2018) Design and performance of a nanofiltration plant for the removal of chromium aimed at the production of safe potable water. J Environ Chem Eng 6:4467–4475. https://doi.org/10.1016/J.JECE.2018.06.055
Gurunathan P, Hari S, Suseela SB et al (2019) Production, characterization and effectiveness of cellulose acetate functionalized ZnO nanocomposite adsorbent for the removal of Se (VI) ions from aqueous media. Environ Sci Pollut Res 26:528–543. https://doi.org/10.1007/S11356-018-3472-2
Han Y, Jiang Y, Gao C (2015) High-flux graphene oxide nanofiltration membrane intercalated by carbon nanotubes. ACS Appl Mater Interfaces 7:8147–8155. https://doi.org/10.1021/acsami.5b00986
He L, Dumée LF, Feng C et al (2015) Promoted water transport across graphene oxide-poly(amide) thin film composite membranes and their antibacterial activity. Desalination 365:126–135. https://doi.org/10.1016/j.desal.2015.02.032
He Y, Liu J, Han G, Chung TS (2018a) Novel thin-film composite nanofiltration membranes consisting of a zwitterionic co-polymer for selenium and arsenic removal. J Memb Sci 555:299–306. https://doi.org/10.1016/j.memsci.2018.03.055
He Y, Tang YP, Chung TS (2016) Concurrent removal of selenium and arsenic from water using polyhedral oligomeric silsesquioxane (POSS)-polyamide thin-film nanocomposite nanofiltration membranes. Ind Eng Chem Res 55:12929–12938. https://doi.org/10.1021/acs.iecr.6b04272
He Y, Tang YP, Ma D, Chung TS (2017) UiO-66 incorporated thin-film nanocomposite membranes for efficient selenium and arsenic removal. J Memb Sci 541:262–270. https://doi.org/10.1016/j.memsci.2017.06.061
He Y, Xiang Y, Zhou Y et al (2018b) Selenium contamination, consequences and remediation techniques in water and soils: a review. Environ Res 164:288–301. https://doi.org/10.1016/j.envres.2018.02.037
Igbinigun E, Fennell Y, Malaisamy R et al (2016) Graphene oxide functionalized polyethersulfone membrane to reduce organic fouling. J Memb Sci 514:518–526. https://doi.org/10.1016/j.memsci.2016.05.024
Jadhav SV, Marathe KV, Rathod VK (2016) A pilot scale concurrent removal of fluoride, arsenic, sulfate and nitrate by using nanofiltration: competing ion interaction and modelling approach. J Water Process Eng 13:153–167. https://doi.org/10.1016/J.JWPE.2016.04.008
Kabir F, Chowdhury S (2017) Arsenic removal methods for drinking water in the developing countries: technological developments and research needs. Environ Sci Pollut Res 24:24102–24120. https://doi.org/10.1007/s11356-017-0240-7
Kazemi M, Peyravi M, Jahanshahi M (2020) Multilayer UF membrane assisted by photocatalytic NZVI@TiO2 nanoparticle for removal and reduction of hexavalent chromium. J Water Process Eng 37:101183. https://doi.org/10.1016/J.JWPE.2020.101183
Kesieme UK, Milne N, Aral H et al (2013) Economic analysis of desalination technologies in the context of carbon pricing, and opportunities for membrane distillation. Desalination 323:66–74. https://doi.org/10.1016/J.DESAL.2013.03.033
Kumari P, Tripathi KM, Jangir LK et al (2021) Recent advances in application of the graphene-based membrane for water purification. Mater Today Chem 22:100597. https://doi.org/10.1016/J.MTCHEM.2021.100597
Lai GS, Lau WJ, Goh PS et al (2018) Tailor-made thin film nanocomposite membrane incorporated with graphene oxide using novel interfacial polymerization technique for enhanced water separation. Chem Eng J 344:524–534. https://doi.org/10.1016/J.CEJ.2018.03.11
Lakhotia SR, Mukhopadhyay M, Kumari P (2019) Iron oxide (FeO) nanoparticles embedded thin-film nanocomposite nanofiltration (NF) membrane for water treatment. Sep Purif Technol 211:98–107. https://doi.org/10.1016/J.SEPPUR.2018.09.034
Lemly AD (1997) Environmental implications of excessive selenium: a review. Biomed Environ Sci 10:415–435
Liu F, Huang JC, Zhou C et al (2019) Development of an algal treatment system for selenium removal: effects of environmental factors and post-treatment processing of Se-laden algae. J Hazard Mater 365:546–554. https://doi.org/10.1016/j.jhazmat.2018.11.017
Liu G, Jin W, Xu N (2015) Graphene-based membranes. Chem Soc Rev 44:5016–5030. https://doi.org/10.1039/C4CS00423J
Mahler J, Persson I, Herbert R (2013) Hydration of arsenic oxyacid species. Dalt Trans 42:1364–1377. https://doi.org/10.1039/c2dt31906c
Malhotra M, Pal M, Pal P (2020) A response surface optimized nanofiltration-based system for efficient removal of selenium from drinking Water. J Water Process Eng 33:101007. https://doi.org/10.1016/j.jwpe.2019.101007
Manna AK, Sen M, Martin AR, Pal P (2010) Removal of arsenic from contaminated groundwater by solar-driven membrane distillation. Environ Pollut 158:805–811. https://doi.org/10.1016/J.ENVPOL.2009.10.002
Nishimura T, Hashimoto H, Nakayama M (2007) Removal of selenium(VI) from aqueous solution with polyamine-type weakly basic ion exchange resin. Sep Sci Technol 42:3155–3167. https://doi.org/10.1080/01496390701513107
Pakade VE, Tavengwa NT, Madikizela LM (2019) Recent advances in hexavalent chromium removal from aqueous solutions by adsorptive methods. RSC Adv 26142. https://doi.org/10.1039/c9ra05188k
Pal M, Malhotra M, Mandal MK et al (2020) Recycling of wastewater from tannery industry through membrane-integrated hybrid treatment using a novel graphene oxide nanocomposite. J Water Process Eng 36:101324. https://doi.org/10.1016/j.jwpe.2020.101324
Pal M, Mondal MK, Paine TK (2018) Pal P (2018) Purifying arsenic and fluoride-contaminated water by a novel graphene-based nanocomposite membrane of enhanced selectivity and sustained flux. Environ Sci Pollut Res 2517(25):16579–16589. https://doi.org/10.1007/S11356-018-1829-1
Pal P (2020) Membrane-based technology for groundwater treatment. Membrane-based technologies for environmental pollution control. Elsevier Science Publishers B.V, New York
Pal P, Chakraborty S, Roy M (2012) Arsenic separation by a membrane-integrated hybrid treatment system: modeling, simulation, and techno-economic evaluation. Sep Sci Technol 47:1091–1101. https://doi.org/10.1080/01496395.2011.652754
Pal P, Sen M, Manna A et al (2009) Contamination of groundwater by arsenic: a review of occurrence, causes, impacts, remedies and membrane-based purification. J Integr Environ Sci 6:295–316. https://doi.org/10.1080/19438150903185077
Pino GH, de Mesquita LMS, Torem ML et al (2006) Biosorption of heavy metals by powder of green coconut shell. Sep Sci Technol 41:3141–3153
Qiu Z, Tian Q, Zhang T et al (2020) Fabrication of dynamic zero-valent iron/MnO2 nanowire membrane for efficient and recyclable selenium separation. Sep Purif Technol 230:115847. https://doi.org/10.1016/J.SEPPUR.2019.115847
Rajamohan N, Bosu S, Rajasimman M, Varjani S (2023) Environmental remediation of selenium using surface modified carbon nano tubes — characterization, influence of variables, equilibrium and kinetic analysis. Environ Res 216:114629. https://doi.org/10.1016/J.ENVRES.2022.114629
Rathi BS, Kumar PS, Show PL (2021) A review on effective removal of emerging contaminants from aquatic systems: current trends and scope for further research. J Hazard Mater 409:124413. https://doi.org/10.1016/j.jhazmat.2020.124413
Richards LA, Richards BS, Schäfer AI (2011) Renewable energy powered membrane technology: salt and inorganic contaminant removal by nanofiltration/reverse osmosis. J Memb Sci 369:188–195. https://doi.org/10.1016/j.memsci.2010.11.069
Shahriary L, Athawale AA (2014) Graphene oxide synthesized by using modified hummers approach. Int J Renew Energy Environ Eng 02:58–63
Shukla AK, Alam J, Alhoshan M et al (2018) Removal of heavy metal ions using a carboxylated graphene oxide-incorporated polyphenylsulfone nanofiltration membrane. Environ Sci Water Res Technol 4:438–448. https://doi.org/10.1039/c7ew00506g
Singh S, Anil AG, Khasnabis S et al (2022) Sustainable removal of Cr(VI) using graphene oxide-zinc oxide nanohybrid: adsorption kinetics, isotherms and thermodynamics. Environ Res 203:111891. https://doi.org/10.1016/J.ENVRES.2021.111891
Suriyaraj SP, Selvakumar R (2016) Advances in nanomaterial based approaches for enhanced fluoride and nitrate removal from contaminated water. RSC Adv 6:10565–10583. https://doi.org/10.1039/C5RA24789F
Taghipour N, Amini H, Mosaferi M et al (2016) National and sub-national drinking water fluoride concentrations and prevalence of fluorosis and of decayed, missed, and filled teeth in Iran from 1990 to 2015: a systematic review. Environ Sci Pollut Res 23:5077–5098. https://doi.org/10.1007/s11356-016-6160-0
Tan LC, Nancharaiah YV, van Hullebusch ED, Lens PNL (2016) Selenium: environmental significance, pollution, and biological treatment technologies. Biotechnol Adv 34:886–907. https://doi.org/10.1016/j.biotechadv.2016.05.005
The European Parliament and the Council of the European Union (2020) Directive (EU) 2020/2184 of the European Parliament and of the Council. Off J Eur Union 2019:1–62
USEPA (2009) National Primary Drinking Water Regulations | US EPA. https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations#one. Accessed 12 May 2022
Wei Y, Zhang Y, Gao X et al (2018) Multilayered graphene oxide membranes for water treatment: a review. Carbon N Y 139:964–981. https://doi.org/10.1016/j.carbon.2018.07.040
Wilkie JA, Hering JG (1996) Adsorption of arsenic onto hydrous ferric oxide: effects of adsorbate/adsorbent ratios and co-occurring solutes. Colloids Surf A Physicochem Eng Asp 107:97–110. https://doi.org/10.1016/0927-7757(95)03368-8
Willcox JAL, Kim HJ (2017) Molecular dynamics study of water flow across multiple layers of pristine, pxidized, and mixed regions of graphene oxide. ACS Nano 11:2187–2193. https://doi.org/10.1021/ACSNANO.6B08538
World Health Organization (2022) Guidelines for drinking-water quality (GDWQ), 4th edn. World Health Organisation
Xu Z, Wu T, Shi J et al (2016) Manipulating migration behavior of magnetic graphene oxide via magnetic field induced casting and phase separation toward high-performance hybrid ultrafiltration membranes. ACS Appl Mater Interfaces 8:18418–18429. https://doi.org/10.1021/acsami.6b04083
Yoon J, Amy G, Chung J et al (2009) Removal of toxic ions (chromate, arsenate, and perchlorate) using reverse osmosis, nanofiltration, and ultrafiltration membranes. Chemosphere 77:228–235. https://doi.org/10.1016/J.CHEMOSPHERE.2009.07.028
Yuan Y, Gao X, Wei Y et al (2017) Enhanced desalination performance of carboxyl functionalized graphene oxide nanofiltration membranes. Desalination 405:29–39. https://doi.org/10.1016/j.desal.2016.11.024
Zamora-Ledezma C, Negrete-Bolagay D, Figueroa F et al (2021) Heavy metal water pollution: a fresh look about hazards, novel and conventional remediation methods. Environ Technol Innov 22:101504. https://doi.org/10.1016/J.ETI.2021.101504
Zhuang Y, Liu Q, Kong Y et al (2019) Enhanced antibiotic removal through a dual-reaction-center Fenton-like process in 3D graphene based hydrogels. Environ Sci Nano 6:388. https://doi.org/10.1039/c8en01339j
Zinadini S, Zinatizadeh AA, Rahimi M et al (2014) Novel high flux antifouling nanofiltration membranes for dye removal containing carboxymethyl chitosan coated Fe3O4 nanoparticles. Desalination 349:145–154. https://doi.org/10.1016/j.desal.2014.07.007
Acknowledgements
The authors acknowledge fellowship support from the Ministry of Education, Government of India, and Department of Science and Technology, GOI, for infrastructural support.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Meenakshi Malhotra, Madhubonti Pal, Sankha Chakrabortty, and Parimal Pal. The first draft of the manuscript was written by Meenakshi Malhotra, and all authors commented on previous versions of the manuscript and contributed to substantial improvements. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Ethical approval for this work is irrelevant as the work does not involve human beings/animals in the investigations.
Consent to participate
All authors have consent to participate in this work. Concerned Institutes do not require such consent.
Consent for publication
All authors have consent to this publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Angeles Blanco
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Malhotra, M., Pal, M., Chakrabortty, S. et al. A single functionalized graphene nanocomposite in cross flow module for removal of multiple toxic anionic contaminants from drinking water. Environ Sci Pollut Res 30, 65250–65266 (2023). https://doi.org/10.1007/s11356-023-26937-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26937-y