Abstract
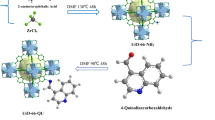
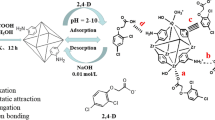
In this study, a green adsorbent (Fe3O4-UiO-66-NH2) with the ability of addressing the issues of separation and recovery of UiO-66-NH2 is obtained using a simple co-precipitation method under environmentally benign conditions. Various characterization techniques are utilized for evaluating the properties of the developed adsorbent. The capability of Fe3O4-UiO-66-NH2 towards 2,4-dichlorophenoxyacetic acid (2,4-D) and glyphosate (GP) from solution is explored. The results revealed that the magnetization process did not destroy the crystal structure of UiO-66-NH2, which ensured that Fe3O4-UiO-66-NH2 had good adsorption performance for 2,4-D and GP. The adsorption processes showed a wide pH application range, high salt tolerance, and regeneration performance as well as an excellent adsorption rate. Results from thermodynamic study showed that both processes were spontaneous and endothermic. The unit uptake ability of Fe3O4-UiO-66-NH2 for 2,4-D and GP reached up to 249 mg·g−1 and 183 mg·g−1 from Langmuir model at 303 K, respectively. When solid–liquid ratio was 2 g·L−1, Fe3O4-UiO-66-NH2 can reduce the content of 2,4-D or GP with the initial density of 100 mg·L−1 below the drinking water requirement limit. In addition, the reusability efficiency of Fe3O4-UiO-66-NH2 towards 2,4-D and GP was found to be 86% and 80% using 5 mmol·L−1 NaOH as eluent. Analysis of simulated water samples indicated that Fe3O4-UiO-66-NH2 could achieve the single or simultaneous removal of 2,4-D and GP from wastewater. Summarily, Fe3O4-UiO-66-NH2 as a green adsorbent can serve as an alternative for removing 2,4-D and GP from water body.








Similar content being viewed by others
Data availability
The dataset generated and analyzed during this study could be obtained from the corresponding author on reasonable request.
References
Aryee AA, Mpatani FM, Zhang XT, Kani AN, Dovi E, Han RP, Li ZH, Qu LB (2020) Iron (III) and iminodiacetic acid functionalized magnetic peanut husk for the removal of phosphate from solution: characterization, kinetic and equilibrium studies. J Clean Prod 268:122191. https://doi.org/10.1016/j.jclepro.2020.122191
Carles L, Martin-Laurent F, Devers M, Spor A, Rouard N, Beguet J, Besse-Hoggan P, Batisson I (2021) Potential of preventive bioremediation to reduce environmental contamination by pesticides in an agricultural context: a case study with the herbicide 2,4-D. J Hazard Mater 416:125740. https://doi.org/10.1016/j.jhazmat.2021.125740
Chen P, He XH, Pang MB, Dong XT, Zhao S, Zhang W (2020) Iodine capture using Zr- based metal–organic frameworks (Zr-MOFs): adsorption performance and mechanism. ACS Appl Mater Inter 12:20429–20439. https://doi.org/10.1021/acsami.0c02129
Chen SS, Zang ZY, Zhang SS, Ouyang GF, Han RP (2021) Preparation of MIL-100(Fe) and multi-walled carbon nanotubes nanocomposite with high adsorption capacity towards oxytetracycline from solution. J Environ Chem Eng 9(2):104780. https://doi.org/10.1016/j.jece.2020.104780
Diel JC, Franco DSP, Igansi AV, Cadaval TRS Jr, Pereira HeA, Nunes IdS, Basso CW, Alves MdCM, Morais J, Pinto D, Dotto GL (2021a) Green synthesis of carbon nanotubes impregnated with metallic nanoparticles: characterization and application in glyphosate adsorption. Chemosphere 283:131193. https://doi.org/10.1016/j.chemosphere.2021.131193
Diel JC, Franco DSP, Nunes IDS, Pereira HA, Moreira KS, Burgo TADL, Foletto EL, Dotto GL (2021b) Carbon nanotubes impregnated with metallic nanoparticles and their application as an adsorbent for the glyphosate removal in an aqueous matrix. J. Environ Chem Eng 9:105178. https://doi.org/10.1016/j.jece.2021.105178
Diel JC, Martinello KdB, da Silveira CL, Pereira HA, Franco DSP, Silva LFO, Dotto GL (2022) New insights into glyphosate adsorption on modified carbon nanotubes via green synthesis: statistical physical modeling and steric and energetic interpretations. Chem Eng J 431:134095. https://doi.org/10.1016/j.cej.2021.134095
Fajardo-Díaz JL, López-Urías F, Muñoz-Sandoval E (2020) Chloride functionalized carbon nanotube sponge: high charge capacity and high magnetic saturation. Carbon 164:324–336. https://doi.org/10.1016/j.carbon.2020.04.016
Fang F, Lv QK, Li P, Tao Y, Zhang Y, Zhou Y, Li XD, Li J (2022) Screening of hierarchical porous UiO-67 for efficient removal of glyphosate from aqueous solution. J Environ Chem Eng 10:107824. https://doi.org/10.1016/j.jece.2022.107824
Franco D, Silva LFO, Martinello KdB, Diel JC, Georgin J, Netto MS, Pereira HA, Lima EC, Dotto GL (2021) Transforming agricultural waste into adsorbent: application of Fagopyrum esculentum wheat husks treated with H2SO4 to adsorption of the 2,4-D herbicide. J Environ Chem Eng 9:106872. https://doi.org/10.1016/j.jece.2021.106872
Govarthanan M, Jeon CH, Kim W (2022) Synthesis and characterization of lanthanum-based metal organic framework decorated polyaniline for effective adsorption of lead ions from aqueous solutions. Environ Pollut 303:119049. https://doi.org/10.1016/j.envpol.2022.119049
Guo FY, Zhou M, Xu JC, Fein JB, Yu Q, Wang YW, Huang QY, Rong XM (2021) Glyphosate adsorption onto kaolinite and kaolinite-humic acid composites: experimental and molecular dynamics studies. Chemosphere 263:127979. https://doi.org/10.1016/j.chemosphere.2020.127979
Herrera-García U, Castillo J, Patiño-Ruiz D, Solano R, Herrera A (2019) Activated carbon from yam peels modified with Fe3O4 for removal of 2,4-dichlorophenoxyacetic acid in aqueous solution. Water 11:2342. https://doi.org/10.3390/w11112342
Li CS, Li YJ, Li Q, Duan JL, Hou JY, Hou Q, Ai SY, Li HS, Yang YC (2021) Regenerable magnetic aminated lignin/Fe3O4/La(OH)3 adsorbents for the effective removal of phosphate and glyphosate. Sci Total Environ 788:147812. https://doi.org/10.1016/j.scitotenv.2021.147812
Li KC, Zou S, Jin G, Yang J, Dou MY, Qin L, Su HF, Huang FC (2022) Efficient removal of selenite in aqueous solution by MOF-801 and Fe3O4/MOF-801: adsorptive behavior and mechanism study. Sep Purif Technol 296:121384. https://doi.org/10.1016/j.seppur.2022.121384
Li SM, Feng F, Chen S, Zhang XL, Liang YX, Shan SS (2020a) Preparation of UiO-66-NH2 and UiO-66-NH2/sponge for adsorption of 2,4-dichlorophenoxyacetic acid in water. Ecotox Environ Safe 194:110440. https://doi.org/10.1016/j.ecoenv.2020.110440
Li YS, Wang YT, He LY, Meng LZ, Lu HJ, Li XL (2020b) Preparation of poly(4-vinylpyridine)-functionalized magnetic Al-MOF for the removal of naproxen from aqueous solution. J Hazard Mater 383:121144. https://doi.org/10.1016/j.jhazmat.2019.121144
Lin KA, Liu YT, Chen SY (2016) Adsorption of fluoride to UiO-66-NH2 in water: stability, kinetic, isotherm and thermodynamic studies. J Colloid Interface Sci 461:79–87. https://doi.org/10.1016/j.jcis.2015.08.061
Liu HT, Guan JJ, Yang SX, Yu YH, Shao R, Zhang ZP, Dou ML, Wang F, Xu Q (2020a) Metal-organic-framework-derived Co2P nanoparticle/multi-doped porous carbon as a trifunctional electrocatalyst. Adv Mater 32:e2003649. https://doi.org/10.1002/adma.202003649
Liu MY, Zhang XT, Li ZH, Qu LB, Han RP (2020b) Fabrication of zirconium (IV)-loaded chitosan/Fe3O4/graphene oxide for efficient removal of alizarin red from aqueous solution. Carbohyd Polym 248:116792. https://doi.org/10.1016/j.carbpol.2020.116792
Liu W, Yang Q, Yang ZL, Wang WJ (2016) Adsorption of 2,4-D on magnetic graphene and mechanism study. Colloid Surface A 509:367–375. https://doi.org/10.1016/j.colsurfa.2016.09.039
Luo HP, Zhou X, Chen QY, Zhou J (2021a) Removal of 2,4-dichlorophenoxyacetic acid by the boron-nitrogen co-doped carbon nanotubes: insights into peroxymonosulfate adsorption and activation. Sep Purif Technol 259:118196. https://doi.org/10.1016/j.seppur.2020.118196
Luo XL, Huang GL, Chen XK, Guo J, Yang WX, Tang WZ, Yue TL, Li ZH (2021b) Ingenious ambient temperature fabrication zirconium-metal organic framework laden polysaccharide aerogel as an efficient glyphosate scavenger. J Environ Chem Eng 9:106808. https://doi.org/10.1016/j.jece.2021.106808
Magnoli K, Carranza CS, Aluffi ME, Magnoli CE, Barberis CL (2020) Herbicides based on 2,4-D: its behavior in agricultural environments and microbial biodegradation aspects. A review. Environ Sci Poll Res 27:38501–38512. https://doi.org/10.1007/s11356-020-10370-6
Mansoorianfar M, Nabipour H, Pahlevani F, Zhao YW, Hussain Z, Hojjati-Najafabadi A, Hoang HY, Pei RJ (2022) Recent progress on adsorption of cadmium ions from water systems using metal-organic frameworks (MOFs) as an efficient class of porous materials. Environ Res 214:114113. https://doi.org/10.1016/j.envres.2022.114113
Meftaul IM, Venkateswarlu K, Dharmarajan R, Annamalai P, Asaduzzaman M, Parven A, Megharaj M (2020) Controversies over human health and ecological impacts of glyphosate: Is it to be banned in modern agriculture? Environ Pollut 263:114372. https://doi.org/10.1016/j.envpol.2020.114372
Mpatani FM, Aryee AA, Han RP, Kani AN, Li ZH, Qu LB (2021) Green fabrication of a novel cetylpyridinium-bagasse adsorbent for sequestration of micropollutant 2,4-D herbicide in aqueous system and its antibacterial properties against S. aureus and E. coli. J Environ Chem Eng 9:106714. https://doi.org/10.1016/j.jece.2021.106714
Mpatani FM, Aryee AA, Kani AN, Guo QH, Dovi E, Qu LB, Li ZH, Han RP (2020) Uptake of micropollutant-bisphenol A, methylene blue and neutral red onto a novel bagasse-beta-cyclodextrin polymer by adsorption process. Chemosphere 259:127439. https://doi.org/10.1016/j.chemosphere.2020.127439
Neetha JN, Ujwal P, Kumar KG, Chidananda B, Goveas L, Sandesh K (2021) Aerobic biodegradation and optimization of 2,4-Dichlorophenoxyacetic acid by E. hormaechei subsp. Xiangfangensis and assessment of biodegraded metabolite toxicity. Environ Technol Inno 24:102055. https://doi.org/10.1016/j.eti.2021.102055
Orduz AE, Acebal C, Zanini G (2021) Activated carbon from peanut shells: 2,4-D desorption kinetics study for application as a green material for analytical purposes. J Environ Chem Eng 9:104601. https://doi.org/10.1016/j.jece.2020.104601
Paz MJ, Vieira T, Enzweiler H, Paulino AT (2022) Chitosan/wood sawdust/magnetite composite membranes for the photodegradation of agrochemicals in water. J Environ Chem Eng 10:106967. https://doi.org/10.1016/j.jece.2021.106967
Pereira HA, Hernandes PRT, Netto MS, Reske GD, Vieceli V, Oliveira LFS, Dotto GL (2020) Adsorbents for glyphosate removal in contaminated waters: a review. Environ Chem Lett 19:1525–1543. https://doi.org/10.1007/s10311-020-01108-4
Qiao Q, Zhou H, Guo F, Shu R, Liu S, Xu L, Dong K, Bai Y (2022) Facile and scalable synthesis of mesoporous composite materials from coal gasification fine slag for enhanced adsorption of malachite green. J Clean Prod 379:134739. https://doi.org/10.1016/j.jclepro.2022.134739
Ramrakhiani L, Ghosh S, Mandal AK, Majumdar S (2019) Utilization of multi-metal laden spent biosorbent for removal of glyphosate herbicide from aqueous solution and its mechanism elucidation. Chem Eng J 361:1063–1077. https://doi.org/10.1016/j.cej.2018.12.163
Sayğılı GA, Sayğılı H (2022) Fabrication of a magnetic hydrochar composite via an in situ one-pot hydrocarbonization strategy for efficient herbicide removal. Diam Relat Mater 128:109302. https://doi.org/10.1016/j.diamond.2022.109302
Sharma A, Mangla D, Shehnaz, Chaudhry SA (2022) Recent advances in magnetic composites as adsorbents for wastewater remediation. J Environ Manag 306:114483. https://doi.org/10.1016/j.jenvman.2022.114483
Singh S, Kumar V, Singh J (2019) Kinetic study of the biodegradation of glyphosate by indigenous soil bacterial isolates in presence of humic acid, Fe(III) and Cu(II) ions. J Environ Chem Eng 7:103098. https://doi.org/10.1016/j.jece.2019.103098
Soares SF, Amorim CO, Amaral JS, Trindade T, Daniel-da-Silva AL (2021) On the efficient removal, regeneration and reuse of quaternary chitosan magnetite nanosorbents for glyphosate herbicide in water. J Environ Chem Eng 9:105189. https://doi.org/10.1016/j.jece.2021.105189
Song YW, Wang YY, Han RP (2023) Adsorption of chlorophenols on activated pine sawdust pyrolytic carbon from solution in batch mode. Environ Sci Pollut Res 30:31294–31308. https://doi.org/10.1007/s11356-022-24403-9
Tchinsa A, Hossain MF, Wang T, Zhou Y (2021) Removal of organic pollutants from aqueous solution using metal organic frameworks (MOFs)-based adsorbents: a review. Chemosphere 284:131393. https://doi.org/10.1016/j.chemosphere.2021.131393
Uddin MJ, Ampiaw RE, Lee W (2021) Adsorptive removal of dyes from wastewater using a metal-organic framework: a review. Chemosphere 284:131314. https://doi.org/10.1016/j.chemosphere.2021.131314
Ueda Yamaguchi N, Bergamasco R, Hamoudi S (2016) Magnetic MnFe2O4–graphene hybrid composite for efficient removal of glyphosate from water. Chem Eng J 295:391–402. https://doi.org/10.1016/j.cej.2016.03.051
Vacca A, Mais L, Mascia M, Usai EM, Rodriguez J, Palmas S (2021) Mechanistic insights into 2,4-D photoelectrocatalytic removal from water with TiO2 nanotubes under dark and solar light irradiation. J Hazard Mater 412:125202. https://doi.org/10.1016/j.jhazmat.2021.125202
Valadi FM, Ekramipooya A, Gholami MR (2020) Selective separation of Congo Red from a mixture of anionic and cationic dyes using magnetic-MOF: experimental and DFT study. J Mol Liq 318:114051. https://doi.org/10.1016/j.molliq.2020.114051
Vinayagam R, Ganga S, Murugesan G, Rangasamy G, Bhole R, Goveas LC, Varadavenkatesan T, Dave N, Samanth A, Radhika Devi V, Selvaraj R (2023) 2,4-Dichlorophenoxyacetic acid (2,4-D) adsorptive removal by algal magnetic activated carbon nanocomposite. Chemosphere 310:136883. https://doi.org/10.1016/j.chemosphere.2022.136883
Vinayagam R, Pai S, Murugesan G, Varadavenkatesan T, Narayanasamy S, Selvaraj R (2022) Magnetic activated charcoal/Fe2O3 nanocomposite for the adsorptive removal of 2,4-dichlorophenoxyacetic acid (2,4-D) from aqueous solutions: Synthesis, characterization, optimization, kinetic and isotherm studies. Chemosphere 286:131938. https://doi.org/10.1016/j.chemosphere.2021.131938
Wang H, Wang S, Wang SX, Tang JL, Chen YB, Zhang LB (2022) Adenosine-functionalized UiO-66-NH2 to efficiently remove Pb(II) and Cr(VI) from aqueous solution: thermodynamics, kinetics and isothermal adsorption. J Hazard Mater 425:127771. https://doi.org/10.1016/j.jhazmat.2021.127771
Wang YF, Zhao W, Qi ZY, Zhang L, Zhang YN, Huang HO, Peng YZ (2020) Designing ZIF-8/hydroxylated MWCNT nanocomposites for phosphate adsorption from water: capability and mechanism. Chem Eng J 394:124992. https://doi.org/10.1016/j.cej.2020.124992
Wu H, Zhang H, Zhang W, Yang X, Zhou H, Pan Z, Wang D (2019) Preparation of magnetic polyimide@ Mg-Fe layered double hydroxides core-shell composite for effective removal of various organic contaminants from aqueous solution. Chemosphere 219:66–75. https://doi.org/10.1016/j.chemosphere.2018.11.209
Yang QF, Wang J, Chen XY, Yang WX, Pei HN, Hu N, Li ZH, Suo YR, Li T, Wang JL (2018) The simultaneous detection and removal of organophosphorus pesticides by a novel Zr-MOF based smart adsorbent. J Mater Chem A 6:2184–2192. https://doi.org/10.1039/c7ta08399h
Yu P, Li X, Zhang X, Zhou H, Xu YH, Sun YJ, Zheng HL (2021) Insights into the glyphosate removal efficiency by using magnetic powder activated carbon composite. Sep Purif Technol 254:117662. https://doi.org/10.1016/j.seppur.2020.117662
Zadeh RJ, Sayadi MH, Rezaei MR (2021) Synthesis of Thiol modified magMCM-41 nanoparticles with rice husk ash as a robust, high effective, and recycling magnetic sorbent for the removal of herbicides. J Environ Chem Eng 9:104804. https://doi.org/10.1016/j.jece.2020.104804
Zeng HJ, Yu ZX, Shao LY, Li XH, Zhu M, Liu YC, Feng XF, Zhu XM (2021) A novel strategy for enhancing the performance of membranes for dyes separation: Embedding PAA@UiO-66-NH2 between graphene oxide sheets. Chem Eng J 403:126281. https://doi.org/10.1016/j.cej.2020.126281
Zhang MM, Sun Q, Wang YJ, Shan WJ, Lou ZN, Xiong Y (2021a) Synthesis of porous UiO-66-NH2-based mixed matrix membranes with high stability, flux and separation selectivity for Ga(III). Chem Eng J 421:129748. https://doi.org/10.1016/j.cej.2021.129748
Zhang S, Wang JQ, Zhang Y, Ma JZ, Huang LTY, Yu SJ, Chen L, Song G, Qiu MQ, Wang XX (2021b) Applications of water-stable metal-organic frameworks in the removal of water pollutants: a review. Environ Pollut 291:118076. https://doi.org/10.1016/j.envpol.2021.118076
Zhang XT, Liu MY, Han RP (2021c) Adsorption of phosphate on UiO-66-NH2 prepared by a green synthesis method. J Environ Chem Eng 9:106672. https://doi.org/10.1016/j.jece.2021.106672
Zhang YJ, Cao H, Lu JR, Li YM, Bao MT (2022) Enhanced photocatalytic activity of glyphosate over a combination strategy of GQDs/TNAs heterojunction composites. J Colloid Interf Sci 607:607–620. https://doi.org/10.1016/j.jcis.2021.08.160
Zhang Z, Chen Y, Hu CY, Zuo C, Wang P, Chen WQ, Ao TQ (2021d) Efficient removal of tetracycline by a hierarchically porous ZIF-8 metal organic framework. Environ Res 198:111254. https://doi.org/10.1016/j.envres.2021.111254
Zhao FK, Fang SQ, Gao YX, Bi JH (2022) Removal of aqueous pharmaceuticals by magnetically functionalized Zr-MOFs: adsorption kinetics, isotherms, and regeneration. J Colloid Interf Sci 615:876–886. https://doi.org/10.1016/j.jcis.2022.02.018
Zhao YF, Wang DF, Wei W, Cui LZ, Cho CW, Wu GP (2021) Effective adsorption of mercury by Zr(IV)-based metal-organic frameworks of UiO-66-NH2 from aqueous solution. Environ Sci Pollut R 28:7068–7075. https://doi.org/10.1007/s11356-020-11080-9
Zhu XY, Tong JQ, Zhu LH, Pan DD (2022) In situ growth of ZIF-8 on carboxymethyl chitosan beads for improved adsorption of lead ion from aqueous solutions. Int J Biol Macromol 205:473–482. https://doi.org/10.1016/j.ijbiomac.2022.02.120
Zhuang ST, Cheng R, Wang JL (2019) Adsorption of diclofenac from aqueous solution using UiO-66-type metal-organic frameworks. Chem Eng J 359:354–362. https://doi.org/10.1016/j.cej.2018.11.150
Funding
The research was supported by the National Key Research and Development Program of China (2018YFD0401402–04) and Zhongyuan Scholars Foundation (202101510005). The authors would like to grateful acknowledge Dr. Aaron Albert Aryee for improving the scientific language of this manuscript.
Author information
Authors and Affiliations
Contributions
Xiaoting Zhang: conceptualization, preparation, methodology, data curation, writing-original draft, writing-review and editing. Yang Liu: software, investigation. Lingbo Qu: writing-review and editing. Runping Han: methodology, data curation, Funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Liu, Y., Qu, L. et al. Adsorption of 2,4-dichlorophenoxyacetic acid and glyphosate from water by Fe3O4-UiO-66-NH2 obtained in a simple green way. Environ Sci Pollut Res 30, 60574–60589 (2023). https://doi.org/10.1007/s11356-023-26737-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26737-4