Abstract
We investigated the effects of metals and physicochemical variables on the microbes and their metabolisms in the sediments of Guarapiranga reservoir, a tropical eutrophic-hypereutrophic freshwater reservoir located in a highly urbanized and industrialized area in Brazil. The metals cadmium, copper, and chromium showed minor contribution to changes in the structure, composition, and richness of sediment microbial communities and functions. However, the effects of metals on the microbiota are increased when taken together with physicochemical properties, including the sediment carbon and sulfur, the bottom water electrical conductivity, and the depth of the water column. Clearly, diverse anthropic activities, such as sewage discharge, copper sulfate application to control algal growth, water transfer, urbanization, and industrialization, contribute to increase these parameters and the metals spatially in the reservoir. Microbes found especially in metal-contaminated sites encompassed Bathyarchaeia, MBG-D and DHVEG-1, Halosiccatus, Candidatus Methanoperedens, Anaeromyxobacter, Sva0485, Thermodesulfovibrionia, Acidobacteria, and SJA-15, possibly showing metal resistance or acting in metal bioremediation. Knallgas bacteria, nitrate ammonification, sulfate respiration, and methanotrophy were inferred to occur in metal-contaminated sites and may also contribute to metal removal. This knowledge about the sediment microbiota and metabolisms in a freshwater reservoir impacted by anthropic activities allows new insights about their potential for metal bioremediation in these environments.
Graphical Abstract


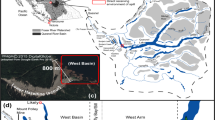
Source: Created with QGIS software v. 3.12.3 (QGIS Development Team 2020)






Similar content being viewed by others
Data availability
All the sequencing data analyzed in this study are available in the NCBI’s Sequence Read Archive, BioProjectID PRJNA690112, accession number SRP301026, experiments SRX9810583, SRX9810576, SRX9810554, SRX9810541, SRX9810538, SRX9810536, SRX9810533, and SRX9810531.
Materials availability
All the sequencing data analyzed in this study are available in the NCBI’s Sequence Read Archive, BioProjectID PRJNA690112, accession number SRP301026, experiments SRX9810583, SRX9810576, SRX9810554, SRX9810541, SRX9810538, SRX9810536, SRX9810533, and SRX9810531.
References
Ahmed AM, Lyautey E, Bonnineau C et al (2018) Environmental concentrations of copper, alone or in mixture with arsenic, can impact river sediment microbial community structure and functions. Front Microbiol 9:1852. https://doi.org/10.3389/fmicb.2018.01852
Ahmed AM, Tardy V, Bonnineau C et al (2020) Changes in sediment microbial diversity following chronic copper-exposure induce community copper-tolerance without increasing sensitivity to arsenic. J Hazard Mater 391:122197. https://doi.org/10.1016/j.jhazmat.2020.122197
Algül F, Beyhan M (2020) Concentrations and sources of heavy metals in shallow sediments in Lake Bafa. Turkey Sci Rep 10:11782. https://doi.org/10.1038/s41598-020-68833-2
Andersen JM (1976) An ignition method for determination of total phosphorus in lake sediments. Water Res 10:329–331. https://doi.org/10.1016/0043-1354(76)90175-5
APHA (2017) Standard methods for the examination of water and wastewater, 23rd edn. American Public Health Association, Washington, D. C., New York
Ayala D, Burgos W, España J et al (2020) Metagenomic and metatranscriptomic study of microbial metal resistance in an acidic pit lake. Microorganisms 8:1350. https://doi.org/10.3390/microorganisms8091350
Bar-Or I, Ben-Dov E, Kushmaro A et al (2015) Methane-related changes in prokaryotes along geochemical profiles in sediments of Lake Kinneret (Israel). Biogeosciences 12:2847–2860. https://doi.org/10.5194/bg-12-2847-2015
Branco R, Chung A-P, Veríssimo A, Morais PV (2005) Impact of chromium-contaminated wastewaters on the microbial community of a river. FEMS Microbiol Ecol 54:35–46. https://doi.org/10.1016/j.femsec.2005.02.014
Brasil (2012) CONAMA - Conselho Nacional do Meio Ambiente. Resolução n° 454, de 01 de novembro de 2012. Diário Oficial da União, Brasília, DF. 01 nov. 2012
Brito F, Miraglia S, Semensatto Junior D (2018) Ecosystem services of the Guarapiranga reservoir watershed (São Paulo, Brazil): value of water supply and implications for management strategies. International Journal of Urban Sustainable Development 10:1–11. https://doi.org/10.1080/19463138.2018.1442336
Cardoso-Silva S, Nishimura PY, Padial PR et al (2014) Compartimentalização e qualidade da água: o caso da Represa Billings. Bioikos 28:31–43
Cardoso-Silva S, López-Doval JC, Moschini-Carlos V, Pompêo M (2017) Metals and limnological variables in an urban reservoir: compartmentalization and identification of potential impacted areas. Environ Monit Assess 190:19. https://doi.org/10.1007/s10661-017-6387-3
Chardin B, Giudici-Orticoni M-T, De Luca G et al (2003) Hydrogenases in sulfate-reducing bacteria function as chromium reductase. Appl Microbiol Biotechnol 63:315–321. https://doi.org/10.1007/s00253-003-1390-8
Chen Y, Liu Y, Wang X (2017) Spatiotemporal variation of bacterial and archaeal communities in sediments of a drinking reservoir, Beijing, China. Appl Microbiol Biotechnol 101:3379–3391. https://doi.org/10.1007/s00253-016-8019-1
Chovanec P, Sparacino-Watkins C, Zhang N et al (2012) Microbial reduction of chromate in the presence of nitrate by three nitrate respiring organisms. Front Microbiol 3:416. https://doi.org/10.3389/fmicb.2012.00416
Costa PS, Reis MP, Ávila MP et al (2015) Metagenome of a microbial community inhabiting a metal-rich tropical stream sediment. PLOS ONE 10:e0119465. https://doi.org/10.1371/journal.pone.0119465
Cox MP, Peterson DA, Biggs PJ (2010) SolexaQA: at-a-glance quality assessment of illumina second-generation sequencing data. BMC Bioinformatics 11:485. https://doi.org/10.1186/1471-2105-11-485
de Andrade MRM, Salim A, Rossini-Penteado D et al (2016) Mapeamento de uso da terra para avaliação da qualidade das águas do reservatório Guarapiranga. Geociências (são Paulo) 34:258–274
Dinno A (2017) dunn.test: Dunn’s test of multiple comparisons using rank sums. Version 1.3.5. https://CRAN.R-project.org/package=dunn.test
Dong C-D, Chen C-W, Chen C-F (2013) Distribution and contamination status of chromium in surface sediments of northern Kaohsiung Harbor, Taiwan. J Environ Sci 25:1450–1457. https://doi.org/10.1016/S1001-0742(12)60200-9
Faheem M, Shabbir S, Zhao J et al (2020) Enhanced adsorptive bioremediation of heavy metals (Cd2+, Cr6+, Pb2+) by methane-oxidizing epipelon. Microorganisms 8:E505. https://doi.org/10.3390/microorganisms8040505
Ferreira K de S, Rani-Borges B, Santos GLM, et al (2021) Metais nos Sedimentos em Reservatórios: há Toxicidade Potencial? Sociedade & Natureza 33:. https://doi.org/10.14393/SN-v33-2021-58794
Gough HL, Stahl DA (2011) Microbial community structures in anoxic freshwater lake sediment along a metal contamination gradient. ISME J 5:543–558. https://doi.org/10.1038/ismej.2010.132
Gupta A, Saha A, Sar P (2021) Thermoplasmata and Nitrososphaeria as dominant archaeal members in acid mine drainage sediment of Malanjkhand Copper Project, India. Arch Microbiol 203:1833–1841. https://doi.org/10.1007/s00203-020-02130-4
Haas S, Desai DK, LaRoche J et al (2019) Geomicrobiology of the carbon, nitrogen and sulphur cycles in Powell Lake: a permanently stratified water column containing ancient seawater. Environ Microbiol 21:3927–3952. https://doi.org/10.1111/1462-2920.14743
Hao X, Zhu J, Rensing C et al (2021) Recent advances in exploring the heavy metal(loid) resistant microbiome. Comput Struct Biotechnol J 19:94–109. https://doi.org/10.1016/j.csbj.2020.12.006
He Q, Yao K (2010) Impact of alternative electron acceptors on selenium(IV) reduction by Anaeromyxobacter dehalogenans. Biores Technol. https://doi.org/10.1016/j.biortech.2010.10.046
He C, Zhang B, Jiang Y et al (2021) Microbial electrolysis cell produced biogas as sustainable electron donor for microbial chromate reduction. Chem Eng J 403:126429. https://doi.org/10.1016/j.cej.2020.126429
Hu Y, Chen N, Li M et al (2019) Insights into simultaneous microbial chromium and nitrate reduction: inhibitory effects and molecular mechanisms. J Chem Technol Biotechnol 94:2589–2596. https://doi.org/10.1002/jctb.6056
Johnson D, Hedrich S, Pakostova E (2017) Indirect redox transformations of iron, copper, and chromium catalyzed by extremely acidophilic bacteria. Frontiers in Microbiology 8:211. https://doi.org/10.3389/fmicb.2017.00211
Kapahi M, Sachdeva S (2019) Bioremediation options for heavy metal pollution. J Health Pollut 9:191203. https://doi.org/10.5696/2156-9614-9.24.191203
Karthikeyan OP, Smith TJ, Dandare SU et al (2021) Metal(loid) speciation and transformation by aerobic methanotrophs. Microbiome 9:156. https://doi.org/10.1186/s40168-021-01112-y
KC Denmark A/S (2021) 12.210 and 12.211 - Van Veen Grab – 1000 cm2 - Manual. https://www.kc-denmark.dk/media/92595/12.210%20-%20van%20veen%20grab%20-%201000%20cm%C2%B2%20-%20manual.pdf. Accessed 20 Jan 2023
Kristjansson JK, Schönheit P (1983) Why do sulfate-reducing bacteria outcompete methanogenic bacteria for substrates? Oecologia 60:264–266. https://doi.org/10.1007/BF00379530
Leal PR, Moschini-Carlos V, López-Doval JC et al (2018) Impact of copper sulfate application at an urban Brazilian reservoir: A geostatistical and ecotoxicological approach. Sci Total Environ 618:621–634. https://doi.org/10.1016/j.scitotenv.2017.07.095
Li C, Quan Q, Gan Y et al (2020) Effects of heavy metals on microbial communities in sediments and establishment of bioindicators based on microbial taxa and function for environmental monitoring and management. Sci Total Environ 749:141555. https://doi.org/10.1016/j.scitotenv.2020.141555
Liao W, Tong D, Li Z et al (2021) Characteristics of microbial community composition and its relationship with carbon, nitrogen and sulfur in sediments. Sci Total Environ 795:148848. https://doi.org/10.1016/j.scitotenv.2021.148848
Lin C-Y, Shei S-H (2008) Heavy metal effects on fermentative hydrogen production using natural mixed microflora. Int J Hydrogen Energy 33:587–593. https://doi.org/10.1016/j.ijhydene.2007.09.030
Liu L, Yang J, Yu X et al (2013) Patterns in the composition of microbial communities from a subtropical river: effects of environmental, spatial and temporal factors. PLOS ONE 8:e81232. https://doi.org/10.1371/journal.pone.0081232
Long Y, Jiang J, Hu X et al (2021) The response of microbial community structure and sediment properties to anthropogenic activities in Caohai wetland sediments. Ecotoxicol Environ Saf 211:111936. https://doi.org/10.1016/j.ecoenv.2021.111936
Louca S, Parfrey LW, Doebeli M (2016) Decoupling function and taxonomy in the global ocean microbiome. Science 353:1272–1277. https://doi.org/10.1126/science.aaf4507
Luo J-H, Wu M, Liu J et al (2019) Microbial chromate reduction coupled with anaerobic oxidation of methane in a membrane biofilm reactor. Environ Int 130:104926. https://doi.org/10.1016/j.envint.2019.104926
Luo X, Xiang X, Huang G et al (2019b) Bacterial abundance and physicochemical characteristics of water and sediment associated with hydroelectric dam on the Lancang River China. Int J Environ Res Public Health 16:2031. https://doi.org/10.3390/ijerph16112031
Lv J, Niu Y, Yuan R, Wang S (2021) Different responses of bacterial and archaeal communities in river sediments to water diversion and seasonal changes. Microorganisms 9:782. https://doi.org/10.3390/microorganisms9040782
Ma Y, Liu F, Kong Z et al (2016) The distribution pattern of sediment archaea community of the Poyang Lake, the largest freshwater lake in China. Archaea 2016:e9278929. https://doi.org/10.1155/2016/9278929
Mahé F, Rognes T, Quince C et al (2014) Swarm: robust and fast clustering method for amplicon-based studies. PeerJ 2:e593. https://doi.org/10.7717/peerj.593
Masella AP, Bartram AK, Truszkowski JM et al (2012) PANDAseq: paired-end assembler for illumina sequences. BMC Bioinformatics 13:31. https://doi.org/10.1186/1471-2105-13-31
McCartney DM, Oleszkiewicz JA (1993) Competition between methanogens and sulfate reducers: effect of COD:sulfate ratio and acclimation. Water Environ Res 65:655–664
Meng S, Peng T, Pratush A et al (2021) Interactions between heavy metals and bacteria in mangroves. Mar Pollut Bull 172:112846. https://doi.org/10.1016/j.marpolbul.2021.112846
Mysara M, Saeys Y, Leys N et al (2015) CATCh, an ensemble classifier for chimera detection in 16S rRNA sequencing studies. Appl Environ Microbiol 81:1573–1584. https://doi.org/10.1128/AEM.02896-14
Ni C, Horton DJ, Rui J et al (2016) High concentrations of bioavailable heavy metals impact freshwater sediment microbial communities. Annals of Microbiology 66:1003–1012
Odubanjo GO, Oyetibo GO, Ilori MO (2021) Ecological risks of heavy metals and microbiome taxonomic profile of a freshwater stream receiving wastewater of textile industry. Front Environ Sci 9:130. https://doi.org/10.3389/fenvs.2021.554490
Oksanen J, Blanchet FG, Friendly M et al (2019) vegan: community ecology package. Version 2.5–6. https://CRAN.R-project.org/package=vegan
Pei Y, Yu Z, Ji J et al (2018) Microbial community structure and function indicate the severity of chromium contamination of the Yellow river. Front Microbiol 9:38. https://doi.org/10.3389/fmicb.2018.00038
Pierangeli GMF, Domingues MR, de Jesus TA et al (2021) Higher abundance of sediment methanogens and methanotrophs do not predict the atmospheric methane and carbon dioxide flows in eutrophic tropical freshwater reservoirs. Front Microbiol 12:647921. https://doi.org/10.3389/fmicb.2021.647921
Pierangeli GMF, Domingues MR, Choueri RB et al (2022) Spatial variation and environmental parameters affecting the abundant and rare communities of bacteria and archaea in the sediments of tropical urban reservoirs. Microb Ecol. https://doi.org/10.1007/s00248-022-02047-z
Pompêo M, Padial PR, Mariani CF et al (2013) Biodisponibilidade de metais no sedimento de um reservatório tropical urbano (reservatório Guarapiranga – São Paulo (SP), Brasil): há toxicidade potencial e heterogeneidade espacial? Geochimica Brasiliensis 27:104–104
QGIS Development Team (2020) QGIS: geographic information system. Version 3.12.3. http://qgis.osgeo.org
Quast C, Pruesse E, Yilmaz P et al (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:D590–D596. https://doi.org/10.1093/nar/gks1219
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria
Rader KJ, Carbonaro RF, van Hullebusch ED et al (2019) The fate of copper added to surface water: field, laboratory, and modeling studies. Environ Toxicol Chem 38:1386–1399. https://doi.org/10.1002/etc.4440
Rajeev M, Sushmitha TJ, Aravindraja C et al (2021) Exploring the impacts of heavy metals on spatial variations of sediment-associated bacterial communities. Ecotoxicol Environ Saf 209:111808. https://doi.org/10.1016/j.ecoenv.2020.111808
Salam LB, Obayori OS, Ilori MO, Amund OO (2020) Effects of cadmium perturbation on the microbial community structure and heavy metal resistome of a tropical agricultural soil. Bioresources and Bioprocessing 7:25. https://doi.org/10.1186/s40643-020-00314-w
Schloss PD, Westcott SL, Ryabin T et al (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541. https://doi.org/10.1128/AEM.01541-09
Shen C, Zhao J, Xie G et al (2021) Identifying microbial distribution drivers of archaeal community in sediments from a black-odorous urban river — a case study of the Zhang River Basin. Water 13:1545. https://doi.org/10.3390/w13111545
Sobolev D, Begonia MFT (2008) Effects of heavy metal contamination upon soil microbes: lead-induced changes in general and denitrifying microbial communities as evidenced by molecular markers. Int J Environ Res Public Health 5:450–456
Sutcliffe B, Hose G, Harford A et al (2019) Microbial communities are sensitive indicators for freshwater sediment copper contamination. Environ Pollut. https://doi.org/10.1016/j.envpol.2019.01.104
Tan S, Liu J, Fang Y et al (2019) Insights into ecological role of a new deltaproteobacterial order Candidatus Acidulodesulfobacterales by metagenomics and metatranscriptomics. ISME J 13:2044–2057. https://doi.org/10.1038/s41396-019-0415-y
Tao K, Liu Y, Ke T et al (2019) Patterns of bacterial and archaeal communities in sediments in response to dam construction and sewage discharge in Lhasa River. Ecotoxicol Environ Saf 178:195–201. https://doi.org/10.1016/j.ecoenv.2019.03.107
Tarekegn MM, Salilih FZ, Ishetu AI (2020) Microbes used as a tool for bioremediation of heavy metal from the environment. Cogent Food & Agriculture 6:1783174. https://doi.org/10.1080/23311932.2020.1783174
Thevenon F, Graham ND, Chiaradia M et al (2011) Local to regional scale industrial heavy metal pollution recorded in sediments of large freshwater lakes in central Europe (lakes Geneva and Lucerne) over the last centuries. Sci Total Environ 412–413:239–247. https://doi.org/10.1016/j.scitotenv.2011.09.025
Voica DM, Bartha L, Banciu HL, Oren A (2016) Heavy metal resistance in halophilic bacteria and archaea. FEMS Microbiology Letters 363:. https://doi.org/10.1093/femsle/fnw146
Wang S, Zhang B, Diao M et al (2018) Enhancement of synchronous bio-reductions of vanadium (V) and chromium (VI) by mixed anaerobic culture. Environ Pollut 242:249–256. https://doi.org/10.1016/j.envpol.2018.06.080
Wang L, Li Y, Zhao Z et al (2021a) Microbial community diversity and composition in river sediments contaminated with tetrabromobisphenol A and copper. Chemosphere 272:129855. https://doi.org/10.1016/j.chemosphere.2021.129855
Wang M, Wang L, Shi H et al (2021b) Soil bacteria, genes, and metabolites stimulated during sulfur cycling and cadmium mobilization under sodium sulfate stress. Environ Res 201:111599. https://doi.org/10.1016/j.envres.2021.111599
Weber P, Behr ER, Knorr CDL et al (2013) Metals in the water, sediment, and tissues of two fish species from different trophic levels in a subtropical Brazilian river. Microchem J 106:61–66. https://doi.org/10.1016/j.microc.2012.05.004
Wu Q, Sanford RA, Löffler FE (2006) Uranium(VI) reduction by Anaeromyxobacter dehalogenans strain 2CP-C. Appl Environ Microbiol 72:3608–3614. https://doi.org/10.1128/AEM.72.5.3608-3614.2006
Wu B, Wang G, Wu J et al (2014) Sources of heavy metals in surface sediments and an ecological risk assessment from two adjacent plateau reservoirs. PLOS ONE 9:e102101. https://doi.org/10.1371/journal.pone.0102101
Xia L, Yin C, Cai L et al (2010) Metabolic changes of Acidithiobacillus caldus under Cu2+ stress. J Basic Microbiol 50:591–598. https://doi.org/10.1002/jobm.201000051
Yu C, Zhang J, Wu L et al (2015) Effects of heavy metal and nutrients on benthic microbial communities in freshwater sediment of Poyang Lake (China). Journal of Residuals Science and Technology 12:105–111. https://doi.org/10.12783/issn.1544-8053/12/2/11
Zhang L, Zhao T, Wang Q et al (2019) Bacterial community composition in aquatic and sediment samples with spatiotemporal dynamics in large, shallow, eutrophic Lake Chaohu, China. J Freshw Ecol 34:575–589. https://doi.org/10.1080/02705060.2019.1635536
Zhang L, Tu D, Li X et al (2020) Impact of long-term industrial contamination on the bacterial communities in urban river sediments. BMC Microbiol 20:254. https://doi.org/10.1186/s12866-020-01937-x
Zhang X, Gai X, Zhong Z et al (2021) Understanding variations in soil properties and microbial communities in bamboo plantation soils along a chromium pollution gradient. Ecotoxicol Environ Saf 222:112507. https://doi.org/10.1016/j.ecoenv.2021.112507
Zhao Q, Li X, Wang Y et al (2023) Long-term bioremediation of cadmium contaminated sediment using sulfate reducing bacteria: perspective on different depths of the sediment profile. Chem Eng J 451:138697. https://doi.org/10.1016/j.cej.2022.138697
Acknowledgements
We thank the São Paulo Research Foundation (FAPESP), the Brazilian National Research Council (CNPq), the Coordination of Superior Level Staff Improvement (CAPES) and the Multiuser Experimental Center from Federal University of ABC.
Funding
This work was supported by the São Paulo Research Foundation (FAPESP—process number: 2017/10355–1), with technical training scholarship (process numbers: 2017/19001–8; 2018/20417–7; 2019/23767–1) and student’s scholarship (process number: 2018/19694–6) and by the Brazilian National Research Council (CNPq), with master’s scholarship (process number: 167185/2018–7).
Author information
Authors and Affiliations
Contributions
The authors Gabrielle Maria Fonseca Pierangeli, Karine Mirelle Rodrigues da Silva, Lucia Helena Gomes Coelho, and Gustavo Bueno Gregoracci contributed to the study conception and design. Methodology: Gabrielle Maria Fonseca Pierangeli, Karine Mirelle Rodrigues da Silva, Lucia Helena Gomes Coelho, and Gustavo Bueno Gregoracci. Formal analysis and investigation: Gabrielle Maria Fonseca Pierangeli and Karine Mirelle Rodrigues da Silva. Writing—original draft preparation: Gabrielle Maria Fonseca Pierangeli, Lucia Helena Gomes Coelho, Roseli Frederigi Benassi, Mércia Regina Domingues, and Gustavo Bueno Gregoracci; Writing, review, and editing: Lucia Helena Gomes Coelho, Roseli Frederigi Benassi, Mércia Regina Domingues, and Gustavo Bueno Gregoracci; funding acquisition and resources: Roseli Frederigi Benassi; supervision: Lucia Helena Gomes Coelho, Roseli Frederigi Benassi, and Gustavo Bueno Gregoracci. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Robert Duran
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pierangeli, G.M.F., da Silva, K.M.R., Coelho, L.H.G. et al. Effects of metal contamination with physicochemical properties on the sediment microbial communities in a tropical eutrophic-hypereutrophic urban reservoir in Brazil. Environ Sci Pollut Res 30, 54961–54978 (2023). https://doi.org/10.1007/s11356-023-26114-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26114-1