Abstract
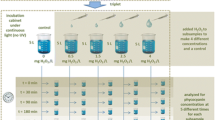
Harmful cyanobacterial blooms plague reservoirs and lakes used for a variety of purposes, such as recreation and drinking water. Chemical controls are frequently used to mitigate the occurrence of cyanobacterial blooms given that many are fast-acting and effective at reducing cyanobacterial abundance. Recent research has identified hydrogen peroxide (H2O2) as an environmentally friendly alternative to algaecides that have typically been used, such as copper sulfate. To build on past studies, these experiments sought to further understand how well H2O2 treatments reduce cyanobacteria in complex eutrophic conditions, as well as to assess treatment effects on a non-target phytoplankter, a green alga. We assessed the effectiveness of H2O2 (at treatments of 2–16 mg L−1) under varying environmental conditions in a controlled laboratory setting, including (1) dissolved organic matter (DOM) concentrations (humic acid; 0–60 mg L−1), (2) temperature (20, 25, and 32 °C), and (3) initial algal biomass (chlorophyll-a; 82–371 µg L−1). In contrast to our expectations, neither DOM concentration nor temperature meaningfully impacted the effectiveness of H2O2 at reducing cyanobacteria. However, initial algal biomass as well as H2O2 treatment dose greatly influenced the effectiveness of the algaecide on cyanobacteria. Treatments of ≥ 8 mg H2O2 L−1 on algal biomass were significantly buffered with higher DOM and lower temperature, and the biological significance of these findings should be explored further. Across all experiments, H2O2 concentrations of 0.03–0.12 mg H2O2 L−1 µg chlorophyll L−1 were effective at significantly reducing cyanobacteria with varying effects on algal biomass. Thus, water resource managers are encouraged to consider how ambient levels of phytoplankton biomass may affect the ability of H2O2 to control cyanobacterial blooms prior to treatment.






Similar content being viewed by others
Data availability
Data and code are available from the corresponding author upon reasonable request.
References
Barrington DJ, Ghadouani A (2008) Application of hydrogen peroxide for the removal of toxic cyanobacteria and other phytoplankton from wastewater. Environ Sci Technol 42:8916–8921. https://doi.org/10.1021/es801717y
Barrington DJ, Ghadouani A, Ivey GN (2011) Environmental factors and the application of hydrogen peroxide for the removal of toxic cyanobacteria from waste stabilization ponds. J Environ Eng 137:952–960. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000401
Barrington DJ, Ghadouani A, Ivey GN (2013) Cyanobacterial and microcystins dynamics following the application of hydrogen peroxide to waste stabilization ponds. Hydrol Earth Syst Sci 17:2097–2105. https://doi.org/10.5194/hess-17-2097-2013
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Beaulieu M, Pick F, Gregory-Eaves I (2013) Nutrients and water temperature are significant predictors of cyanobacterial biomass in a 1147 lakes data set. Limnol Oceanogr 58(5):1736–1746. https://doi.org/10.4319/lo.2013.58.5.1736
Boyd CE, Shelton JL (1984) Observations on the hydrology and morphometry of ponds on the Auburn University Fisheries Research Unit. Bulletin 558:1–64
Boyd CE (2015) Water Quality: An Introduction, 2nd (ed) Springer International Publishing https://doi.org/10.1007/978-3-319-17446-4
Buley RP, Adams C, Belfiore AP, Fernandez-Figueroa EG, Gladfelter MF, Garner B, Straus DL, Wilson AE (2021) Field evaluation of seven products to control cyanobacterial blooms in aquaculture. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-021-12708-0
Chen Z, Li J, Chen M, Yuen Koh K, Du Z, Yew-Hoong Gin K, He Y, Ong CN, Chen JP (2021) Microcystis aeruginosa removal by peroxides of hydrogen peroxide, peroxymonosulfate and peroxydisulfate without additional activators. Water Res 201 https://doi.org/10.1016/j.watres.2021.117263
Crafton EA, Cutright TJ, Bishop WM, Ott DW (2019) Modulating the effect of iron and total organic carbon on the efficiency of a hydrogen peroxide-based algaecide for suppressing cyanobacteria. Water Air Soil Pollution 230:56. https://doi.org/10.1007/s11270-019-4112-2
Doskočil L, Grasset L, Válková D, Pekař M (2014) Hydrogen peroxide oxidation of humic acids and lignite. Fuel 134:406–413. https://doi.org/10.1016/j.fuel.2014.06.011
Drábková M, Admiraal W, Maršálek B (2007) Combined exposure to hydrogen peroxide and light selective effects on cyanobacteria, green algae, and diatoms. Environ Sci Technol 41(1):309–314. https://doi.org/10.1021/es060746i
Florida Department of Environmental Protection (FDEP) (2013) Implementation of Florida’s numeric nutrient standards. Accessed 3/2/2021. https://floridadep.gov/sites/default/files/NNC_Implementation.pdf
Francy DS, Brady AMG, Ecker CD, Graham JL, Stelzer EA, Struffolino P, Dwyer DF, Loftin KA (2016) Estimating microcystin levels at recreational sites in western Lake Erie and Ohio. Harmful Algae 58:23–34. https://doi.org/10.1016/j.hal.2016.07.003
Gao L, Pan X, Zhang D, Mu S, Lee DJ, Halik U (2015) Extracellular polymeric substances buffer against the biocidal effect of H2O2 on the bloom-forming cyanobacterium Microcystis aeruginosa. Water Res 69:51–58. https://doi.org/10.1016/j.watres.2014.10.060
Goes MCA, et al (2021) Dynamics of metabolites production and application of hydrogen peroxide for the removal of noxious cyanobacteria from isolated cultures and wastewater. Master’s Thesis. Universidade Federal Do Ceara. pp105. https://repositorio.ufc.br/handle/riufc/58089?locale=es
Greenfield DI, Duquette A, Goodson A, Keppler CJ, Williams SH, Brock LM, Stackley KD, White D, Wilde SB (2014) The effects of three chemical algaecides on cell numbers and toxin content of the cyanobacteria Microcystis aeruginosa and Anabaenopsis sp. Environ Manage 54:1110–1120. https://doi.org/10.1007/s00267-014-0339-2
Gross A, Boyd C (1998) A digestion procedure for the simultaneous determination of total nitrogen and total phosphorus in pond water. J World Aquac Soc-Wiley Online Lib 29:300–303. https://doi.org/10.1111/j.1749-7345.1998.tb00650.x
Jiang Y, Fang Y, Liu Y, Liu B, Zhang J (2022) Community succession during the preventive control of cyanobacterial bloom by hydrogen peroxide in an aquatic microcosm. Ecotoxicol Environ Saf 237:113546. https://doi.org/10.1016/j.ecoenv.2022.113546
Kasinak JME, Holt BM, Chislock MF, Wilson AE (2015) Benchtop fluorometry of phycocyanin as a rapid approach for estimating cyanobacterial biovolume. J Plankton Res 37:248–257. https://doi.org/10.1093/plankt/fbu096
Kehoe M, Chun K, Baulch H (2015) Who smells? Forecasting taste and odor in a drinking water reservoir. Environ Sci Technol 49:10984–10992. https://doi.org/10.1021/acs.est.5b00979
Kinley-Baird C, Calomeni A, Berthold DE, Lefler FW, Barbosa M, Rodgers JH, Laughinghouse HD (2021) Laboratory-scale evaluation of algaecide effectiveness for control of microcystin-producing cyanobacteria from Lake Okeechobee, Florida (USA). Ecotoxicol Environ Saf 207:111233. https://doi.org/10.1016/j.ecoenv.2020.111233
Lenth RV, Buerkner P, Herve M, Love J, Riebl H, Singmann H (2021) Estimated marginal means, aka least-squares means. https://cran.r-project.org/web/packages/emmeans/emmeans.pdf
Lürling M, Meng D, Faassen EJ (2014) Effects of hydrogen peroxide and ultrasound on biomass reduction and toxin release in the cyanobacterium, Microcystis aeruginosa. Toxins 6:3260–3280. https://doi.org/10.3390/toxins6123260
Lusty MW, Gobler CJ (2023) Repeated hydrogen peroxide dosing briefly reduces cyanobacterial blooms and microcystin while increasing fecal bacteria indicators in a eutrophic pond. J Environ Sci (china) 124:522–543. https://doi.org/10.1016/j.jes.2021.11.031
Matthijs HCP, Visser PM, Reeze B, Meeuse J, Slot PC, Wijn G, Talens R, Huisman J (2012) Selective suppression of harmful cyanobacteria in an entire lake with hydrogen peroxide. Water Res, Cyanobacteria: Impacts of Climate Change on Occurrence, Toxicity and Water Quality Management 46:1460–1472. https://doi.org/10.1016/j.watres.2011.11.016
Morris JE, Mischke CC (1999) Phytoplankton management for fish culture. Iowa State University. NCRAC Technical bulletin. 12–1999. https://www.ncrac.org/files/biblio/TB114.pdf
Paerl HW, Huisman J (2008) Blooms like it hot. Science 320:57–58. https://doi.org/10.1126/science.1155398
Paerl HW, Bebout BM, Joye SB, Marais DJD (1993) Microscale characterization of dissolved organic matter production and uptake in marine microbial mat communities. Limnol Oceanogr 38:1150–1161. https://doi.org/10.4319/lo.1993.38.6.1150
Paerl HW, Fulton RS, Moisander PH, Dyble J (2001) Harmful freshwater algal blooms, with an emphasis on cyanobacteria. Sci World J 1:76–113. https://doi.org/10.1100/tsw.2001.16
Piel T, Sandrini G, Muyzer G, Brussaard CPD, Slot PC, van Herk MJ, Huisman J, Visser PM (2021) Resilience of microbial communities after hydrogen peroxide treatment of a eutrophic lake to suppress harmful cyanobacterial blooms. Microorganisms 7:1495. https://doi.org/10.3390/microorganisms9071495
Pinheiro J, DebRoy B, Sarkar D (2021) Linear and nonlinear mixed effects models. https://cran.r-project.org/web/packages/nlme/citation.html. Accessed 16 April 2021
Reitsema RE, Meire P, Schoelynck J (2018) The future of freshwater macrophytes in a changing world: dissolved organic carbon quantity and quality and its interactions with macrophytes. Front Plant Sci 9 https://doi.org/10.3389/fpls.2018.00629
Rippka R, Deruelles J, Waterbury JB, Herdmam M, Stanier RYY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 111:1–61. https://doi.org/10.1099/00221287-111-1-1
Romero-Martínez L, Rivas-Zaballos I, Moreno-Andrés J, Moreno-Garrido I, Acevedo-Merino A, Nebot E (2021) Improving the microalgae inactivating efficacy of ultraviolet ballast water treatment in combination with hydrogen peroxide or peroxymonosulfate salt. Mar Pollut Bull 162:111886. https://doi.org/10.1016/j.marpolbul.2020.111886
Rouse DR, Boyd C, Lichtkoppler F (1979) Water quality management and pond fish culture. Research and Development Series number 22. International Center for Aquaculture. Agricultural Experiment Station, Auburn University. https://aurora.auburn.edu/bitstream/handle/11200/1088/0192FISH.pdf?sequence=1
Santos A, Guedes DO, Barros MUG, Oliveira S, Pacheco ABF, Azevedo S, Magalhães VF, Pestana CJ, Edwards C, Lawton LA, Capelo-Neto J (2021) Effect of hydrogen peroxide on natural phytoplankton and bacterioplankton in a drinking water reservoir: Mesocosm-scale study. Water Res 197. https://doi.org/10.1016/j.watres.2021.117069.
Sarnelle O (2007) Initial conditions mediate the interaction between Daphnia and bloom-forming cyanobacteria. Limnol Oceanogr 52:2120–2127. https://doi.org/10.4319/lo.2007.52.5.2120
Sartory DP, Grobbelaar JU (1984) Extraction of chlorophyll a from Ffeshwater phytoplankton for spectrophotometric analysis. Hydrobiol 114:177–187. https://doi.org/10.1007/BF00031869
Schrader KK, Tucker CS, Brown TW, Torrans EL, Whitis GN (2016) Comparison of phytoplankton communities in catfish split-pond aquaculture systems with conventional ponds. N Am J Aquac 78:384–395. https://doi.org/10.1080/15222055.2016.1201557
Sinha AK, Eggleton MA, Lochmann RT (2018) An environmentally friendly approach for mitigating cyanobacterial bloom and their toxins in hypereutrophic ponds: potentiality of a newly developed granular hydrogen peroxide-based compound. Sci Total Environ 637–638:524–537. https://doi.org/10.1016/j.scitotenv.2018.05.023
US Environmental Protection Agency (EPA) (2016) Human health recreational ambient water quality criteria or swimming advisories for microcystins and cylindrospermopsin. Office of Water. Document number: 822-P-16–002. 1–185. https://www.epa.gov/sites/default/files/2016-12/documents/draft-hh-rec-ambient-water-swimming-document.pdf. Accessed 3/2/2021
Wang Z, Li D, Qin H, Li Y (2012) An integrated method for removal of harmful cyanobacterial blooms in eutrophic lakes. Environ Pollut 160:34–41. https://doi.org/10.1016/j.envpol.2011.09.003
Weenink EFJ, Matthijs HCP, Schuurmans JM, Piel T, van Herk MJ, Sigon CAM, Visser PM (2021) Huisman (2021) Interspecific protection against oxidative stress: green algae protect harmful cyanobacteria against hydrogen peroxide. Environ Microbiol 23(5):2404–2419. https://doi.org/10.1111/1462-2920.15429
Wilson AE, Chislock MF, Yang Z, Barros MUG, Roberts JF (2018) Pond bank access as an approach for managing toxic cyanobacteria in beef cattle pasture drinking water ponds. Environ Monit Assess 190:247. https://doi.org/10.1007/s10661-018-6595-5
Wisconsin Department of Natural Resources (WDNR) (2019) Water quality standards rule packages technical support document: waterbody assessments, bicriteria, and phosphorus response indicators (Rule No. WY-23–13). V1. 6–17–2019. Accessed 3/2/2021. https://dnr.wi.gov/news/input/documents/rules/WY2313TechnicalSupportDocument.pdf
Yang Z, Buley RP, Fernandez-Figueroa EG, Barros MUG, Rajendran S, Wilson AE (2018) Hydrogen peroxide treatment promotes chlorophytes over toxic cyanobacteria in a hyper-eutrophic aquaculture pond. Environ Pollut 240:590–598. https://doi.org/10.1016/j.envpol.2018.05.012
Yang X, Rosario-Ortiz FL, Lei Y, Pan Y, Lei X, Westerhoff P (2022) Multiple roles of dissolved organic matter in advanced oxidation processes. Environ Sci Technol 56:16. https://doi.org/10.1021/acs.est.2c01017
Yazıcı E, Deveci H (2010) Factors affecting decomposition of hydrogen peroxide. Proceedings of the XIIth International Mineral Processing Symposium, 609–616. https://doi.org/10.13140/RG.2.1.1530.0648
Yu D, Qiu W, Zhang Z, Glass K, Su J, DeMeo DL, Tantisira K, Weiss ST (1982) corTest: robust tests for equal correlation. Educ Psychol Meas 42(4):987–100. https://doi.org/10.1177/001316448204200407
Zepp R, Faust B, Hoigne J (1992) Hydroxyl radical formation in aqueous reactions (pH 3–8) of iron(II) with hydrogen peroxide: the photo-Fenton reaction. Environ Sci Technol 26:313–319. https://doi.org/10.1021/es00026a011
Znachor P, Nedoma J (2010) Importance of dissolved organic carbon for phytoplankton nutrition in a eutrophic reservoir. J Plankton Res 32:367–376. https://doi.org/10.1093/plankt/fbp129
Funding
This study was supported by the National Science Foundation [grant DBI-1658694], U.S. Department of Agriculture [2017–70007-27132 and 58–6010-0–006], and the Alabama Agricultural Experiment Station and the Hatch program of the National Institute of Food and Agriculture [ALA016-1–16007].
Author information
Authors and Affiliations
Contributions
Author contributed the following aspects of the manuscript:
Riley Buley: Conceptualization, Methodology, Data curation, Writing original and later edits, Visualization.
Alan Wilson: Conceptualization, Methodology, Edits to original manuscript, Supervision, Funding acquisition.
Matthew Gladfelter: Formal analysis, Investigation, Edits to original manuscript.
Edna Fernandez-Figueroa: Methodology; Investigation, Edits to original manuscript.
Corresponding author
Ethics declarations
Ethics approval
No human participants were used in this study.
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Sami Rtimi
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. The effect of hydrogen peroxide on cyanobacteria was assessed under varying conditions.
2. Neither dissolved organic matter concentration nor temperature meaningfully reduced the efficacy of hydrogen peroxide.
3. Initial algal biomass negatively influenced the efficacy of hydrogen peroxide.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Buley, R.P., Gladfelter, M.F., Fernandez-Figueroa, E.G. et al. Complex effects of dissolved organic matter, temperature, and initial bloom density on the efficacy of hydrogen peroxide to control cyanobacteria. Environ Sci Pollut Res 30, 43991–44005 (2023). https://doi.org/10.1007/s11356-023-25301-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-25301-4