Abstract
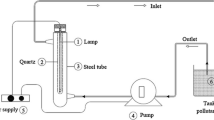
Due to continuous industrialization, the discharge of hazardous dyes has enormously disrupted the ecosystem causing environmental problems. Due to the stable recalcitrant nature of dyes, advanced catalytic ozonation processes with the latest catalyst are under investigation. Fe-RGO is an effective oxidation catalyst, and the metal loaded platform provides enhanced catalytic performance. This study aims to investigate the effectiveness of Fe-RGO/O3 process for the removal of dyes. In the current research, the application of iron-coated reduced graphene oxide (Fe-RGO) was studied as a catalyst in the heterogeneous catalytic ozonation process to remove dyes. Methylene blue (MB) was selected as a model pollutant. RGO was prepared using the improved Hummers method and was coated with iron (Fe) implying the impregnation method. The FTIR, SEM–EDX, XRD, and BET analyses of RG and Fe-RGO were performed to characterize the catalyst. The effect of various parameters such as pH (3–10), catalyst dose (0.01–0.04 g), and radical scavengers (NaHCO3, NaCl) on removal efficiency was elucidated. The result revealed an excellent catalytic efficiency of Fe-RGO in the ozonation process. At optimum conditions, 96% removal efficiency was achieved in catalytic ozonation at pH 7 with a catalyst dose of 0.02 g and ozone dose 0.5 mg/min, after 10 min. Interestingly, a slight decrease in removal efficiency was observed in the catalytic ozonation process in hydroxyl radical scavengers (NaCl and NaHCO3), which makes the proposed catalyst more applicable in real conditions. Therefore, it is concluded that Fe-RGO can be used as an excellent catalyst for the removal of dyes in real conditions where radical scavengers may be present in a significant amount.











Similar content being viewed by others
Data availability
Not applicable.
References
Afzal A, Abuilaiwi FA, Javaid R et al (2020a) Solid-state synthesis of heterogeneous Ni0.5Cu0.5-xZnxFe2O4 spinel oxides with controlled morphology and tunable dielectric properties. J Mater Sci Mater Electron 31:14261–14270. https://doi.org/10.1007/s10854-020-03982-8
Afzal A, Mujahid A, Iqbal N et al (2020b) Enhanced high-temperature (600 ◦C) NO2 response of ZnFe2O4 nanoparticle-based exhaust gas sensors. Nanomaterials 10:2133
Akerdi AG, Bahrami SH, Arami M, Pajootan E (2016) Photocatalytic discoloration of Acid Red 14 aqueous solution using titania nanoparticles immobilized on graphene oxide fabricated plate. Chemosphere 159:293–299. https://doi.org/10.1016/j.chemosphere.2016.06.020
Andrijanto E, Shoelarta S, Subiyanto G, Rifki S (2016) Facile synthesis of graphene from graphite using ascorbic acid as reducing agent. In: AIP Conference Proceedings. Am Inst Phys Inc. 020003. https://doi.org/10.1063/1.4945457
Bano N, Hussain I, El-Naggar AM, Albassam AA (2019) Reduced graphene oxide nanocomposites for optoelectronics applications. Appl Phys A Mater Sci Process 125:215. https://doi.org/10.1007/s00339-019-2518-8
Bhattacharya S, Mukhopadhyay A, Chattopadhyay D et al (2013) Role of nanotechnology in water treatment and purification: potential applications and implications. Int J Chem Sci Technol 3:59–64
Brienza M, Katsoyiannis IA (2017) Sulfate radical technologies as tertiary treatment for the removal of emerging contaminants from wastewater 9(9):1604. https://doi.org/10.3390/su9091604
Emiru TF, Ayele DW (2017) Controlled synthesis, characterization and reduction of graphene oxide: a convenient method for large scale production. Egypt J Basic Appl Sci 4:74–79. https://doi.org/10.1016/j.ejbas.2016.11.002
Ernst M, Lurot F, Schrotter JC (2004) Catalytic ozonation of refractory organic model compounds in aqueous solution by aluminum oxide. Appl Catal B Environ 47:15–25. https://doi.org/10.1016/S0926-3373(03)00290-X
Fil BA, Özmetin C, Korkmaz M (2012) Cationic dye (methylene blue) removal from aqueous solution by montmorillonite. Bull Korean Chem Soc 33:3184–3190. https://doi.org/10.5012/bkcs.2012.33.10.3184
Flörke M, Kynast E, Bärlund I et al (2013) Domestic and industrial water uses of the past 60 years as a mirror of socio-economic development: a global simulation study. Glob Environ Chang 23:144–156. https://doi.org/10.1016/j.gloenvcha.2012.10.018
Forzatti P, Lietti L (1999) Catalyst deactivation. Catal Today 52:165–181. https://doi.org/10.1016/S0920-5861(99)00074-7
Gebreegziabher GG, Asemahegne AS, Ayele DW et al (2019) One-step synthesis and characterization of reduced graphene oxide using chemical exfoliation method. Mater Today Chem 12:233–239. https://doi.org/10.1016/j.mtchem.2019.02.003
Hu L, Hao GX, Luo HD et al (2018) Bifunctional 2D Cd(II)-based metal-organic framework as efficient heterogeneous catalyst for the formation of C-C bond. Cryst Growth Des 18:2883–2889. https://doi.org/10.1021/acs.cgd.7b01728
Ikhlaq A, Brown DR, Kasprzyk-Hordern B (2012) Mechanisms of catalytic ozonation on alumina and zeolites in water: formation of hydroxyl radicals. Appl Catal B Environ 123–124:94–106. https://doi.org/10.1016/j.apcatb.2012.04.015
Ikhlaq A, Fatima R, Qazi UY et al (2021) Combined iron-loaded zeolites and ozone-based process for the purification of drinking water in a novel hybrid reactor: removal of faecal coliforms and arsenic. Catalysts 11:1–21. https://doi.org/10.3390/catal11030373
Ikhlaq A, Munir HMS, Khan A et al (2019) Comparative study of catalytic ozonation and Fenton-like processes using iron-loaded rice husk ash as catalyst for the removal of methylene blue in wastewater. Ozone Sci Eng 41:250–260. https://doi.org/10.1080/01919512.2018.1525276
Ikhlaq A, Javaid R, Akram A, Qazi UY, Erfan J, Madkour M, Abdelbagi MEM, Ibn Shamsah SM, Qi F (2022) Application of attapulgite clay-based Fe-Zeolite 5A in UV-assisted catalytic ozonation for the removal of ciprofloxacin. https://doi.org/10.1155/2022/2846453
Javaid R (2021) Catalytic hydrogen production, storage and application. Catalysts 11:836
Javaid R, Aoki Y, Nanba T (2020) Highly efficient Ru/MgO–Er2O3 catalysts for ammonia synthesis. J Phys Chem Solids 146:109570. https://doi.org/10.1016/j.jpcs.2020.109570
Javaid R, Kawanami H, Chatterjee M et al (2011) Sonogashira C-C coupling reaction in water using tubular reactors with catalytic metal inner surface. Chem Eng J 167:431–435. https://doi.org/10.1016/j.cej.2010.08.080
Javaid R, Kawanami H, Chatterjee M et al (2010) Fabrication of microtubular reactors coated with thin catalytic layer (M=Pd, Pd-Cu, Pt, Rh, Au). Catal Commun 11:1160–1164. https://doi.org/10.1016/j.catcom.2010.05.018
Javaid R, Kawasaki S-I, Ookawara R et al (2013a) Continuous dehydrogenation of aqueous formic acid under sub-critical conditions by use of hollow tubular reactor coated with thin palladium oxide layer. J Chem Eng Japan 46:751–758. https://doi.org/10.1252/jcej.13we184
Javaid R, Kawasaki S-I, Suzuki A, Suzuki TM (2013b) Simple and rapid hydrogenation of p-nitrophenol with aqueous formic acid in catalytic flow reactors. Beilstein J Org Chem 9:1156–1163. https://doi.org/10.3762/bjoc.9.129
Javaid R, Matsumoto H, Nanba T (2019) Influence of reaction conditions and promoting role of ammonia produced at higher temperature conditions in its synthesis process over Cs-Ru/MgO Catalyst. ChemistrySelect 4:2218–2224. https://doi.org/10.1002/slct.201803813
Javaid R, Nanba T (2020) Effect of reaction conditions and surface characteristics of Ru/CeO2 on catalytic performance for ammonia synthesis as a clean fuel. Int J Hydrogen Energy 46:18107–18115. https://doi.org/10.1016/j.ijhydene.2020.07.222
Javaid R, Nanba T (2021a) Effect of texture and physical properties of catalysts on ammonia synthesis. Catal Today. https://doi.org/10.1016/j.cattod.2021.06.007
Javaid R, Nanba T (2020b) MgFe 2 O 4 -supported Ru catalyst for ammonia synthesis: promotive effect of chlorine. ChemistrySelect 5:4312–4315. https://doi.org/10.1002/slct.202000883
Javaid R, Qazi UY (2019) Catalytic oxidation process for the degradation of synthetic dyes: An overview. Int J Environ Res Public Health 16:2066. https://doi.org/10.3390/ijerph16112066
Javaid R, Qazi UY, Ikhlaq A et al (2021) Subcritical and supercritical water oxidation for dye decomposition. J Environ Manage 290:112605. https://doi.org/10.1016/j.jenvman.2021.112605
Javaid R, Qazi UY, Kawasaki SI (2016) Highly efficient decomposition of Remazol Brilliant Blue R using tubular reactor coated with thin layer of PdO. J Environ Manage 180:551–556. https://doi.org/10.1016/j.jenvman.2016.05.075
Javaid R, Qazi UY, Kawasaki SI (2015a) Efficient and continuous decomposition of hydrogen peroxide using a silica capillary coated with a thin palladium or platinum layer. Bull Chem Soc Jpn 88:976–980. https://doi.org/10.1246/bcsj.20150052
Javaid R, Tanaka DAP, Kawanami H, Suzuki TM (2009) Silica capillary with thin metal (Pd and Pt) inner wall: application to continuous decomposition of hydrogen peroxide. Chem Lett 38:146–147. https://doi.org/10.1246/cl.2009.146
Javaid R, Nanba T (2021) Effect of preparation method and reaction parameters on catalytic activity for ammonia synthesis. Int J Hydrog Energy 46(71):35209–35218. https://doi.org/10.1016/j.ijhydene.2021.08.082
Javaid R, Urata K, Furukawa S, Komatsu T (2015b) Factors affecting coke formation on H-ZSM-5 in naphtha cracking. Appl Catal A Gen 491.https://doi.org/10.1016/j.apcata.2014.12.002
Kasanen J, Salstela J, Suvanto M, Pakkanen TT (2011) Photocatalytic degradation of methylene blue in water solution by multilayer TiO 2 coating on HDPE. Appl Surf Sci 258:1738–1743. https://doi.org/10.1016/j.apsusc.2011.10.028
Khuntia S, Majumder SK, Ghosh P (2016) Catalytic ozonation of dye in a microbubble system: hydroxyl radical contribution and effect of salt. J Environ Chem Eng 4:2250–2258. https://doi.org/10.1016/j.jece.2016.04.005
Kumar A, Sadanandhan AM, Jain SL (2019) Silver doped reduced graphene oxide as a promising plasmonic photocatalyst for oxidative coupling of benzylamines under visible light irradiation. New J Chem 43:9116–9122. https://doi.org/10.1039/c9nj00852g
Lavin-Lopez MP, Paton-Carrero A, Sanchez-Silva L et al (2017) Influence of the reduction strategy in the synthesis of reduced graphene oxide. Adv Powder Technol 28:3195–3203. https://doi.org/10.1016/j.apt.2017.09.032
Liu C, Liu L, Tian X et al (2019) Coupling metal–organic frameworks and g-C3N4 to derive Fe@N-doped graphene-like carbon for peroxymonosulfate activation: upgrading framework stability and performance. Appl Catal B Environ 255:117763. https://doi.org/10.1016/j.apcatb.2019.117763
Liu X, Hou Y, Guo J, et al (2015) Catalytic ozone aqueous decomposition of methylene blue using composite metal oxides. In: IOP Conference Series: Materials Science and Engineering. Institute of Physics Publishing, p 012031
Lucas MS, Peres JA (2007) Degradation of Reactive Black 5 by Fenton/UV-C and ferrioxalate/H2O2/solar light processes. Dye Pigment 74:622–629. https://doi.org/10.1016/j.dyepig.2006.04.005
Masoumbeigi H, Rezaee A (2015) Removal of Methylene Blue (MB) dye from synthetic wastewater using UV / H 2 O 2 advanced oxidation process. Heal Policy Sustain Heal 2:60–166
Misra AK (2014) Climate change and challenges of water and food security. Int J Sustain Built Environ 3:153–165
Mobasser S, Akbar Firoozi A (2016) Review of nanotechnology applications in science and engineering. J Civil Eng Urban 6(4):84–93
Mondal S (2008) Methods of dye removal from dye house effluent—an overview. Environ Eng Sci 25:383–396
Munasir, Kusumawati RP (2019) Synthesis and characterization of Fe3O4@rGO composite with wet-mixing (ex-situ) process. Journal of Physics: Conference Series. 1171(1):12048. https://doi.org/10.1088/1742-6596/1171/1/012048
Muzyka R, Drewniak S, Pustelny T et al (2018) Characterization of graphite oxide and reduced graphene oxide obtained from different graphite precursors and oxidized by different methods using Raman spectroscopy. Materials (basel) 11:1050. https://doi.org/10.3390/ma11071050
Nagarkar MG, Chitodakar VD, Mandake MB (2017) Catalytic ozonation: a rising advanced oxidation technology for textile dyes. Int J Innov Res Sci Eng Technol 6:6789–6796. https://doi.org/10.15680/IJIRSET.2017.0604098
Nanba T, Javaid R, Matsumoto H (2019a) Ammonia synthesis catalyst for effective storage of renewable energy. Fine Chem 48:6–11
Nanba T, Javaid R, Matsumoto H (2019b) Ammonia synthesis by using hydrogen produced from renewable energy. 触媒 Catalysts Catal 61:66–71
Nanba T, Nagata Y, Kobayashi K, et al (2021) Explorative study of a Ru/CeO2 catalyst for NH3 synthesis from renewable hydrogen and demonstration of NH3 synthesis under a range of reaction conditions. J Jpn Petrol Inst 64(1):1–9
Naseem T, Zain-ul-Abdin WM et al (2020) Reduced graphene oxide/zinc oxide nanocomposite: from synthesis to its application for wastewater purification and antibacterial activity. J Inorg Organomet Polym Mater 30:3907–3919. https://doi.org/10.1007/s10904-020-01529-2
Pease C, Rücker T, Birk T (2016) Review of the evidence from epidemiology, toxicology, and lung bioavailability on the carcinogenicity of inhaled iron oxide particulates. Chem Res Toxicol 29:237–254
Qazi UY (2021) Silver nanoparticles formation by nanosecond pulsed laser irradiation in an aqueous solution of silver nitrate; effect of sodium bis (2-ethyl hexyl) sulfosuccinate. J New Mater Electrochem Syst 24:38–42. https://doi.org/10.14447/jnmes.v24i1.a07
Qazi UY (2021) Influence of surfactant additives on photochemical synthesized silver nanoparticles using UV pulsed laser irradiations in aqueous silver nitrate solution. J New Mater Electrochem Syst 24:103–110. https://doi.org/10.14447/jnmes.v24i2.a07
Qazi UY, Javaid R (2016) A review on metal nanostructures: preparation methods and their potential applications. Adv Nanoparticles 05:27–43. https://doi.org/10.4236/anp.2016.51004
Qazi UY, Javaid R, Tahir N et al (2020) Design of advanced self-supported electrode by surface modification of copper foam with transition metals for efficient hydrogen evolution reaction. Int J Hydrogen Energy 45:3396–33406. https://doi.org/10.1016/j.ijhydene.2020.09.026
Qazi UY, Javaid R, Zahid M et al (2021) Bimetallic NiCo–NiCoO2 nano-heterostructures embedded on copper foam as a self-supported bifunctional electrode for water oxidation and hydrogen production in alkaline media. Int J Hydrogen Energy 46:18936–18948. https://doi.org/10.1016/j.ijhydene.2021.03.046
Qazi UY, Shervani Z, Javaid R et al (2017a) Formation and growth of silver nanocubes upon nanosecond pulsed laser irradiation: effects of laser intensity and irradiation time. Adv Nanoparticles 06:148–157. https://doi.org/10.4236/anp.2017.64013
Qazi UY, Shervani Z, Javaid R (2018) Green synthesis of silver nanoparticles by pulsed laser irradiation: effect of hydrophilicity of dispersing agents on size of particles. Front Nanosci Nanotechnol 4:61–69. https://doi.org/10.4236/anp.2015.43008
Qazi UY, Yuan CZ, Ullah N et al (2017b) One-step growth of iron-nickel bimetallic nanoparticles on FeNi alloy foils: highly efficient advanced electrodes for the oxygen evolution reaction. ACS Appl Mater Interfaces 9:28627–28634. https://doi.org/10.1021/acsami.7b08922
Rahimi R, Moshari M, Rabbani M, Azad A (2016) Photooxidation of benzyl alcohols and photodegradation of cationic dyes by Fe3O4@sulfur/reduced graphene oxide as catalyst. RSC Adv 6:41156–41164. https://doi.org/10.1039/c6ra00137h
Razumovskii SD, Konstantinova ML, Grinevich TV et al (2010) Mechanism and kinetics of the reaction of ozone with sodium chloride in aqueous solutions. Kinet Catal 51:492–496. https://doi.org/10.1134/S0023158410040051
Saputra E, Saputra R, Nugraha MW, et al (2018) Removal of Methylene Blue from aqueous solution using spent bleaching earth. IOP Conference Series: Mater Sci and Eng 345(1):012008. https://doi.org/10.1088/1757-899X/345/1/012008
Senthilkumaar S, Varadarajan PR, Porkodi K, Subbhuraam CV (2005) Adsorption of methylene blue onto jute fiber carbon: kinetics and equilibrium studies. J Colloid Interface Sci 284:78–82. https://doi.org/10.1016/j.jcis.2004.09.027
Sivakumar B (2011) Water crisis: from conflict to cooperation—an overview. Hydrol Sci J 56:531–552. https://doi.org/10.1080/02626667.2011.580747
Staehelln J, Hoigne J (1985) Decomposition of ozone in water in the presence of organic solutes acting as promoters and inhibitors of radical chain reactions. Environ Sci Technol 19:1206–1213. https://doi.org/10.1021/es00142a012
Sun C, Yang ST, Gao Z et al (2019) Fe3O4/TiO2/reduced graphene oxide composites as highly efficient Fenton-like catalyst for the decoloration of methylene blue. Mater Chem Phys 223:751–757. https://doi.org/10.1016/j.matchemphys.2018.11.056
Wang B, Zhang H, Wang F et al (2019) Application of heterogeneous catalytic ozonation for refractory organics in wastewater. Catalysts 9:241. https://doi.org/10.3390/catal9030241
Wang J, Chen H (2020) Catalytic ozonation for water and wastewater treatment: recent advances and perspective. Sci Total Environ 704:135249
Wang Y, Xie Y, Sun H et al (2016) Efficient catalytic ozonation over reduced graphene oxide for p-hydroxylbenzoic acid (PHBA) destruction: active site and mechanism. ACS Appl Mater Interfaces 8:9710–9720. https://doi.org/10.1021/acsami.6b01175
Yang S, Li M, Guo Z et al (2019) Facile synthesis of Fe-MOF/rGO nanocomposite as an efficient electrocatalyst for nonenzymatic H 2 O 2 sensing. Int J Electrochem Sci 14:7703–7716. https://doi.org/10.20964/2019.08.42
Yu X, Qiang L (2012) Preparation for graphite materials and study on electrochemical degradation of phenol by graphite cathodes. Adv Mater Phys Chem 02:63–68. https://doi.org/10.4236/ampc.2012.22011
Yuan CZ, Sun ZT, Jiang YF et al (2017) One-step in situ growth of iron–nickel sulfide nanosheets on FeNi alloy foils: high-performance and self-supported electrodes for water oxidation. Small 13:1–8. https://doi.org/10.1002/smll.201604161
Zhang D, Liu J, Chang H et al (2015) Characterization of a hybrid composite of SnO2 nanocrystal-decorated reduced graphene oxide for ppm-level ethanol gas sensing application. RSC Adv 5:18666–18672. https://doi.org/10.1039/c4ra14611e
Zhang S, Wang D, Zhang S et al (2013) Ozonation and carbon-assisted ozonation of methylene blue as model compound: effect of solution pH. Procedia Environ Sci 18:493–502. https://doi.org/10.1016/j.proenv.2013.04.066
Zhang X, Cai W, Hao L, et al (2017) Preparation of Fe3O4/reduced graphene oxide nanocomposites with good dispersibility for delivery of paclitaxel. J Nanomater 2017.https://doi.org/10.1155/2017/6702890
Zhang Y, Shaad K, Vollmer D, Ma C (2021) Treatment of textile wastewater using advanced oxidation processes—A critical review 13(24):3515. https://doi.org/10.3390/w13243515
Zhu S, Guo J, Dong J et al (2013) Sonochemical fabrication of Fe3O4 nanoparticles on reduced graphene oxide for biosensors. Ultrason Sonochem 20:872–880. https://doi.org/10.1016/j.ultsonch.2012.12.001
Acknowledgements
Dr. Umair Yaqub Qazi and Dr. Rahat Javaid extend their appreciation to the Deanship of Scientific Research, University of Hafr Al Batin for funding this work through the research group project no. IFP-A-2022-2-4-14. We highly appreciate the support of the Institute of Environmental Engineering, UET Lahore, Pakistan.
Funding
Funding from Deanship of Scientific Research, University of Hafr Al Batin under research group project no. IFP-A-2022–2-4–14 was used to conduct experimental and characterization studies in this paper.
Author information
Authors and Affiliations
Contributions
All the authors have contributed to make this study publishable. The author contribution is as follows: conceptualization, A. Ikhlaq and R. Javaid; methodology, U.Y. Qazi F. Javed and R. Iftikhar; validation, R. Javaid, U.Y. Qazi and I. Riaz; formal analysis, U.Y. Qazi, R. Nusrat; resources, A. Ikhlaq, and R. Jalil; writing—original draft preparation, U.Y. Qazi, and R. Iftikhar; writing—review and editing, A. Ikhlaq, R. Javaid and U.Y. Qazi; visualization, U.Y. Qazi and I. Riaz; supervision, A. Ikhlaq; project administration, U.Y. Qazi, A. Ikhlaq.; funding acquisition, U.Y. Qazi. All authors have read and agreed to the publication of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Ricardo A. Torres-Palma
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Qazi, U.Y., Iftikhar, R., Ikhlaq, A. et al. Application of Fe-RGO for the removal of dyes by catalytic ozonation process. Environ Sci Pollut Res 29, 89485–89497 (2022). https://doi.org/10.1007/s11356-022-21879-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21879-3