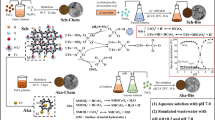
Abstract
Akaganéite (β-FeOOH) with a tunnel structure typically occupied by chloride can undergo anion-exchange reactions in aqueous solutions for pollutant removal. In this work, we studied bioformation of akaganéite in FeCl2 solutions with Acidithiobacillus ferrooxidans cells at pH 2.9, during 36-h incubation. The obtained products were analyzed and characterized by XRD, FTIR, EDS, FETEM, and HRTEM. Results showed that in acidic media with pH 2.9, the cells facilitated ferrous biooxidation and ferric precipitation. The resulting ferric precipitates were identified as polycrystalline akaganéite powders and had a morphology of nanospindles with a length of less 100 nm. The correlatively chemical formula for akaganéite collected at 1 h was reckoned as Fe8O8(OH)6.71(Cl)1.29 with 6.6% Cl. It was observed that ferric precipitates along exterior structures of cells or their extruded organic polymers grew and assembled into cellular shape. The evolved cell-shape akaganéite assemblages were twice of cells (about 2 µm) in size. These results could contribute to understanding of laboratorial bioformation of akaganéite and its biomineralization in acidic environments and promoting its practical applications.






Similar content being viewed by others
Data availability
Not applicable.
References
Badawi AK, Ismail B, Baaloudj O et al (2022) Advanced wastewater treatment process using algal photo-bioreactor associated with dissolved-air flotation system: a pilot-scale demonstration. J Water Process Eng 46:102565. https://doi.org/10.1016/j.jwpe.2022.102565
Carter CJ, Dolska M, Owczarek A et al (2009) In vitro selection of RNA sequences capable of mediating the formation of iron oxide nanoparticles. J Mater Chem 19:8320–8326. https://doi.org/10.1039/B912423C
Chan CS, De Stasio G, Welch SA et al (2004) Microbial polysaccharides template assembly of nanocrystal fibers. Science 303:1656–1658. https://doi.org/10.1126/science.1092098
Chan CS, Fakra SC, Edwards DC et al (2009) Iron oxyhydroxide mineralization on microbial extracellular polysaccharides. Geochim Cosmochim Acta 73(13):3807–3818. https://doi.org/10.1016/j.gca.2009.02.036
Chan CS, Emerson D, Luther GW (2016) The role of microaerophilic Fe-oxidizing micro-organisms in producing banded iron formations. Geobiology 14(5):509–528. https://doi.org/10.1111/gbi.12192
Chen WK, Dai PQ, Hong CF et al (2018) One-step synthesis and assembly of spindle-shaped akaganéite nanoparticles via sonochemistry. CrystEngComm 20:2989–2995. https://doi.org/10.1039/C8CE00470F
Chung D, Park C, Choi W et al (2020) Structural refinement and density functional theory study of synthetic Ge-akaganéite (β-FeOOH). Crystals 10:1–13. https://doi.org/10.3390/cryst10040239
Feng K, Wang XM, Zhou B et al (2021) Hydroxyl, Fe2+, and Acidithiobacillus ferrooxidans jointly determined the crystal growth and morphology of schwertmannite in a sulfate-rich acidic environment. Acs Omea 6:3194–3201. https://doi.org/10.1021/acsomega.0c05606
Feng K, Wang XM, Ding BT et al (2022) Acidithiobacillus ferrooxidans mediates morphology evolution of schwertmannite in the presence of Fe2+. Chem Geol 598:1–8. https://doi.org/10.1016/j.chemgeo.2022.120828
Fortin D, Langley S (2005) Formation and occurrence of biogenic iron-rich minerals. Earth-Sci Rev 72:1–19. https://doi.org/10.1016/j.earscirev.2005.03.002
Fry IV, Lazaroff N, Packer L (1986) Sulfate-dependent iron oxidation by Thiobacillus ferrooxidans: characterization of a new EPR detectable electron transport component on the reducing side of rusticyanin. Arch Biochem Biophys 246:650–654. https://doi.org/10.1016/0003-9861(86)90321-8
Fu XH, Jia LC, Wang A et al (2020) Thermal stability of akaganéite and its desiccation process under conditions relevant to Mars. Icarus 336:1–11. https://doi.org/10.1016/j.icarus.2019.113435
Futterer S, Andrusenko I, Kolb U et al (2013) Structural characterization of iron oxide/hydroxide nanoparticles in nine different parenteral drugs for the treatment of iron deficiency anaemia by electron diffraction (ED) and X-ray powder diffraction (XRPD). J Pharmaceut Biomed 86:151–160. https://doi.org/10.1016/j.jpba.2013.08.005
Gehrke T, Telegdi J, Thierry D et al (1998) Importance of extracellular polymeric substances from Thiobacillus ferrooxidans for bioleaching. Appl Environ Microbiol 64:2743–2747. https://doi.org/10.1128/AEM.64.7.2743-2747.1998
Harijan DKL, Chandra V (2017) Akaganéite nanorods decorated graphene oxide sheets for removal and recovery of aqueous phosphate. J Water Process Eng 19:120–125. https://doi.org/10.1016/j.jwpe.2017.07.019
Huang ZX, Han FS, Li MT et al (2019) Which phase of iron oxyhydroxides (FeOOH) is more competent in overall water splitting as a photocatalyst, goethite, akaganéite or lepidocrocite-a DFT-based investigation. Comp Mater Sci 169:1–8. https://doi.org/10.1016/j.commatsci.2019.109110
JCPDS (Joint Committee on Powder Diffraction Standards) (2002) Mineral powder diffraction files. International center for diffraction data: Swarthmore, PA
Kang FF, Wu M, Xiao B et al (2020) Facile synthesis of schwertmannite@akaganéite core/shell nanostructure from pickling waste liquor: formation mechanism and potential application. J Clean Prod 260:1–11. https://doi.org/10.1016/j.jclepro.2020.120961
Karam A, Bakhoum ES, Zaher K (2021) Coagulation/flocculation process for textile mill effluent treatment: experimental and numerical perspectives. Int J Sustain Eng 14:983–995. https://doi.org/10.1080/19397038.2020.1842547
Lazaroff N, Sigal W, Wasserman A (1982) Iron oxidation and precipitation of ferric hydroxysulfates by resting Thiobacillus ferrooxidans cells. Appl Environ Microbiol 43:924–938. https://doi.org/10.1128/aem.43.4.924-938.1982
Liu LD, Chen WT, Kihara S et al (2021) Green synthesis of acute accent akaganéite (β-FeOOH) nanocomposites as peroxidase-mimics and application for discoloration of methylene blue. J Environ Manag 296:1–13. https://doi.org/10.1016/j.jenvman.2021.113163
Lizama HM, Suzuki I (1988) Bacterial leaching of a sulfide ore by Thiobacillus ferrooxidans and Thiobacillus thiooxidans: I. Shake flask studies. Biotechnol Bioeng 32:110–116. https://doi.org/10.1002/bit.260320116
Nguyen VD, Nguyen HTH, Vranova V et al (2021) Artificial neural network modeling for Congo red adsorption on microwave-synthesized akaganéite nanoparticles: optimization, kinetics, mechanism, and thermodynamics. Environ Sci Pollut Res 28:9133–9145. https://doi.org/10.1007/s11356-020-10633-2
Papper RA, Couperthwaite SJ, Millar GJ et al (2018) A novel akaganéite sorbent synthesized from waste red mud: application for treatment of arsenate in aqueous solutions. J Environ Chem Eng 7:6308–6316. https://doi.org/10.1016/j.jece.2018.09.036
Pepper RA, Couperthwaite SJ, Millar GJ (2018) Value adding red mud waste: Impact of red mud composition upon fluoride removal performance of synthesized akaganéite sorbents. J Environ Chem Eng 6:2063–2074. https://doi.org/10.1016/j.jece.2018.02.048
Peretyazhko TS, Ming DW, Morris RV et al (2021) Formation of Fe(III) (hydr)oxides from Fe(II) sulfides: implications for akaganéite detection on Mars. ACS Earth Space Chem 5:1934–1947. https://doi.org/10.1021/acsearthspacechem.1c00075
Peretyazhko TS, Ming DW, Rampe EB et al (2018) Effect of solution pH and chloride concentration on akaganéite precipitation: Implications for akaganéite formation on Mars. J Geophys Res-Planet 123:2211–2222. https://doi.org/10.1029/2018JE005630
Peretyazhko TS, Pan MJ, Ming DW et al (2019) Reaction of akaganéite with Mars-relevant anions. ACS Earth Space Chem 3:314–323. https://doi.org/10.1021/acsearthspacechem.8b00173
Peterson KM, Heaney PJ, Post JE et al (2018) Evolution in the structure of akaganéite and hematite during hydrothermal growth: an in situ synchrotron X-ray diffraction analysis. Powder Diffr 33:287–297. https://doi.org/10.1017/S0885715618000623
Schwermann U, Cornell RM (2000) Iron oxides in the laboratory: preparation and characterization. Wiley-VCH, Weinheim. https://doi.org/10.1002/9783527613229.fmatter
Shokry A, El Tahan A, Ibrahim H et al (2019) Polyaniline/akaganéite superparamagnetic nanocomposite for cadmium uptake from polluted water. Desalin Water Treat 171:205–215. https://doi.org/10.5004/dwt.2019.24835
Spiridonov VV, Antonova YA, Kusaya VS et al (2021) One-step low-temperature synthesis of akaganéite and maghemite magnetic nanoparticles. Russ Chem B 70:1675–1681. https://doi.org/10.1007/s11172-021-3269-1
Villacorta V, Garcia KE, Greneche JM et al (2019) Influences of As(V), Sb(III), and Hg(II) ions on the nucleation and growth of akagenéite. CrystEngComm 21:7155–7165. https://doi.org/10.1039/c9ce01345h
Villacorta V, Barrero CA, Turrion MB et al (2020) Removal of As3+, As5+, Sb3+, and Hg2+ ions from aqueous solutions by pure and co-precipitated akaganéite nanoparticles: adsorption kinetics studies. RSC Adv 10:42688–42698. https://doi.org/10.1039/d0ra08075f
Wang ML, Gao QH, Zhang ML et al (2020a) In-situ formation of durable akaganéite (β-FeOOH) nanorods on sulfonate-modified poly(ethylene terephthalate) fabric for dual-functional wastewater treatment. J Hazard Mater 386:1–10. https://doi.org/10.1016/j.jhazmat.2019.121647
Wang T, Zhang PC, Ni SJ et al (2020b) Synthesis of nano-akaganéite powder and its chromium adsorption behavior. Ferroelectrics 540:184–192. https://doi.org/10.1080/10584587.2018.1454747
Watkinson DE, Emmerson NJ (2017) The impact of aqueous washing on the ability of β-FeOOH to corrode iron. Environ Sci Pollut Res 24:2138–2149. https://doi.org/10.1007/s11356-016-6749-3
Xiong HX, Guo R (2011) Effects of Chloride acclimation on iron oxyhydroxides and cell morphology during cultivation of Acidithiobacillus ferrooxidans. Environ Sci Technol 45:235–240. https://doi.org/10.1021/es1019146
Xiong HX, Liao YH, Zhou LX (2008) Influence of chloride and sulfate on formation of akaganéite and schwertmannite through ferrous biooxidation by Acidithiobacillus ferrooxidans cells. Environ Sci Technol 42:8681–8696. https://doi.org/10.1021/es801646j
Xiong HX, Shen XY, Cui C et al (2020) Synthesis of nanospindle-akaganéite and its photocatalytic degradation for methyl orange. Water Sci Technol 82:481–491. https://doi.org/10.2166/wst.2020.348
Xu YQ, Yang M, Yao T et al (2014) Isolation, identification and arsenic-resistance of Acidithiobacillus ferrooxidans HX3 producing schwertmannite. J Environ Sci 26:1463–1470. https://doi.org/10.1016/j.jes.2014.05.012
Yan W, Guo W, Wang LY et al (2021) Extracellular polymeric substances from Shewanella oneidensis MR-1 biofilms mediate the transformation of ferrihydrite. Sci Total Environ 784:1–8. https://doi.org/10.1016/j.scitotenv.2021.147245
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support by National Natural Science Foundations of China [41472034] and Natural Science General Fund of Jiangsu Province [BK20191444] for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
Huixin Xiong: conceptualization, methodology, validation, supervision, funding acquisition, writing—review and editing. Suning Peng: investigation, visualization, formal analysis, writing—original draft. Bailin Zhang: investigation, methodology, validation.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to publication
All the authors agreed to be published.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible editor: George Z. Kyzas
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xiong, H., Peng, S. & Zhang, B. Cell-shape assemblage and nanostructure of akaganéite bioformed in FeCl2 solutions. Environ Sci Pollut Res 29, 75566–75574 (2022). https://doi.org/10.1007/s11356-022-21084-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21084-2