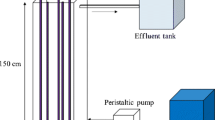
Abstract
Phenol is a widely used synthetic organic compound, which according to global estimations, is discharged into the environment at a rate of 10 tons/year through industrial waste. Phenol is a recalcitrant pollutant, and human exposure to water containing phenolic substances can lead to health issues. It has been found both in drinking water and wastewater. Solar heterogeneous photocatalytic phenol degradation, measured through chemical oxygen demand, was performed on a thin film tilted plate reactor with TiO2 immobilized onto different support materials. A full factorial experimental design (4 × 3 × 3) was carried out to statistically evaluate if the independent variables’ effects were significant. Four advanced oxidation processes (photolysis, photolysis + H2O2, heterogeneous photocatalysis, and heterogeneous photocatalysis + H2O2), three support materials (quartz, calcite, and glass), and three pH levels (3, 5.4, and 9) were evaluated. Reaction kinetics were fitted to the pseudo-first-order reaction rate and data was analyzed with an ANCOVA and means test, considering solar light intensity as a covariate. Photolysis/calcite at pH 5.4 and heterogeneous photocatalysis + H2O2/glass plate at pH 3 gave the best results, with a reaction rate constant kph = 3.047 × 10−3 min−1 and kphC = 4.498 × 10−3 min−1, respectively. The three independent variables and their interactions had a significant effect in the phenol degradation (p < 0.05).




Similar content being viewed by others
Data availability
All data and materials have been provided within the manuscript.
References
Adishkumar S, Kanmani S (2010) Treatment of phenolic wastewaters in single baffle reactor by solar/TiO2/H2O2 process. Desalin Water Treat 24:67–73. https://doi.org/10.5004/dwt.2010.1183
Alrousan DMA, Dunlop PSM, McMurray TA, Byrne JA (2009) Photocatalytic inactivation of E. coli in surface water using immobilised nanoparticle TiO2 films. Water Res 43:47–54. https://doi.org/10.1016/j.watres.2008.10.015
Alshabib M, Onaizi SA (2019) A review on phenolic wastewater remediation using homogeneous and heterogeneous enzymatic processes: current status and potential challenges. Sep Purif Technol 219:186–207. https://doi.org/10.1016/j.seppur.2019.03.028
Asencios YJO, Lourenço VS, Carvalho WA (2022) Removal of phenol in seawater by heterogeneous photocatalysis using activated carbon materials modified with TiO2. Catal Today 388–389:247–258. https://doi.org/10.1016/j.cattod.2020.06.064
Ataman E, Andersson MP, Ceccato M et al (2016) Functional group adsorption on calcite: I. Oxygen containing and nonpolar organic molecules. J Phys Chem C 120:16586–16596. https://doi.org/10.1021/acs.jpcc.6b01349
Azizpour F, Qaderi F (2019) Optimization, modeling and uncertainty investigation of phenolic wastewater treatment by photocatalytic process in cascade reactor. Environ Dev Sustain. https://doi.org/10.1007/s10668-019-00480-8
Bali U, Çatalkaya EÇ, Şengül F (2003) Photochemical degradation and mineralization of phenol: a comparative study. J Environ Sci Heal Part A 38:2259–2275. https://doi.org/10.1081/ESE-120023373
Belhouchet N, Hamdi B, Chenchouni H, Bessekhouad Y (2019) Photocatalytic degradation of tetracycline antibiotic using new calcite/titania nanocomposites. J Photochem Photobiol A Chem 372:196–205. https://doi.org/10.1016/j.jphotochem.2018.12.016
Bell S, Will G, Bell J (2013) Light intensity effects on photocatalytic water splitting with a titania catalyst. Int J Hydrogen Energy 38:6938–6947. https://doi.org/10.1016/j.ijhydene.2013.02.147
Burrows K, Fthenakis V (2015) Glass needs for a growing photovoltaics industry. Sol Energy Mater Sol Cells 132:455–459. https://doi.org/10.1016/j.solmat.2014.09.028
Cámara RM, Portela R, Gutiérrez-Martín F, Sánchez B (2016) Photocatalytic activity of TiO2 films prepared by surfactant-mediated sol-gel methods over commercial polymer substrates. Chem Eng J 283:535–543. https://doi.org/10.1016/j.cej.2015.07.080
Chatzisymeon E, Fierro S, Karafyllis I et al (2010) Anodic oxidation of phenol on Ti/IrO2 electrode: experimental studies. Catal Today 151:185–189. https://doi.org/10.1016/j.cattod.2010.02.076
Colmenares JC (2019) Selective redox photocatalysis: is there any chance for solar bio-refineries? Curr Opin Green Sustain Chem 15:38–46. https://doi.org/10.1016/j.cogsc.2018.08.008
Hach Company (1999) Hach methods approved/accepted by the USEPA. Hach- Downloads - Hach Methods EPA Acceptance Letters. 1999. https://www.hach.com/cms/documents/pdf/EPA/HachMethodsapprovedacceptedbytheUSEPA.pdf. Accessed 6 Mar 2021
Cui W, Li J, Chen L et al (2020) Nature-inspired CaCO3 loading TiO2 composites for efficient and durable photocatalytic mineralization of gaseous toluene. Sci Bull 65:1626–1634. https://doi.org/10.1016/j.scib.2020.05.024
Dadsetan S, Siad H, Lachemi M, Sahmaran M (2021) Evaluation of the tridymite formation as a technique for enhancing geopolymer binders based on glass waste. J Clean Prod 278:123983. https://doi.org/10.1016/j.jclepro.2020.123983
El Gaidoumi A, Arrahli A, Loqman A et al (2021) Efficient sol-gel nanocomposite TiO2-clay in photodegradation of phenol: comparison to labe-made and commercial photocatalysts. SILICON. https://doi.org/10.1007/s12633-021-01275-1
Eskandarian MR, Choi H, Fazli M, Rasoulifard MH (2016) Effect of UV-LED wavelengths on direct photolytic and TiO2 photocatalytic degradation of emerging contaminants in water. Chem Eng J 300:414–422. https://doi.org/10.1016/j.cej.2016.05.049
Giménez J, Curcó D, Queral M (1999) Photocatalytic treatment of phenol and 2,4-dichlorophenol in a solar plant in the way to scaling-up. Catal Today 54:229–243. https://doi.org/10.1016/S0920-5861(99)00185-6
De la Hoz F, Rivera D, Arumí JL, Chávez F (2009) Towards in-channel irrigation water disinfection using solar photocatalysis. Appl Eng Agric 25:685–692. https://doi.org/10.13031/2013.28858
Humayun M, Raziq F, Khan A, Luo W (2018) Modification strategies of TiO2 for potential applications in photocatalysis: a critical review. Green Chem Lett Rev 11:86–102. https://doi.org/10.1080/17518253.2018.1440324
Hussain A, Dubey SK, Kumar V (2015) Kinetic study for aerobic treatment of phenolic wastewater. Water Resour Ind 11:81–90. https://doi.org/10.1016/j.wri.2015.05.002
Irigoyen-Campuzano R, González-Béjar M, Pino E et al (2017) A metal-free, nonconjugated polymer for solar photocatalysis. Chem - A Eur J 23:2867–2876. https://doi.org/10.1002/chem.201605064
Jada A, Ait Akbour R, Douch J (2006) Surface charge and adsorption from water onto quartz sand of humic acid. Chemosphere 64:1287–1295. https://doi.org/10.1016/j.chemosphere.2005.12.063
Jay L, Chirwa EMN (2019) The effect of reaction conditions on the degradation of phenol by UV/TiO2 photocatalysis. Chem Eng Trans 76:847–852. https://doi.org/10.3303/CET1976142
Kanakaraju D, Glass BD, Oelgemöller M (2018) Advanced oxidation process-mediated removal of pharmaceuticals from water: a review. J Environ Manage 219:189–207. https://doi.org/10.1016/j.jenvman.2018.04.103
Kiran S, Ali S, Asgher M (2013) Degradation and mineralization of azo dye reactive blue 222 by sequential photo-Fenton’s oxidation followed by aerobic biological treatment using white rot fungi. Bull Environ Contam Toxicol 90:208–215. https://doi.org/10.1007/s00128-012-0888-0
Lee E, Lee H, Kim YK et al (2011) Hydrogen peroxide interference in chemical oxygen demand during ozone based advanced oxidation of anaerobically digested livestock wastewater. Iran J Environ Heal Sci Eng 8:381–388
Li Y, Yang B, Liu B (2021) MOF assisted synthesis of TiO2/Au/Fe2O3 hybrids with enhanced photocatalytic hydrogen production and simultaneous removal of toxic phenolic compounds. J Mol Liq 322:114815. https://doi.org/10.1016/j.molliq.2020.114815
Litter MI (2015) Mechanisms of removal of heavy metals and arsenic from water by TiO2-heterogeneous photocatalysis. Pure Appl Chem 87:557–567. https://doi.org/10.1515/pac-2014-0710
Litter MI (2017) Last advances on TiO2-photocatalytic removal of chromium, uranium and arsenic. Curr Opin Green Sustain Chem 6:150–158. https://doi.org/10.1016/j.cogsc.2017.04.002
Liu G, Wang G, Hu Z et al (2019) Ag2O nanoparticles decorated TiO2 nanofibers as a p-n heterojunction for enhanced photocatalytic decomposition of RhB under visible light irradiation. Appl Surf Sci 465:902–910. https://doi.org/10.1016/j.apsusc.2018.09.216
Malakootian M, Heidari MR (2018) Removal of phenol from steel wastewater by combined electrocoagulation with photo-Fenton. Water Sci Technol 78:1260–1267. https://doi.org/10.2166/wst.2018.376
Malato S, Fernández-Ibáñez P, Maldonado MI et al (2009) Decontamination and disinfection of water by solar photocatalysis: recent overview and trends. Catal Today 147:1–59. https://doi.org/10.1016/j.cattod.2009.06.018
Mino L, Zecchina A, Martra G et al (2016) A surface science approach to TiO2 P25 photocatalysis: an in situ FTIR study of phenol photodegradation at controlled water coverages from sub-monolayer to multilayer. Appl Catal B Environ 196:135–141. https://doi.org/10.1016/j.apcatb.2016.05.029
Mishra A, Mehta A, Basu S (2018) Clay supported TiO2 nanoparticles for photocatalytic degradation of environmental pollutants: a review. J Environ Chem Eng 6:6088–6107. https://doi.org/10.1016/j.jece.2018.09.029
Morales-Mejía JC, Almanza R, Gutiérrez F (2014) Solar photocatalytic oxidation of hydroxy phenols in a CPC reactor with thick TiO2 films. Energy Procedia 57:597–606. https://doi.org/10.1016/j.egypro.2014.10.214
Mozia S, Brożek P, Przepiórski J et al (2012) Immobilized TiO2 for phenol degradation in a pilot-scale photocatalytic reactor. J Nanomater 2012:1–10. https://doi.org/10.1155/2012/949764
Muhammad-Sukki F, Ramirez-Iniguez R, McMeekin SG et al (2010) Solar concentrators. Int. J Appl Sci 1:1–15
Nguyen AT, Juang RS (2015) Photocatalytic degradation of p-chlorophenol by hybrid H2O2 and TiO2 in aqueous suspensions under UV irradiation. J Environ Manage 147:271–277. https://doi.org/10.1016/j.jenvman.2014.08.023
Nidheesh PV, Zhou M, Oturan MA (2018) An overview on the removal of synthetic dyes from water by electrochemical advanced oxidation processes. Chemosphere 197:210–227. https://doi.org/10.1016/j.chemosphere.2017.12.195
Ola O, Maroto-Valer MM (2015) Review of material design and reactor engineering on TiO2 photocatalysis for CO2 reduction. J Photochem Photobiol C Photochem Rev 24:16–42. https://doi.org/10.1016/j.jphotochemrev.2015.06.001
Pan Y, Wang Y, Li A et al (2017) Detection, formation and occurrence of 13 new polar phenolic chlorinated and brominated disinfection byproducts in drinking water. Water Res 112:129–136. https://doi.org/10.1016/j.watres.2017.01.037
Ren T, Jin Z, Yang J et al (2019) Highly efficient and stable p-LaFeO3/n-ZnO heterojunction photocatalyst for phenol degradation under visible light irradiation. J Hazard Mater 377:195–205. https://doi.org/10.1016/j.jhazmat.2019.05.070
Saroj DP, Kumar A, Bose P et al (2005) Mineralization of some natural refractory organic compounds by biodegradation and ozonation. Water Res 39:1921–1933. https://doi.org/10.1016/j.watres.2005.03.020
Selvam R, Muniraj S, Duraisamy T, Muthunarayanan V (2018) Identification of disinfection by-products (DBPs) halo phenols in drinking water. Appl Water Sci 8:1–8. https://doi.org/10.1007/s13201-018-0771-1
Sharma A, Ahmad J, Flora SJS (2018) Application of advanced oxidation processes and toxicity assessment of transformation products. Environ Res 167:223–233. https://doi.org/10.1016/j.envres.2018.07.010
Silva CG, Faria JL (2009) Effect of key operational parameters on the photocatalytic oxidation of phenol by nanocrystalline sol-gel TiO2 under UV irradiation. J Mol Catal A Chem 305:147–154. https://doi.org/10.1016/j.molcata.2008.12.015
Singh S, Mishra R, Sharma RS, Mishra V (2017) Phenol remediation by peroxidase from an invasive mesquite: turning an environmental wound into wisdom. J Hazard Mater 334:201–211. https://doi.org/10.1016/j.jhazmat.2017.04.007
Srikanth B, Goutham R, Badri Narayan R et al (2017) Recent advancements in supporting materials for immobilised photocatalytic applications in waste water treatment. J Environ Manage 200:60–78. https://doi.org/10.1016/j.jenvman.2017.05.063
Vilhunen S, Puton J, Virkutyte J, Sillanpää M (2011) Efficiency of hydroxyl radical formation and phenol decomposition using UV light emitting diodes and H2O2. Environ Technol 32:865–872. https://doi.org/10.1080/09593330.2010.516770
Wang JL, Xu LJ (2012) Advanced oxidation processes for wastewater treatment: formation of hydroxyl radical and application. Crit Rev Environ Sci Technol 42:251–325. https://doi.org/10.1080/10643389.2010.507698
Wang N, Lei L, Zhang XM et al (2011) A comparative study of preparation methods of nanoporous TiO2 films for microfluidic photocatalysis. Microelectron Eng 88:2797–2799. https://doi.org/10.1016/j.mee.2010.12.051
Wang J, Sun S, Pan L et al (2019a) Preparation and properties of CaCO3-supported nano-TiO2 composite with improved photocatalytic performance. Materials (basel) 12:3369. https://doi.org/10.3390/ma12203369
Wang Y, Zhao J, Xiong X et al (2019b) Role of Ni2+ ions in TiO2 and Pt/TiO2 photocatalysis for phenol degradation in aqueous suspensions. Appl Catal B Environ 258:117903. https://doi.org/10.1016/j.apcatb.2019.117903
Wang H, Li X, Zhao X et al (2022) A review on heterogeneous photocatalysis for environmental remediation: from semiconductors to modification strategies. Chinese J Catal 43:178–214. https://doi.org/10.1016/S1872-2067(21)63910-4
Weber M, Weber M, Weber V (2020) Phenol. In: Ullmann’s encyclopedia of industrial chemistry. Wiley, pp 1–20
Wei TY, Wan CC (1991) Heterogeneous photocatalytic oxidation of phenol with titanium dioxide powders. Ind Eng Chem Res 30:1293–1300. https://doi.org/10.1021/ie00054a033
Xiang L, Xiang Y, Wen Y, Wei F (2004) Formation of CaCO3 nanoparticles in the presence of terpineol. Mater Lett 58:959–965. https://doi.org/10.1016/j.matlet.2003.07.034
Yaroshevsky AA (2006) Abundances of chemical elements in the Earth’s crust. Geochemistry Int 44:48–55. https://doi.org/10.1134/S001670290601006X
Ye J, Li X, Hong J et al (2015) Photocatalytic degradation of phenol over ZnO nanosheets immobilized on montmorillonite. Mater Sci Semicond Process 39:17–22. https://doi.org/10.1016/j.mssp.2015.04.039
Yohi S, Wu C, Koodali RT (2022) A kinetic study of photocatalytic degradation of phenol over titania–silica mixed oxide materials under UV illumination. Catalysts 12:193. https://doi.org/10.3390/catal12020193
Zhang C, Li Y, Shen H, Shuai D (2021) Simultaneous coupling of photocatalytic and biological processes: a promising synergistic alternative for enhancing decontamination of recalcitrant compounds in water. Chem Eng J 403:126365. https://doi.org/10.1016/j.cej.2020.126365
Zhong JB, Li JZ, Feng FM et al (2012) Improved photocatalytic performance of SiO2-TiO2 prepared with the assistance of SDBS. J Mol Catal A Chem 357:101–105. https://doi.org/10.1016/j.molcata.2012.01.026
Acknowledgements
The authors would like to thank CONACyT for the student scholarship, to Planta de Tratamiento de Aguas Residuales (PTAR) “Oriente” del Estado de Durango and Instituto Politécnico Nacional for its support to this project.
Author information
Authors and Affiliations
Contributions
Felipe Silerio-Vázquez contributed to the study conception and design, material preparation, data collection, data analysis, and first draft writing; María T. Alarcón-Herrera contributed to visualize, review, and edit the final draft; José Bernardo Proal-Nájera contributed to the study conception and design, visualize, review, and edit to the final draft, as well as with funding acquisition.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have consented the manuscript content for publication.
Conflict of interests
The authors declare no competing interests.
Additional information
Responsible Editor: Sami Rtimi
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Silerio-Vázquez, F., Alarcón-Herrera, M.T. & Proal-Nájera, J.B. Solar heterogeneous photocatalytic degradation of phenol on TiO2/quartz and TiO2/calcite: a statistical and kinetic approach on comparative efficiencies towards a TiO2/glass system. Environ Sci Pollut Res 29, 42319–42330 (2022). https://doi.org/10.1007/s11356-022-19379-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-19379-5