Abstract
Many materials use polybrominated diphenyl ethers (PBDEs) as flame retardants. As one of the most common congeners of PBDEs, decabromodiphenyl ether (PBDE-209) is reported to harm reproductive health. However, little is known research on attenuating the reproductive toxicity induced by PBDE-209. The present study aimed to investigate the effects of hesperidin against PBDE-209-induced reproductive toxicity in male mice. Pubertal male C57BL/6 J mice were exposed to PBDE-209 groups (20, 100, 500 mg/kg·bw) and hesperidin groups (100 mg/kg·bw PBDE-209 + 100 mg/kg·bw hesperidin) for 8 weeks. The results showed that PBDE-209 increased the amount of abnormal morphological sperms and decreased the sex hormone levels. PBDE-209 induced the histopathological lesions of seminiferous tubules and blood-testis barrier in mice testis. Expressions of apoptosis-associated proteins and mRNA (Bax, Bcl-2, etc.) were altered by the PBDE-209 treatment. PBDE-209 prominently increased the malondialdehyde (MDA) levels, the biomarker of oxidative stress. Hesperidin treatment partly alleviated PBDE-209-induced histopathological lesions and apoptosis in mice testis. These findings suggested that hesperidin partly protects against PBDE-induced reproductive toxicity in pubertal mice. We conclude that more work needs to be done to explore the appropriate dosage of hesperidin or find other drugs to protect against the reproductive toxicity of PBDEs.
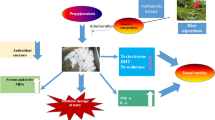
Graphical abstract







Similar content being viewed by others
Data availability
All data during this study are included in the manuscript, tables, and figures.
Abbreviations
- ATF6:
-
Activating transcription factor 6
- Bax:
-
BCL-2-associated X protein
- Bcl-2:
-
B-cell lymphoma-2
- BTB:
-
Blood-testis barrier
- ER:
-
Endoplasmic reticulum
- GSH:
-
Glutathione
- H&E:
-
Hematoxylin and eosin
- HES:
-
Hesperidin
- IHC:
-
Immunohistochemistry
- IRE1:
-
Inositol-requiring enzyme 1
- MDA:
-
Malondialdehyde
- PARP-1:
-
Poly(ADP-Ribose) polymerase-1
- PBDEs:
-
Polybrominated diphenyl ethers
- PBDE-209:
-
Decabromodiphenyl ether
- PCR:
-
Polymerase chain reaction
- TEM:
-
Transmission electron microscopy
- TUNEL:
-
Terminal deoxynucleotidyl transferase (TdT) mediated dUTP nick end labeling
- ZO-1:
-
Zonula occludens-1
References
Ahmadi A, Shadboorestan A, Nabavi SF, Setzer WN, Nabavi SM (2015) The role of hesperidin in cell signal transduction pathway for the prevention or treatment of cancer. Curr Med Chem 22:3462–3471. https://doi.org/10.2174/092986732230151019103810
Alharbi OM, Khattab RA, Ali I (2018) Health and environmental effects of persistent organic pollutants. J Mol Liquids 263:442–453. https://doi.org/10.1016/j.molliq.2018.05.029
Arafa HM, Aly HA, Abd-Ellah MF, El-Refaey HM (2009) Hesperidin attenuates benzo[alpha] pyrene-induced testicular toxicity in rats via regulation of oxidant/antioxidant balance. Toxicol Ind Health 25:417–427. https://doi.org/10.1177/0748233709106624
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Vandesompele J, Wittwer CT (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622. https://doi.org/10.1373/clinchem.2008.112797
Cardiff RD, Miller CH, Munn RJ (2014) Manual hematoxylin and eosin staining of mouse tissue sections. Cold Spring Harb Protoc 2014:655–658. https://doi.org/10.1101/pdb.prot073411
Chen Y, Kayano T, Takagi M (2000) Dysregulated expression of bcl-2 and bax in oral carcinomas: evidence of post-transcriptional control. J Oral Pathol Med Off Publ Int Assoc Oral Pathol Am Acad Oral Pathol 29:63–69. https://doi.org/10.1034/j.1600-0714.2000.290203.x
Costa LG, Giordano G, Tagliaferri S, Caglieri A, Mutti A (2008) Polybrominated diphenyl ether (PBDE) flame retardants: environmental contamination, human body burden and potential adverse health effects. Acta Biomed 79:172–183
Danis RB, Samplaski MK (2019) Sperm morphology: history, challenges, and impact on natural and assisted fertility. Curr Urol Rep 20:43. https://doi.org/10.1007/s11934-019-0911-7
FDA (2005) Guidance for industry: estimating the maximum safe starting dose in initial clinical trials for therapeutics in adult healthy volunteers. Cent Drug Eval Res
García-Vázquez FA, Gadea J, Matás C, Holt WV (2016) Importance of sperm morphology during sperm transport and fertilization in mammals. Asian J Androl 18:844–850. https://doi.org/10.4103/1008-682x.186880
Haddad JJ (2004) Redox and oxidant-mediated regulation of apoptosis signaling pathways: immuno-pharmaco-redox conception of oxidative siege versus cell death commitment. Int Immunopharmacol 4:475–493. https://doi.org/10.1016/j.intimp.2004.02.002
Herbstman JB, Sjödin A, Kurzon M, Lederman SA, Jones RS, Rauh V, Needham LL, Tang D, Niedzwiecki M, Wang RY, Perera F (2010) Prenatal exposure to PBDEs and neurodevelopment. Environ Health Perspect 118:712–719. https://doi.org/10.1289/ehp.0901340
Hsu P-C, Li Z-K, Lai C-S, Tseng L-H, Lee C-W, Cheng F-J, Chang C-Y, Chen J-RJC (2021) Transgenerational effects of BDE-209 on male reproduction in F3 offspring rats. Chemosphere 272:129829. https://doi.org/10.1016/j.chemosphere.2021.129829
Jacobson MH, Barr DB, Marcus M, Muir AB, Lyles RH, Howards PP, Pardo L, Darrow LA (2016) Serum polybrominated diphenyl ether concentrations and thyroid function in young children. Environ Res 149:222–230. https://doi.org/10.1016/j.envres.2016.05.022
Kaya K, Ciftci O, Cetin A, Doğan H, Başak N (2015) Hesperidin protects testicular and spermatological damages induced by cisplatin in rats. Andrologia 47:793–800. https://doi.org/10.1111/and.12332
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG (2010) Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol 8:e1000412. https://doi.org/10.1371/journal.pbio.1000412
Klincic D, Dvorscak M, Jagic K, Mendas G, Herceg Romanic S (2020) Levels and distribution of polybrominated diphenyl ethers in humans and environmental compartments: a comprehensive review of the last five years of research. Environ Sci Pollut Res Int 27:5744–5758. https://doi.org/10.1007/s11356-020-07598-7
Li C, Schluesener H (2017) Health-promoting effects of the citrus flavanone hesperidin. Crit Rev Food Sci Nutr 57:613–631. https://doi.org/10.1080/10408398.2014.906382
Li W, Zhu L, Zha J, Wang Z (2014) Effects of decabromodiphenyl ether (BDE-209) on mRNA transcription of thyroid hormone pathway and spermatogenesis associated genes in Chinese rare minnow (Gobiocypris rarus). Environ Toxicol 29:1–9. https://doi.org/10.1002/tox.20767
Li C, Zug C, Qu H, Schluesener H, Zhang Z (2015) Hesperidin ameliorates behavioral impairments and neuropathology of transgenic APP/PS1 mice. Behav Brain Res 281:32–42. https://doi.org/10.1016/j.bbr.2014.12.012
Li X, Zhu Y, Zhang C, Liu J, Zhou G, Jing L, Shi Z, Sun Z, Zhou X (2019) BDE-209 induces male reproductive toxicity via cell cycle arrest and apoptosis mediated by DNA damage response signaling pathways. Environ Pollut 255:113097. https://doi.org/10.1016/j.envpol.2019.113097
Li X, Liu J, Zhou G, Sang Y, Zhang Y, Jing L, Shi Z, Zhou X, Sun Z (2021) BDE-209 and DBDPE induce male reproductive toxicity through telomere-related cell senescence and apoptosis in SD rat. Environ Int 146:106307. https://doi.org/10.1016/j.envint.2020.106307
Li J, Zhang W, Zhou P, Tong X, Guo D, Lin H (2022) Selenium deficiency induced apoptosis via mitochondrial pathway caused by Oxidative Stress in porcine gastric tissues. Res Vet Sci 144:142–148. https://doi.org/10.1016/j.rvsc.2021.10.017
Liang SX, Gao HX, Zhao YY, Ma XM, Sun HW (2010) Effects of repeated exposure to decabrominated diphenyl ether (BDE-209) on mice nervous system and its self repair. Environ Toxicol Pharmacol 29:297–301. https://doi.org/10.1016/j.etap.2010.02.005
Lie PP, Cheng CY, Mruk DD (2013) Signalling pathways regulating the blood-testis barrier. Int J Biochem Cell Biol 45:621–625. https://doi.org/10.1016/j.biocel.2012.12.009
Linares V, Bellés M, Domingo JL (2015) Human exposure to PBDE and critical evaluation of health hazards. Arch Toxicol 89:335–356. https://doi.org/10.1007/s00204-015-1457-1
Liu JB, Li ZF, Lu L, Wang ZY, Wang L (2022) Glyphosate damages blood-testis barrier via NOX1-triggered oxidative stress in rats: long-term exposure as a potential risk for male reproductive health. Environ Int 159:107038. https://doi.org/10.1016/j.envint.2021.107038
Marcon L, Boissonneault G (2004) Transient DNA strand breaks during mouse and human spermiogenesis new insights in stage specificity and link to chromatin remodeling. Biol Reprod 70:910–918. https://doi.org/10.1095/biolreprod.103.022541
Mruk DD, Cheng CY (2004) Sertoli-Sertoli and Sertoli-germ cell interactions and their significance in germ cell movement in the seminiferous epithelium during spermatogenesis. Endocr Rev 25:747–806. https://doi.org/10.1210/er.2003-0022
Nakamura D, Yanagiba Y, Duan Z, Ito Y, Okamura A, Asaeda N, Tagawa Y, Li C, Taya K, Zhang SY, Naito H, Ramdhan DH, Kamijima M, Nakajima T (2010) Bisphenol A may cause testosterone reduction by adversely affecting both testis and pituitary systems similar to estradiol. Toxicol Lett 194:16–25. https://doi.org/10.1016/j.toxlet.2010.02.002
Opris R, Toma V, Olteanu D, Baldea I, Baciu AM, Lucaci FI, Berghian-Sevastre A, Tatomir C, Moldovan B, Clichici S, David L, Florea A, Filip GA (2019) Effects of silver nanoparticles functionalized with Cornus mas L. extract on architecture and apoptosis in rat testicle. Nanomedicine (lond) 14:275–299. https://doi.org/10.2217/nnm-2018-0193
Pawlicki P, Hejmej A, Milon A, Lustofin K, Płachno BJ, Tworzydlo W, Gorowska-Wojtowicz E, Pawlicka B, Kotula-Balak M, Bilinska B (2019) Telocytes in the mouse testicular interstitium: implications of G-protein-coupled estrogen receptor (GPER) and estrogen-related receptor (ERR) in the regulation of mouse testicular interstitial cells. Protoplasma 256:393–408. https://doi.org/10.1007/s00709-018-1305-2
Rahman F, Langford KH, Scrimshaw MD, Lester JN (2001) Polybrominated diphenyl ether (PBDE) flame retardants. Sci Total Environ 275:1–17. https://doi.org/10.1016/s0048-9697(01)00852-x
Rani L, Thapa K, Kanojia N, Sharma N, Singh S, Grewal AS, Srivastav AL, Kaushal JJJoCP (2020) An extensive review on the consequences of chemical pesticides on human health and environment. 124657. https://doi.org/10.1016/j.jclepro.2020.124657
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochim Biophys Acta 1863:2977–2992. https://doi.org/10.1016/j.bbamcr.2016.09.012
Rehman K, Fatima F, Waheed I, Akash MSH (2018) Prevalence of exposure of heavy metals and their impact on health consequences. J Cell Biochem 119:157–184. https://doi.org/10.1002/jcb.26234
Sarkar D, Joshi D, Singh SK (2019) Maternal BDE-209 exposure during lactation causes testicular and epididymal toxicity through increased oxidative stress in peripubertal mice offspring. Toxicol Lett 311:66–79. https://doi.org/10.1016/j.toxlet.2019.04.028
Schecter A, Päpke O, Harris TR, Tung KC, Musumba A, Olson J, Birnbaum L (2006) Polybrominated diphenyl ether (PBDE) levels in an expanded market basket survey of U.S. food and estimated PBDE dietary intake by age and sex. Environ Health Perspect 114:1515–1520. https://doi.org/10.1289/ehp.9121
Sellers RS, Morton D, Michael B, Roome N, Johnson JK, Yano BL, Perry R, Schafer K (2007) Society of Toxicologic Pathology position paper: organ weight recommendations for toxicology studies. Toxicol Pathol 35:751–755. https://doi.org/10.1080/01926230701595300
Sharkey M, Harrad S, Abou-Elwafa Abdallah M, Drage DS, Berresheim H (2020) Phasing-out of legacy brominated flame retardants: the UNEP Stockholm Convention and other legislative action worldwide. Environ Int 144:106041. https://doi.org/10.1016/j.envint.2020.106041
Song S, Tan J, Miao Y, Li M, Zhang Q (2017) Crosstalk of autophagy and apoptosis: involvement of the dual role of autophagy under ER stress. J Cell Physiol 232:2977–2984. https://doi.org/10.1002/jcp.25785
Squadrito F, Micali A, Rinaldi M, Irrera N, Marini H, Puzzolo D, Pisani A, Lorenzini C, Valenti A, Laurà R, Germanà A, Bitto A, Pizzino G, Pallio G, Altavilla D, Minutoli L (2016) Polydeoxyribonucleotide, an Adenosine-A2(A) Receptor agonist, preserves blood testis barrier from cadmium-induced injury. Front Pharmacol 7:537. https://doi.org/10.3389/fphar.2016.00537
Tait SW, Green DR (2010) Mitochondria and cell death: outer membrane permeabilization and beyond. Nat Rev Mol Cell Biol 11:621–632. https://doi.org/10.1038/nrm2952
Tang Z, Li Y, Jiang Y, Cheng J, Xu S, Zhang J (2019) Cellular metabolomics reveals glutamate and pyrimidine metabolism pathway alterations induced by BDE-47 in human neuroblastoma SK-N-SH cells. Ecotoxicol Environ Saf 182:109427. https://doi.org/10.1016/j.ecoenv.2019.109427
Trivedi PP, Tripathi DN, Jena GB (2011) Hesperetin protects testicular toxicity of doxorubicin in rat: role of NFκB, p38 and caspase-3. Food Chem Toxicol 49:838–847. https://doi.org/10.1016/j.fct.2010.12.005
UNEP (2017) Report of the Conference of the Parties to the Stockholm Convention on Persistent Organic Pollutants on the work of its eighth meeting. http://chm.pops.int/TheConvention/ConferenceoftheParties/Meetings/COP8/tabid/5309/Default.aspx
USEPA (2008) 2,2’,3,3’,4,4’,5,5’,6,6’-Decabromodiphenyl ether (BDE-209); CASRN 1163–19-5. Environmental Protection Agency, U.S
Vijaya Bharathi B, Jaya Prakash G, Krishna KM, Ravi Krishna CH, Sivanarayana T, Madan K, Rama Raju GA, Annapurna A (2015) Protective effect of alpha glucosyl hesperidin (G-hesperidin) on chronic vanadium induced testicular toxicity and sperm nuclear DNA damage in male Sprague Dawley rats. Andrologia 47:568–578. https://doi.org/10.1111/and.12304
Viswanatha GL, Shylaja H, Sandeep Rao KS, Santhosh Kumar VR, Jagadeesh M (2012) Hesperidin ameliorates immobilization-stress-induced behavioral and biochemical alterations and mitochondrial dysfunction in mice by modulating nitrergic pathway. ISRN Pharmacology 2012:479570. https://doi.org/10.5402/2012/479570
Vogel C, Marcotte EM (2012) Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat Rev Genet 13:227–232. https://doi.org/10.1038/nrg3185
Vonderheide AP, Mueller KE, Meija J, Welsh GL (2008) Polybrominated diphenyl ethers: causes for concern and knowledge gaps regarding environmental distribution, fate and toxicity. Sci Total Environ 400:425–436. https://doi.org/10.1016/j.scitotenv.2008.05.003
Xu L, Gao S, Zhao H, Wang L, Cao Y, Xi J, Zhang X, Dong X, Luan Y (2021) Integrated proteomic and metabolomic analysis of the testes characterizes BDE-47-induced reproductive toxicity in mice. Biomolecules 11. https://doi.org/10.3390/biom11060821
Yu L, Liu C, Chen Q, Zhou B (2014) Endocrine disruption and reproduction impairment in zebrafish after long-term exposure to DE-71. Environ Toxicol Chem 33:1354–1362. https://doi.org/10.1002/etc.2562
Zhang Y, Li X, Jing L, Zhou G, Sang Y, Gao L, Jiang S, Shi Z, Ge W, Sun Z, Zhou X (2021) Decabromodiphenyl ether induces male reproductive toxicity by activating mitochondrial apoptotic pathway through glycolipid metabolism dysbiosis. Chemosphere 285:131512. https://doi.org/10.1016/j.chemosphere.2021.131512
Zhang Y, Xi B, Tan W (2021) Release, transformation, and risk factors of polybrominated diphenyl ethers from landfills to the surrounding environments: a review. Environ Int 157:106780. https://doi.org/10.1016/j.envint.2021.106780
Zirkin BR, Papadopoulos V (2018) Leydig cells: formation, function, and regulation. Biol Reprod 99:101–111. https://doi.org/10.1093/biolre/ioy059
Acknowledgements
We express our great thanks to the editor(s) and reviewers that will/have spend time to guide toward a successful publication.
Funding
This work was supported by the National Key R&D Program of China (2017YFC1600302).
Author information
Authors and Affiliations
Contributions
Shiqi Li contributed to the experiments and data analysis and prepared the manuscript draft. Siyan Che and Sunni Chen performed the experiments. Zheng Ruan contributed to the manuscript’s conception, design, revision, and finalization. Li Zhang contributed to editing and supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The Animal Experiment Ethics Committee of Nanchang University (license No. SYXK (GAN) 2015–0001) approved the animal experiment.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. PBDE-209 induced oxidative stress and apoptosis in mice testes.
2. PBDE-209 induced histopathologic changes in seminiferous tubules of mice.
3. PBDE-209 disrupted the blood-testis barrier integrity in mice testes.
4. Hesperidin attenuated PBDE-209-induced reproductive toxicity in mice to some extent.
Rights and permissions
About this article
Cite this article
Li, S., Che, S., Chen, S. et al. Hesperidin partly ameliorates the decabromodiphenyl ether-induced reproductive toxicity in pubertal mice. Environ Sci Pollut Res 29, 90391–90403 (2022). https://doi.org/10.1007/s11356-022-20944-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-20944-1