Abstract
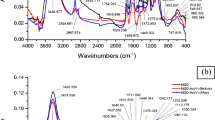
Heavy metals present in industrial effluents, when discharged into water channels, not only affect humans but also negatively impact plants and aquatic organisms. Sawdust is available readily in developing countries and can be used by small-scale industries for effluent water treatment containing low concentrations of bivalent zinc ions. This study explores the potential of sawdust-derived biosorbents, after boiling (SDB), chemical modification with formaldehyde (SDF), and sulfuric acid (SDS), for sequestration of Zn(II) from simulated wastewater as well as industrial effluents. The morphological analysis of the three biosorbents indicated a suitable porous structure with a pore size of 232.928 m2/g (SDB), 291.102 m2/g (SDF), and 498.873 m2/g (SDS). The functional analysis of native and metal-laden biosorbents indicated the role of − OH, − C = O, and − NH functional groups in Zn(II) binding. The process parameters were optimized and the spontaneous adsorption of Zn(II) was found to proceed by multilayer formation by following pseudo-second-order kinetics. SDS adsorbent (0.1 g) exhibited a greater potential for removal of Zn(II) from industrial effluents as compared to SDB and SDF at pH = 6.0 with the equilibrium adsorption capacity of 45.87 mg/g. Therefore, SDS could be a promising adsorbent for the treatment of wastewater in small-scale industries.






Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmad A, Rafatullah M, Sulaiman O et al (2009) Removal of Cu(II) and Pb(II) ions from aqueous solutions by adsorption on sawdust of Meranti wood. Desalination 247:636–646. https://doi.org/10.1016/j.desal.2009.01.007
Ali MM, Bhakta JN (2020) Biosorption of zinc from aqueous solution using leaves of Corchorus olitorius as a low-cost biosorbent. Water Environ Res 92:821–828. https://doi.org/10.1002/wer.1274
Ali Z, Ahmad R, Aslam Farooq W, et al (2020) Synthesis and characterization of functionalized nanosilica for zinc ion mitigation; experimental and computational investigations. Molecules 25https://doi.org/10.3390/molecules25235534
Al-Malack MH, Dauda M (2017) Competitive adsorption of cadmium and phenol on activated carbon produced from municipal sludge. J Environ Chem Eng 5:2718–2729. https://doi.org/10.1016/j.jece.2017.05.027
APHA (1998) Standard methods for the examination of water and wastewater. 20th Edition, American Public Health Association, American Water Works Association and Water Environmental Federation, Washington DC
Arshad N, Imran S (2020) Indigenous waste plant materials: an easy and cost-effective approach for the removal of heavy metals from water. Curr Res Green Sustain Chem 3:100040. https://doi.org/10.1016/j.crgsc.2020.100040
Bansal M, Garg R, Garg VK et al (2022) Sequestration of heavy metal ions from multi-metal simulated wastewater systems using processed agricultural biomass. Chemosphere 296:133966. https://doi.org/10.1016/j.chemosphere.2022.133966
Cai T, Liu X, Zhang J et al (2021) Silicate-modified oiltea camellia shell-derived biochar: a novel and cost-effective sorbent for cadmium removal. J Clean Prod 281:125390. https://doi.org/10.1016/j.jclepro.2020.125390
Castro L, Blázquez ML, González F et al (2017) Biosorption of Zn(II) from industrial effluents using sugar beet pulp and F. vesiculosus: from laboratory tests to a pilot approach. Sci Total Environ 598:856–866. https://doi.org/10.1016/j.scitotenv.2017.04.138
Castro D, Rosas-Laverde NM, Aldás MB et al (2021) Chemical modification of agro-industrial waste-based bioadsorbents for enhanced removal of Zn(II) ions from aqueous solutions. Materials (basel) 14:2134. https://doi.org/10.3390/ma14092134
Dey S, Basha SR, Babu GV, Nagendra T (2021) Characteristic and biosorption capacities of orange peels biosorbents for removal of ammonia and nitrate from contaminated water. Clean Mater 1:100001. https://doi.org/10.1016/j.clema.2021.100001
Eddy NO, Garg R, Garg R et al (2022) Waste to resource recovery: mesoporous adsorbent from orange peel for the removal of trypan blue dye from aqueous solution. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-022-02571-5
El Hajam M, Kandri NI, Plavan GI et al (2020) Pb2+ ions adsorption onto raw and chemically activated Dibetou sawdust: application of experimental designs. J King Saud Univ - Sci 32:2176–2189. https://doi.org/10.1016/j.jksus.2020.02.027
Ferreira AS, Mota AA, Oliveira AM et al (2019) Equilibrium and kinetic modelling of adsorption: evaluating the performance of adsorbent in softening water for irrigation and animal consumption. Rev Virtual Quim 11:1752–1766. https://doi.org/10.21577/1984-6835.20190123
Garg R, Garg R, Thakur A, Arif SM (2020) Water remediation using biosorbent obtained from agricultural and fruit waste. Mater Today Proc 46:6669–6672. https://doi.org/10.1016/j.matpr.2021.04.132
Garg R, Garg R, Okon Eddy N et al (2022b) Biosynthesized silica-based zinc oxide nanocomposites for the sequestration of heavy metal ions from aqueous solutions. J King Saud Univ - Sci 34:101996. https://doi.org/10.1016/j.jksus.2022.101996
Garg R, Garg R, Khan MA, et al (2022a) Utilization of biosynthesized silica-supported iron oxide nanocomposites for the adsorptive removal of heavy metal ions from aqueous solutions. Environ Sci Pollut Res 1–10. https://doi.org/10.21203/rs.3.rs-1394501/v1
Ghasemi M, Naushad M, Ghasemi N, Khosravi-fard Y (2014) A novel agricultural waste based adsorbent for the removal of Pb(II) from aqueous solution: kinetics, equilibrium and thermodynamic studies. J Ind Eng Chem 20:454–461. https://doi.org/10.1016/j.jiec.2013.05.002
Gopal B, Gupta A (2019) Integrated approach for hazardous Cr(VI) removal: reduction, extraction, and conversion into a photoactive composite, CuO/CuCr2O4. ACS Omega 4:20443–20449. https://doi.org/10.1021/acsomega.9b01452
Hassan SS, Al-Ghouti MA, Abu-Dieyeh M, McKay G (2020) Novel bioadsorbents based on date pits for organophosphorus pesticide remediation from water. J Environ Chem Eng 8https://doi.org/10.1016/j.jece.2019.103593
Hawari A, Rawajfih Z, Nsour N (2009) Equilibrium and thermodynamic analysis of zinc ions adsorption by olive oil mill solid residues. J Hazard Mater 168:1284–1289. https://doi.org/10.1016/j.jhazmat.2009.03.014
Hsieh YY, Tsai YC, He JR et al (2017) Rice husk agricultural waste-derived low ionic content carbon–silica nanocomposite for green reinforced epoxy resin electronic packaging material. J Taiwan Inst Chem Eng 78:493–499. https://doi.org/10.1016/j.jtice.2017.06.010
Hu CY, Lo SL, Kuan WH, Lee YD (2005) Removal of fluoride from semiconductor wastewater by electrocoagulation- flotation. Water Res 39:895–901. https://doi.org/10.1016/j.watres.2004.11.034
Ibeji CU, Akpomie KG, Ugwu CI et al (2020) Sequestration of Pb(II) onto phosphate modified fibrous Prunus dulcis seed shell: kinetic, thermodynamic, isotherm, desorption and reusability. J Nat Fibers 00:1–14. https://doi.org/10.1080/15440478.2020.1848702
Ibrahim NA, Abdellatif FHH, Hasanin MS, Abdellatif MM (2022) Fabrication, characterization, and potential application of modified sawdust sorbents for efficient removal of heavy metal ions and anionic dye from aqueous solutions. J Clean Prod 332:130021. https://doi.org/10.1016/j.jclepro.2021.130021
Idowu AA, Temilade FA, Peter A et al (2019) Agro waste material as ecofriendly adsorbent for the removal of zn(Ii): isotherm, kinetic, thermodynamic and optimization studies. Desalin Water Treat 155:250–258. https://doi.org/10.5004/dwt.2019.23977
Jabasingh SA, Belachew H, Yimam A (2018) Iron oxide induced bagasse nanoparticles for the sequestration of Cr6+ ions from tannery effluent using a modified batch reactor. J Appl Polym Sci 135:1–15. https://doi.org/10.1002/app.46683
Jacob MM, Ponnuchamy M, Kapoor A, Sivaraman P (2020) Bagasse based biochar for the adsorptive removal of chlorpyrifos from contaminated water. J Environ Chem Eng 8:103904. https://doi.org/10.1016/j.jece.2020.103904
Jaradat AQ, Telfah DB, Ismail R (2021) Heavy metals removal from landfill leachate by coagulation/flocculation process combined with continuous adsorption using eggshell waste materials. Water Sci Technol 84:3817–3832. https://doi.org/10.2166/wst.2021.493
Joshi S, Kataria N, Garg VK, Kadirvelu K (2020) Pb2+ and Cd2+ recovery from water using residual tea waste and SiO2@TW nanocomposites. Chemosphere 257:127277. https://doi.org/10.1016/j.chemosphere.2020.127277
Kalavathy MH, Miranda LR (2010) Comparison of copper adsorption from aqueous solution using modified and unmodified Hevea brasiliensis saw dust. Desalination 255:165–174. https://doi.org/10.1016/j.desal.2009.12.028
Kayranli B (2021) Mechanism of interaction and removal of zinc with lignocellulosic adsorbents, closing the cycle with a soil conditioner. J King Saud Univ - Sci 33:101607. https://doi.org/10.1016/j.jksus.2021.101607
Khayyun TS, Mseer AH (2019) Comparison of the experimental results with the Langmuir and Freundlich models for copper removal on limestone adsorbent. Appl Water Sci 9:1–8. https://doi.org/10.1007/s13201-019-1061-2
Kononova ON, Bryuzgina GL, Apchitaeva OV, Kononov YS (2019) Ion exchange recovery of chromium (VI) and manganese (II) from aqueous solutions. Arab J Chem 12:2713–2720. https://doi.org/10.1016/j.arabjc.2015.05.021
Kulasooriya TPK, Priyantha N, Navaratne AN (2020) Removal of textile dyes from industrial effluents using burnt brick pieces: adsorption isotherms, kinetics and desorption. SN Appl Sci 2:1–11. https://doi.org/10.1007/s42452-020-03533-0
Lin S, Guo Y, Zhou L, Yang H (2022) Quantitative effects of surface oxidation on biochar derived from long-root Eichhornia Crassipes plants as Cd2+ adsorbent. Bioresour Technol Reports 17:100937. https://doi.org/10.1016/j.biteb.2021.100937
Luo JH, Li J, Guo L et al (2017) Removal of chromium(III) from aqueous waste solution by liquid-liquid extraction in a circular microchannel. Water Sci Technol 76:2313–2320. https://doi.org/10.2166/wst.2017.371
Muthumareeswaran MR, Alhoshan M, Agarwal GP (2017) Ultrafiltration membrane for effective removal of chromium ions from potable water. Sci Rep 7:1–12. https://doi.org/10.1038/srep41423
Rani S, Bansal M, Kaur K, Sharma S (2019) Adsorption isotherm, kinetics and thermodynamics of bivalent nickel scavenging utilising sawdust carbon. Rasayan J Chem 12:1315–1325. https://doi.org/10.31788/RJC.2019.1235279
Ren B, Jin Y, Zhao L et al (2022) Enhanced Cr(VI) adsorption using chemically modified dormant Aspergillus niger spores: process and mechanisms. J Environ Chem Eng 10:106955. https://doi.org/10.1016/j.jece.2021.106955
Rezgui A, Guibal E, Boubakera T (2017) Sorption of Hg(II) and Zn(II) ions using lignocellulosic sorbent (date pits). Can J Chem Eng 95:775–782. https://doi.org/10.1002/cjce.22728
Saruchi, Kumar V (2019) Adsorption kinetics and isotherms for the removal of rhodamine B dye and Pb +2 ions from aqueous solutions by a hybrid ion-exchanger. Arab J Chem 12:316–329. https://doi.org/10.1016/j.arabjc.2016.11.009
Segovia-Sandoval SJ, Ocampo-Pérez R, Berber-Mendoza MS et al (2018) Walnut shell treated with citric acid and its application as biosorbent in the removal of Zn(II). J Water Process Eng 25:45–53. https://doi.org/10.1016/j.jwpe.2018.06.007
Shikuku VO, Mishra T (2021) Adsorption isotherm modeling for methylene blue removal onto magnetic kaolinite clay: a comparison of two-parameter isotherms. Appl Water Sci 11:1–9. https://doi.org/10.1007/s13201-021-01440-2
Singha AS, Guleria A (2015) Utility of chemically modified agricultural waste okra biomass for removal of toxic heavy metal ions from aqueous solution. Eng Agric Environ Food 8:52–60. https://doi.org/10.1016/j.eaef.2014.08.001
Sreedhar I, Reddy NS (2019) Heavy metal removal from industrial effluent using bio-sorbent blends. SN Appl Sci 1:1–15. https://doi.org/10.1007/s42452-019-1057-4
Zand AD, Abyaneh MR (2020) Adsorption of lead, manganese, and copper onto biochar in landfill leachate: implication of non-linear regression analysis. Sustain Environ Res 30https://doi.org/10.1186/s42834-020-00061-9
Zhang W, An Y, Li S et al (2020) Enhanced heavy metal removal from an aqueous environment using an eco-friendly and sustainable adsorbent. Sci Rep 10:1–19. https://doi.org/10.1038/s41598-020-73570-7
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception, design, literature review, data collection, and analysis. All authors read, edited, and approved the final manuscript. Savita Rani: conceptualization, investigation, data curation, and writing — original draft. Seema Sharma: supervision, methodology, validation, resources, and writing — review and editing. Manjeet Bansal: supervision, methodology, validation, resources, and writing — review and editing. Rajni Garg: conceptualization, methodology, formal analysis, and writing — review and editing. Rishav Garg: conceptualization, visualization, formal analysis, and writing — review and editing.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All authors approve the ethics and consent to participate in this research.
Consent for publish
All authors have consented to publish this paper.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rani, S., Sharma, S., Bansal, M. et al. Enhanced Zn(II) adsorption by chemically modified sawdust based biosorbents. Environ Sci Pollut Res 30, 99046–99061 (2023). https://doi.org/10.1007/s11356-022-22963-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22963-4