Abstract
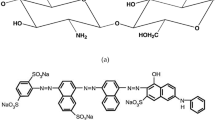
Biosorption is a key phenomenon that has been used to remove the aquatic pollutants like dyes and heavy metals present in industrial effluents. The current study aims at the development of a chitosan-pectinase blend (CPB) to separate the methylene blue (MB) dye from its synthetic solution. Pectinase, an enzyme isolated from a consortium of Bacillus species, is imbibed in the blend. The electron micrographs revealed the rough surface of the adsorbent, and its amorphous nature was evident from broader peaks in diffraction patterns. The FTIR analyses indicated the perfect blend formation through the presence and shifts in the characteristic peaks. Maximum adsorption was exhibited at pH 7.0, 30 °C, 30 min of contact time and an adsorbent dosage of 2.5 g/L. On comparison, the pseudo-second-order model was found to be the suitable fit with the highest R2 value closer to 1. Different isotherm models were experimentally fitted and the maximum adsorption capacity was obtained at 16.81 mg/g and the Temkin isotherm suits the best. The polymer blend showed an agreeable extent of desorption of MB dye which was evident from the desorption studies and, thereby, the biosorbent could be reused for removing the dye to the maximum extent.












Similar content being viewed by others
Data availability
Not applicable.
References
Abdeen Z, Mohammad SG, Mahmoud MS (2015) Adsorption of Mn (II) ion on polyvinyl alcohol/chitosan dry blending from aqueous solution. Environ Nanotechnol Monit Manag 3:1–9
Ahmad M, Ahmed S, Swami BL, Ikram S (2015) Adsorption of heavy metal ions: role of chitosan and cellulose for water treatment. Int J Pharmacogn 2:280–289
Al-Bastaki N (2004) Removal of methyl orange dye and Na2SO4 salt from synthetic wastewater using reverse osmosis. Chem Eng Process 43:1561–1567
Anitha T (2018) Novel chitosan blended polymers for the removal of rose Bengal dye: adsorption isotherms, kinetics and mechanism. Int J Environ Waste Manage 22:87–110
Anitha T, Kumar PS, Kumar KS, Sriram K, Ahmed JF (2016) Biosorption of lead(II) ions onto nano-sized chitosan particle blended polyvinyl alcohol (PVA): adsorption isotherms, kinetics and equilibrium studies. Desalin Water Treat 57:1–11
Apostol LC, Pereira L, Pereira R, Gavrilescu M, Alves MM (2012) Biological decolourization of xanthene dyes by anaerobic granular biomass. Biodegradation 23:725–737
Arslan-Alaton I, Balcioglu IA, Bahnemann DW (2000) Advanced chemical oxidation of reactive dyes in simulated dyehouse effluents by ferrioxalate-Fenton/UV-A and TiO2/UV-A processes. Dyes Pigm 47:207–218
Arvand M, Pakseresht MA (2013) Cadmium adsorption on modified chitosan-coated bentonite: batch experimental studies. J Chem Technol Biotechnol 88:572–578
Boyd GE, Adamson AW, Myers LS (1947) The exchange adsorption of ions from aqueous Solutions by organic zeolites. II Kinetics J Am Chem Soc 69:2836–2848
Chiou M-S, Ho P-Y, Li H-Y (2004) Adsorption of anionic dyes in acid solutions using chemically cross-linked chitosan beads. Dyes Pigm 60:69–84
Crini G (2006) Non-conventional low-cost adsorbents for dye removal: a review. Bioresour Technol 97:1061–1085
Crini G, Badot P-M (2008) Application of chitosan, a natural aminopolysaccharide, for dye removal from aqueous solutions by adsorption processes using batch studies: a review of recent literature. Prog Polym Sci 33:399–447
Dubinin MM, Radushkevich LV (1947) Equation of the characteristic curve of activated charcoal. Chem Zentralbl 1:875–890
Dutta S, Bhattacharyya A, Ganguly A, Gupta S, Basu S (2011) Application of response surface methodology for preparation of low-cost adsorbent from citrus fruit peel and for removal of methylene blue. Desalination 275:26–36
El Mouzdahir Y, Elmchaouri A, Mahboub R, Gil A, Korili SA (2010) Equilibrium modeling for the adsorption of methylene blue from aqueous solutions on activated clay minerals. Desalination 250:335–338
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–470
Gecgel U, Ozcan G, Gurpinar GC (2013) Removal of methylene blue from aqueous solution by activated carbon prepared from pea shells (Pisum sativum). J chem 1–9
Georgiou D, Aivazidis A, Hatiras J, Gimouhopoulos K (2003) Treatment of cotton textile wastewater using lime and ferrous sulfate. Water Res 37:2248–2250
Gozalvez-Zafrilla JM, Sanz-Escribano D, Lora-Garcia J, Hidalgo MCL (2008) Nanofiltration of secondary effluent for wastewater reuse in the textile industry. Desalination 222:272–279
Gurses A, Karaca S, Dogar C, Bayrak R, Cikyildiz M, Yalcin M (2004) Determination of adsorptive properties of clay/water system: methylene blue sorption. J Colloid Interface Sci 269:310–314
Hassaan MA, El Nemr A (2017) Health and environmental impacts of dyes: mini review. Am J Environ Sci Eng 1:64–67
Hemavathy RV, Kumar PS, Kanmani K, Jahnavi N (2019) Adsorptive separation of Cu(II) ions from aqueous medium using thermally/chemically treated Cassia fistula based biochar. J Clean Prod 249:119390
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465
Jarvis MC (2006) Structure and properties of pectin gels in plant cell walls. Plant Cell Environ 7:153–164
Jin X, Jiang M, Shan X, Pei Z, Chen Z (2008) Adsorption of methylene blue and orange onto unmodified and surfactant-modified zeolite. J Colloid and Interface Sci 328:243–247
Khan S, Malik A (2014) Environmental and health effects of textile industry wastewater. In: Malik A, Grohmann E, Akhtar R. ed. Environmental deterioration and human health. Natural and Anthropogenic Determinants, Netherlands, Springer 55-71
Kumar MNVR (2000) A review of chitin and chitosan applications. React Funct Polym 46:1–27
Kumar S, Koh J (2012) Physiochemical, optical and biological activity of chitosan- chromone derivative for biomedical applications. Int J Mol Sci 13:6102–6116
Kumar PS, Senthamarai C, Deepthi ASLS, Bharani R (2013) Adsorption isotherms, kinetics and mechanism of Pb(II) ions removal from aqueous solution using chemically modified agricultural waste. Can J Chem Eng 91:1950–1956
Lagergren S (1898) About the theory of so-called adsorption of soluble substances. Kungliga Svenska Vetensk Handl 4:1–39
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403
Martin RW Jr, Baillod CR, Mihelcic JR (2005) Low-temperature inhibition of the activated sludge process by an industrial discharge containing the azo dye acid black 1. Water Res 39:17–28
Merzouk B, Madani K, Sekki A (2010) Using electrocoagulation–electroflotation technology to treat synthetic solution and textile wastewater, two case studies. Desalination 250:573–577
Moussavi G, Mahmoudi M (2009) Removal of azo and anthraquinone reactive dyes from industrial wastewaters using MgO nanoparticles. J Hazard Mater 168:806–812
Murcia-Salvador A, Pellicer JA, Fortea MI, Gomez-Lopez VM, Rodriguez-Lopez MI, Nunez-Delicado E, Gabaldon JA (2019) Adsorption of direct blue 78 using chitosan and cyclodextrins as adsorbents. Polymers (basel) 11:1003
Naqash F, Masoodi FA, Rather SA, Wani SM, Gani A (2017) Emerging concepts in the nutraceutical and functional properties of pectin - a review. Carbohydr Polym 168:227–239
Ngah WSW, Teong LC, Hanafiah MAKM (2011) Adsorption of dyes and heavy metal ions by chitosan composites: a review. Carbohydr Polym 83:1446–1456
Pearce CI, Lloyd JR, Guthrie JT (2003) The removal of colour from textile wastewater using whole bacterial cells: a review. Dyes Pigm 58:179–196
Pooladi A, Bazargan-Lari R (2020) Simultaneous removal of copper and zinc ions by chitosan/hydroxyapatite/nano-magnetite composite. J Mater Res Tech 9:14841–14852
Ranganathan K, Jeyapaul S, Sharma DC (2007) Assessment of water pollution in different bleaching-based paper manufacturing and textile dyeing industries in India. Environ Monit Assess 134:363–372
Reymond P, Grunberger S, Paul K, Muller M, Farmer EE (1995) Oligogalacturonide defense signals in plants: large fragments interact with the plasma membrane in vitro. PNAS 92:4145–4149
Rieman W, Walton HF (1970) Ion exchange in analytical chemistry. International series of monographs on analytical chemistry. 38. Oxford, Pergamon Press
Sakkayawong N, Thiravetyan P, Nakbanpote W (2005) Adsorption mechanism of synthetic reactive dye wastewater by chitosan. J Colloid Interface Sci 286:36–42
Sathya K, Nagarajan K, Malar CG, Rajalakshmi S, Rajalakshmi P (2022) A comprehensive review on comparison among effluent treatment methods and modern methods of treatment of industrial wastewater effluent from different sources. Appl Water Sci 12:70
Seenuvasan M, Suganthi JRG, Sarojini G, Malar GCG, Priya ME, Kumar MA (2018) Effective utilization of crustacean shells for preparing chitosan composite beads: applications in ameliorating the biosorption of an endocrine disrupting heavy metal. Desalin Water Treat 121:28–35
Seenuvasan M, Malar CG, Growther L (2021) Production of a biopolymer film from biological wastes and its statistical analysis. Biores Tech Rep 13:100610
Shi B, Li G, Wang D, Feng C, Tang H (2007) Removal of direct dyes by coagulation: the performance of preformed polymeric aluminum species. J Hazard Mater 143:567–574
Sila DN, Buggenhout SV, Duvetter T, Fraeye I, Roeck AD, Loey AV, Hendrickx M (2009) Pectins in processed fruits and vegetables: part II-structure-function relationships. Compr Rev Food Sci Food Saf 8:86–104
Temkin MJ, Pyzhev V (1940) Recent modifications to langmuir isotherms. Acta Physicochim URSS 12:217–225
Tony BD, Goyal D, Khanna S (2009) Decolourization of textile azo dyes by aerobic bacterial consortium. Int Biodeterior Biodegradation 63:462–469
Trikkaliotis DG, Christoforidis AK, Mitropoulos AC, Kyzas GZ (2020) Adsorption of copper ions onto chitosan/poly(vinyl alcohol) beads functionalized with poly(ethylene glycol). Carbohydr Polym 234:115890
Vadivelan V, Kumar V (2005) Equilibrium kinetics, mechanism and process design for the sorption of methylene blue onto rice husk. J Colloid Interface Sci 286:90–100
Wang C, Yediler A, Lienert D, Wang Z, Kettrup A (2003) Ozonation of an azo dye C.I. Remazol Black 5 and toxicological assessment of its oxidation products. Chemosphere 52:1225–1232
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div, Am Soc Civ Eng 89:31–60
Yasin Y, Hussein MZ, Ahmad FH (2007) Adsorption of methylene blue onto treated activated carbon. The Malaysian J Analytical Sci 11:400–406
Yong SK, Shrivastava M, Srivastava P, Kunhikrishnan A, Bolan N (2015) Environmental applications of chitosan and its derivatives. Rev Environ Contam Toxicol 233:1–43
Zhao M, Tang Z, Liu P (2008) Removal of methylene blue from aqueous solution with silica nano-sheets derived from vermiculate. J Hazard Mater 158:43–51
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the study and to the manuscript preparation. Material preparation, data collection, and analysis were performed by Anitha Thulasisingh. Supervision was done by Seenuvasan Muthulingam. The first draft of the manuscript was written by Mohan Kumar BS, Naveenraj Rajasekar, and Shantanu Mohanraj. Manuscript drafting and editing were done by Carlin Geor Malar. All the authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable
Consent to participate
Not applicable
Consent for publication
All the authors have accepted for publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thulasisingh, A., Muthulingam, S., Kumar, M. et al. Biosorption of methylene blue dye using a novel chitosan pectinase blend. Environ Sci Pollut Res 30, 48948–48961 (2023). https://doi.org/10.1007/s11356-022-24996-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-24996-1