Abstract
There has been a substantially increasing demand for energy critical elements (ECEs) in recent years as energy-related technology has advanced rapidly. Spent catalysts are known as potential sources of ECCs such as Ni, Co, Mo, W, V, and rare earth elements. This study developed a novel environmentally friendly process for recovering cobalt and molybdenum from spent hydroprocessing catalysts using deep eutectic solvents (DESs). DESs based on p-toluenesulfonic acid achieved high metal extraction at 100 °C and a pulp density of 20 g/L for 48 h which 93% of cobalt and 87% of molybdenum were dissolved. FT-IR and H-NMR analyses were conducted to determine whether hydrogen bonds form between p-toluenesulfonic acid-based DES components. Leaching kinetic models were also developed for DES systems. The experimental results were well-matched with the shrinking core models. The leaching controlling step of DES-1 was determined to be the diffusion through the product layer based on kinetic studies, with an activation energy of 22.56 kJ/mol for Co and 29.34 kJ/mol for Mo in DES-1. Similarly, the mixed control reaction with an activation energy of 38.09 kJ/mol for Co and 31.48 kJ/mol for Mo in DES-2 was found to control the leaching kinetic mechanism of the DES-2 sample.
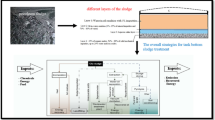
Graphical Abstract








Similar content being viewed by others
Data availability
All data are true and valid and can be available by request.
References
Abbott AP, Boothby D, Capper G et al (2004) Deep eutectic solvents formed between choline chloride and carboxylic acids: versatile alternatives to ionic liquids. J Am Chem Soc 126:9142–9147. https://doi.org/10.1021/ja048266j
Abbott AP, Capper G, Davies DL et al (2006) Solubility of metal oxides in deep eutectic solvents based on choline chloride. J Chem Eng Data 51:1280–1282. https://doi.org/10.1021/je060038c
Abbott AP, Collins J, Dalrymple I et al (2009) Processing of electric arc furnace dust using deep eutectic solvents. Aust J Chem 62:341–347. https://doi.org/10.1071/CH08476
Abbott AP, Capper G, Davies DL, et al (2003) Novel solvent properties of choline chloride/urea mixtures. Chem Commun 70–71. https://doi.org/10.1039/b210714g
Abdollahi H, Saneie R, Shafaei SZ et al (2021) Bioleaching of cobalt from magnetite-rich cobaltite-bearing ore. Hydrometallurgy 204:105727. https://doi.org/10.1016/j.hydromet.2021.105727
Akcil A, Vegliò F, Ferella F et al (2015) A review of metal recovery from spent petroleum catalysts and ash. Waste Manag 45:420–433. https://doi.org/10.1016/j.wasman.2015.07.007
Amiri F, Yaghmaei S, Mousavi SM (2011) Bioleaching of tungsten-rich spent hydrocracking catalyst using Penicillium simplicissimum. Bioresour Technol 102:1567–1573. https://doi.org/10.1016/j.biortech.2010.08.087
Anand S, Das SC, Das RP, Jena PK (1988) Leaching of manganese nodules at elevated temperature and pressure in the presence of oxygen. Hydrometallurgy 20:155–167. https://doi.org/10.1016/0304-386X(88)90049-7
Arslanoğlu H, Yaraş A (2019) Recovery of precious metals from spent Mo–Co–Ni/Al2O3 catalyst in organic acid medium: process optimization and kinetic studies. Pet Sci Technol 37:2081–2093. https://doi.org/10.1080/10916466.2019.1618867
Bakkar A (2014) Recycling of electric arc furnace dust through dissolution in deep eutectic ionic liquids and electrowinning. J Hazard Mater 280:191–199. https://doi.org/10.1016/j.jhazmat.2014.07.066
Bakkar A, Neubert V (2019) Recycling of cupola furnace dust: extraction and electrodeposition of zinc in deep eutectic solvents. J Alloys Compd 771:424–432. https://doi.org/10.1016/j.jallcom.2018.08.246
Beolchini F, Fonti V, Ferella F, Vegliò F (2010) Metal recovery from spent refinery catalysts by means of biotechnological strategies. J Hazard Mater 178:529–534. https://doi.org/10.1016/j.jhazmat.2010.01.114
Binnemans K, Jones PT (2017) Solvometallurgy: an emerging branch of extractive metallurgy. J Sustain Metall 3:570–600. https://doi.org/10.1007/s40831-017-0128-2
Chauhan G, Pant KK, Nigam KDP (2013) Metal recovery from hydroprocessing spent catalyst: a green chemical engineering approach. Ind Eng Chem Res 52:16724–16736. https://doi.org/10.1021/ie4024484
Chen Y, Lu Y, Liu Z et al (2020) Efficient dissolution of lithium-ion batteries cathode LiCoO2by polyethylene glycol-based deep eutectic solvents at mild temperature. ACS Sustain Chem Eng 8:11713–11720. https://doi.org/10.1021/acssuschemeng.0c03624
Dai Y, van Spronsen J, Witkamp GJ et al (2013) Natural deep eutectic solvents as new potential media for green technology. Anal Chim Acta 766:61–68. https://doi.org/10.1016/j.aca.2012.12.019
Ebrahimi E, Abdollahi H, Ziaedin Shafaei S, et al (2023) Green extraction of pure ferromagnetic nickel from spent hydroprocessing catalysts via deep eutectic solvents. Sep Purif Technol 123461. https://doi.org/10.1016/j.seppur.2023.123461
Eyman DP, Drago RS (1966) Nuclear magnetic resonance studies of hydrogen bonding. J Am Chem Soc 88:1617–1620. https://doi.org/10.1021/ja00960a005
Fogler HS (2016) Elements of Chemical Reaction Engineering, 5 th. Pearson, Philadelphia
Free ML (2022) Hydrometallurgy fundamentals and applications, 3rd edn. Springer, Cham, pp 23–66. https://doi.org/10.1007/978-3-030-88087-3
Gholami RM, Borghei SM, Mousavi SM (2011) Bacterial leaching of a spent Mo–Co–Ni refinery catalyst using Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans. Hydrometallurgy 106:26–31. https://doi.org/10.1016/j.hydromet.2010.11.011
Habashi F (2017) Principles of extractive metallurgy. Routledge, Québec, Québec City Canada
Habashi F (1999) Principles of EXTRACTIVE METALLURGY. Routledge
Jiang J, Bai X, Zhao X et al (2019) Poly-quasi-eutectic solvents (PQESs): versatile solvents for dissolving metal oxides. Green Chem 21:5571–5578. https://doi.org/10.1039/c9gc02604e
Kim HI, Park KH, Mishra D (2009) Sulfuric acid baking and leaching of spent Co-Mo/Al2O3 catalyst. J Hazard Mater 166:1540–1544. https://doi.org/10.1016/j.jhazmat.2008.11.051
Le MN, Lee MS (2020) Hydrometallurgical treatment of elemental sulfur in spent catalysts by aqueous and nonaqueous solutions at low temperature. Miner Process Extr Metall Rev 41:217–226. https://doi.org/10.1080/08827508.2019.1634563
Le MN, Lee MS (2021) A review on hydrometallurgical processes for the recovery of valuable metals from spent catalysts and life cycle analysis perspective. Miner Process Extr Metall Rev 42:335–354. https://doi.org/10.1080/08827508.2020.1726914
Levenspiel O (1998) Chemical reaction engineering, 3rd edn. John Wiley & Sons, India
Lim MSW, Yang TCK, Yap YH et al (2021) Intensification and optimisation of nickel recovery from spent hydrogenation catalysts via ultrasound-augmented hydrometallurgy. J Environ Chem Eng 9:105771. https://doi.org/10.1016/j.jece.2021.105771
Liu C, Yan Q, Zhang X et al (2020) Efficient recovery of end-of-life ndfeb permanent magnets by selective leaching with deep eutectic solvents. Environ Sci Technol 54:10370–10379. https://doi.org/10.1021/acs.est.0c03278
Medvedev AS, Malochkina NV (2007) Sublimation of molybdenum trioxide from exhausted catalysts employed for the purification of oil products. Russ J Non-Ferrous Met 48:114–117. https://doi.org/10.3103/S1067821207020071
Meshram P, Pandey BD, Mankhand TR (2015) Leaching of base metals from spent Ni-metal hydride batteries with emphasis on kinetics and characterization. Hydrometallurgy 158:172–179. https://doi.org/10.1016/j.hydromet.2015.10.028
MeshramAbhilash P (2020) Recovery and recycling of cerium from primary and secondary resources- a critical review. Miner Process Extr Metall Rev 41:279–310. https://doi.org/10.1080/08827508.2019.1677647
Mishra D, Kim DJ, Ralph DE et al (2007) Bioleaching of vanadium rich spent refinery catalysts using sulfur oxidizing lithotrophs. Hydrometallurgy 88:202–209. https://doi.org/10.1016/j.hydromet.2007.05.007
Mohapatra D, Park KH (2007) Selective recovery of Mo, Co and Al from spent Co/Mo/γ -Al 2O3 catalyst: effect of calcination temperature. J Environ Sci Health-Part A Toxic/Hazard Subst Environ Eng 42:507–515. https://doi.org/10.1080/10934520601188409
Nguyen TH, Lee MS (2015) Development of a hydrometallurgical process for the recovery of calcium molybdate and cobalt oxalate powders from spent hydrodesulphurization (HDS) catalyst. J Clean Prod 90:388–396. https://doi.org/10.1016/j.jclepro.2014.11.048
Nuss P, Blengini GA (2018) Towards better monitoring of technology critical elements in Europe: coupling of natural and anthropogenic cycles. Sci Total Environ 613–614:569–578. https://doi.org/10.1016/j.scitotenv.2017.09.117
Oza R, Patel S (2011) Recovery of nickel from spent Ni/Al 2 O 3 catalysts using acid leaching, chelation and ultrasonication. Res J Recent Sci ResJRecentSci 1:434–443
Panagopoulos A (2022) Brine management (saline water & wastewater effluents): sustainable utilization and resource recovery strategy through minimal and zero liquid discharge (MLD & ZLD) desalination systems. Chem Eng Process-Process Intensif 176:108944. https://doi.org/10.1016/j.cep.2022.108944
Panagopoulos A, Giannika V (2022) Decarbonized and circular brine management/valorization for water & valuable resource recovery via minimal/zero liquid discharge (MLD/ZLD) strategies. J Environ Manage 324:116239. https://doi.org/10.1016/j.jenvman.2022.116239
Park KH, Reddy BR, Mohapatra D, Nam C-W (2006) Hydrometallurgical processing and recovery of molybdenum trioxide from spent catalyst. Int J Miner Process 80:261–265. https://doi.org/10.1016/j.minpro.2006.05.002
Pellecchia M (2005) Solution nuclear magnetic resonance spectroscopy techniques for probing intermolecular interactions. Chem Biol 12:961–971. https://doi.org/10.1016/j.chembiol.2005.08.013
Rezaee M, Abdollahi H, Saneie R et al (2022) A cleaner approach for high-efficiency regeneration of base and precious metals from waste printed circuit boards through stepwise oxido-acidic and thiocyanate leaching. Chemosphere 298:134283. https://doi.org/10.1016/j.chemosphere.2022.134283
Rezaee M, Saneie R, Mohammadzadeh A et al (2023) Eco-friendly recovery of base and precious metals from waste printed circuit boards by step-wise glycine leaching: process optimization, kinetics modeling, and comparative life cycle assessment. J Clean Prod 389:136016. https://doi.org/10.1016/j.jclepro.2023.136016
Rocchetti L, Fonti V, Vegliò F, Beolchini F (2013) An environmentally friendly process for the recovery of valuable metals from spent refinery catalysts. Waste Manag Res J a Sustain Circ Econ 31:568–576. https://doi.org/10.1177/0734242X13476364
Rodriguez Rodriguez N, MacHiels L, Binnemans K (2019) P-Toluenesulfonic acid-based deep-eutectic solvents for solubilizing metal oxides. ACS Sustain Chem Eng 7:3940–3948. https://doi.org/10.1021/acssuschemeng.8b05072
Ruiz V, Meux E, Schneider M, Georgeaud V (2011) Hydrometallurgical treatment for valuable metals recovery from spent CoMo/Al2O3 Catalyst. 2. Oxidative leaching of an unroasted catalyst using H2O2. Ind Eng Chem Res 50:5307–5315. https://doi.org/10.1021/ie102428r
Santana-Mayor Á, Rodríguez-Ramos R, Herrera-Herrera AV et al (2021) Deep eutectic solvents. The new generation of green solvents in analytical chemistry. TrAC-Trends Anal Chem 134:116108. https://doi.org/10.1016/j.trac.2020.116108
Shahbazi M, Abdollahi H, Ziaedin S et al (2022) Desulfurization of Tabas coal using chemical (Meyers, Molten caustic leaching) and biological (bioleaching) methods. Int J Min Geo-Engineering 1:249–255. https://doi.org/10.22059/IJMGE.2022.345748.594981
Stanislaus A, Marafi M, Absi-Halabi M (1993) Studies on the rejuvenation of spent catalysts: effectiveness and selectivity in the removal of foulant metals from spent hydroprocessing catalysts in coked and decoked forms. Appl Catal a, Gen 105:195–203. https://doi.org/10.1016/0926-860X(93)80248-O
Tang S, Zhang M, Guo M (2022) A novel deep-eutectic solvent with strong coordination ability and low viscosity for efficient extraction of valuable metals from spent lithium-ion batteries. ACS Sustain Chem Eng 10:975–985. https://doi.org/10.1021/acssuschemeng.1c06902
Tayar SP, Yeste MP, Ramírez M et al (2020) Nickel recycling through bioleaching of a Ni/Al2O3 commercial catalyst. Hydrometallurgy 195:105350. https://doi.org/10.1016/j.hydromet.2020.105350
Tran MK, Rodrigues MTF, Kato K et al (2019) Deep eutectic solvents for cathode recycling of Li-ion batteries. Nat Energy 4:339–345. https://doi.org/10.1038/s41560-019-0368-4
Tran TT, Liu Y, Lee MS (2021) Recovery of pure molybdenum and vanadium compounds from spent petroleum catalysts by treatment with ionic liquid solution in the presence of oxidizing agent. Sep Purif Technol 255:117734. https://doi.org/10.1016/j.seppur.2020.117734
Zhao Z, Guo M, Zhang M (2015) Extraction of molybdenum and vanadium from the spent diesel exhaust catalyst by ammonia leaching method. J Hazard Mater 286:402–409. https://doi.org/10.1016/j.jhazmat.2014.12.063
Acknowledgements
The authors acknowledge with thanks the Staff of the Mineral Processing Laboratory of the University of Tehran for their technical support.
Author information
Authors and Affiliations
Contributions
Ehsan Ebrahimi: conceptualization, investigation, methodology, funding acquisition, software, formal analysis, validation, data curation, writing—original draft, writing—review and editing, visualization, and resources. Hassan Safari: investigation, methodology, validation, writing—review and editing, and funding acquisition. Mohammad Rezaee: investigation, methodology, validation, writing—review and editing, and funding acquisition. Ali Rezaei: resources and equipment. Hadi Abdollahi: conceptualization, methodology, supervision, project administration, funding acquisition, validation, and resources.
Corresponding author
Ethics declarations
Consent to participate
All the authors listed consent to participate.
Consent for publication
All the authors listed have approved the manuscript that is enclosed.
Conflict of interest
The authors declare no competing interests.
I would like to declare on behalf of my coauthors that the work described was original research that has not been published previously.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ebrahimi, E., Safari, H., Rezaee, M. et al. An environmentally friendly method for extraction of cobalt and molybdenum from spent catalysts using deep eutectic solvents (DESs). Environ Sci Pollut Res 30, 90243–90255 (2023). https://doi.org/10.1007/s11356-023-26806-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26806-8