Abstract
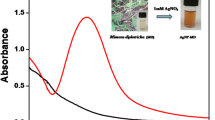
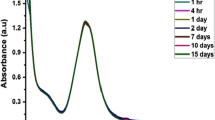
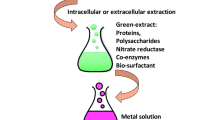
In this study, a simple, one-pot, and eco-friendly biosynthesis of silver nanoparticles (AgNPs) was accomplished with the use of aqueous leaves extract of Cestrum nocturnum L.(AECN). Different techniques like ultraviolet–visible (UV–Vis) spectrophotometry, Fourier transform infrared (FTIR), X-ray diffraction (XRD), transmission electron microscopy (TEM), and scanning area electron diffraction were used to investigate the optical, operational, and physical properties of the green synthesized AECN-AgNPs.The AECN-AgNPs were further used for the detection of Hg2+ by UV–Vis and electrochemical methods. The disintegration of the AECN-AgNPs solution caused the formation of an Ag-Hg amalgam, which caused discoloration of the solution. Sensing performance for a variety of metals such as Na+, K+, Mg2+, Ca2+, Ni2+, Cu 2+, Fe3+, Zn2+, Co2+, Cd2+, Pb2+, As3+, and Mn2+ at 10-mM concentrations was measured in order to determine the selectivity of the sensor towards the Hg2+. For the electrochemical determination of 2 + Hg2+ , AECN-AgNPs were immobilized on a glassy carbon (GC) electrode, and the resulting modified electrode (GC/AECN-AgNPs) was characterized by cyclic voltammetry. This phenomenon is advantageously used for the sensitive determination of trace level Hg2+. GC/AECN-AgNPs demonstrated a linear calibration range of 100 nM to 10 μM and a limit of detection of 21 nM for Hg2+ determination.





Similar content being viewed by others
Data availability
Data used in this study are freely available to the public from the described sources in the method section.
References
Afkhami A, Madrakian T, Sabounchei SJ, Rezaei M, Samiee S, Pourshahbaz M (2012) Construction of a modified carbon paste electrode for the highly selective simultaneous electrochemical determination of trace amounts of mercury(II) and cadmium(II). Sens Actuators B Chem. https://doi.org/10.1016/j.snb.2011.10.073
Agraz R, Sevilla MT, Hernandez L (1995) Voltammetric quantification and speciation of mercury compounds. J Electroanal Chem. https://doi.org/10.1016/0022-0728(95)03955-G
Al-Reza SM, Rahman A, Kang SC (2009) Chemical composition and inhibitory effect of essential oil and organic extracts of Cestrum nocturnum L. on food-borne pathogens. Int J Food Sci Technol. https://doi.org/10.1111/j.1365-2621.2009.01939.x
Aranda PR, Colombo L, Perino E, de Vito IE, Raba J (2013) Solid-phase preconcentration and determination of mercury(II) using activated carbon in drinking water by X-ray fluorescence spectrometry. X-Ray Spectrom. https://doi.org/10.1002/xrs.2440
Azevedo BF, Furieri LB, Peçanha FMI, Wiggers GA et al (2012) Toxic effects of mercury on the cardiovascular and central nervous systems. J Biomed Biotechnol. https://doi.org/10.1155/2012/949048
Bard AJ, Faulkner LR (1980) Electrochemical methods fundamentals and applications, 2nd edn. Wiley, New York. https://doi.org/10.1021/ed060pa25
Bhattacharjee Y, Chakraborty A (2014) Label-free cysteamine-capped silver nanoparticle-based colorimetric assay for Hg (II) detection in water with subnanomolar exactitude. ACS Sustain Chem Eng. https://doi.org/10.1021/sc500339n
Chang LW (1992) The concept of direct and indirect neurotoxicity and the concept of toxic metal/essential element interactions as a common biomechanism underlying metal toxicity. In: Isaacson RL, Jensen KF (eds) The vulnerable brain and environmental risks. Springer, Boston, pp 61–82. https://doi.org/10.1021/ac00153a011
Chen L, Fu X, Lu W, Chen L (2013) Highly sensitive and selective colorimetric sensing of Hg2+ based on the morphology transition of silver nanoprisms. ACS Appl Mater Interfaces. https://doi.org/10.1021/am3020857
Cinti S, Santella F, Moscone D, Arduini F (2016) Hg2+ detection using a disposable and miniaturized screen-printed electrode modified with nanocomposite carbon black and gold nanoparticles. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-016-6118-2
Coates J (2006) Interpretation of infrared spectra a practical approach. In: Meyers RA (ed) Encyclopedia of analytical chemistry. Wiley, Chichester. https://doi.org/10.1002/9780470027318.a5606
Ding Y, Wang S, Li J, Chen L (2016) Nanomaterial-based optical sensors for mercury ions. Trends Anal Chem. https://doi.org/10.1016/j.trac.2016.05.015
Fang Y, Pan Y, Li P, Xue M, Pei F, Yang W, Ma N, Hu Q (2016) Simultaneous determination of arsenic and mercury species in rice by ion-pairing reversed phase chromatography with inductively coupled plasma mass spectrometry. Food Chem. https://doi.org/10.1016/j.foodchem.2016.07.003
Goulart LA, Gonçalves R, Correa AA, Pereira EC, Mascaro LH (2018) Synergic effect of silver nanoparticles and carbon nanotubes on the simultaneous voltammetric determination of hydroquinone, catechol, bisphenol A and phenol. Microchim Acta. https://doi.org/10.1007/s00604-017-2540-5
Ioannidou MD, Zachariadis GA, Anthemidis AN, Stratis JA (2005) Direct determination of toxic trace metals in honey and sugars using inductively coupled plasma atomic emission spectrometry. Talanta. https://doi.org/10.1016/j.talanta.2004.05.018
Kumar V, Bano D, Mohan S, Singh DK, Hasan SH (2016a) Sunlight-induced green synthesis of silver nanoparticles using aqueous leaf extract of Polyalthia longifolia and its antioxidant activity. Mater Lett. https://doi.org/10.1016/j.matlet.2016.05.097
Kumar V, Singh DK, Mohan S, Hasan SH (2016b) Photo-induced biosynthesis of silver nanoparticles using aqueous extract of Erigeron bonariensis and its catalytic activity against Acridine Orange. J Photochem Photobiol B Biol. https://doi.org/10.1016/j.jphotobiol.2015.12.011
Kumar V, Singh DK, Mohan S, Bano D, Gundampati RK, Hasan SH (2017) Green synthesis of silver nanoparticle for the selective and sensitive colorimetric detection of mercury (II) ion. J Photochem Photobiol B Biol. https://doi.org/10.1016/J.JPHOTOBIOL.2017.01.022
Lin Y, Yang Y, Li Y, Yang L, Hou X, Feng X, Zheng C (2016) Ultrasensitive speciation analysis of mercury in rice by headspace solid phase microextraction using porous carbons and gas chromatography-dielectric barrier discharge optical emission spectrometry. Environ Sci Technol. https://doi.org/10.1021/acs.est.5b04328
Lou T, Chen Z, Wang Y, Chen L (2011) Blue-to-red colorimetric sensing strategy for Hg2+ and Ag+ via redox-regulated surface chemistry of gold nanoparticles. ACS Appl Mater. https://doi.org/10.1021/am200130e
Park JD, Zheng W (2012) Human exposure and health effects of inorganic and elemental mercury. J Prev Med Public Health. https://doi.org/10.3961/jpmph.2012.45.6.344
Qadir MI, al Murad MS, Ali M, Saleem M, Farooqi AA (2014) Hepatoprotective effect of leaves of aqueous ethanol extract of Cestrum nocturnum against paracetamol-induced hepatotoxicity. Bangladesh J Pharmacol. https://doi.org/10.3329/bjp.v9i2.17770
Rastogi PK, Ganesan V, Krishnamoorthi S (2014) A promising electrochemical sensing platform based on a silvernanoparticles decorated copolymer for sensitive nitrite determination. J Mater Chem A. https://doi.org/10.1039/C3TA13794E
Rey-Raap N, Gallardo A (2012) Determination of mercury distribution inside spent compact fluorescent lamps by atomic absorption spectrometry. Waste Manage. https://doi.org/10.1016/j.wasman.2011.12.001
Rice KM, Walker EM, Wu M, Gillette C, Blough ER (2014) Environmental mercury and its toxic effects. J Prev Med Public Health. https://doi.org/10.3961/jpmph.2014.47.2.74
Some IT, Sakira AK, Mertens D, Ronkart SN, Kauffmann JM (2016) Determination of groundwater mercury (II) content using a disposable gold modified screen printed carbon electrode. Talanta. https://doi.org/10.1016/j.talanta.2016.02.033
Sonkar PK, Ganesan V (2015) Synthesis and characterization of silver nanoparticle-anchored amine-functionalized mesoporous silica for electrocatalytic determination of nitrite. J Solid State Electrochem. https://doi.org/10.1007/s10008-014-2725-3
Sonkar PK, Ganesan V, Gupta R, Yadav DK (2016) Simple route to anchor silver nanoparticles into thiol-functionalized mesoporous silica: synthesis, characterization and electrochemical applications. J Nanoparticle Res. https://doi.org/10.1007/s11051-016-3607-7
Suaarez MF, Mills A, Egdell RG, Compton RG (2000) Anodic stripping voltammetry with photochemical preconcentration at nanocrystalline TiO2 films: detection of Ag and Hg2+. Electroanalysis. https://doi.org/10.1002/(SICI)1521-4109(20000401)12:63.3.CO;2-9
Suherman AL, Ngamchuea K, Tanner EE, Sokolov SV, Holter J, Young NP, Compton RG (2017) Electrochemical detection of ultratrace (picomolar) levels of Hg2+ using a silver nanoparticle-modified glassy carbon electrode. Anal Chem. https://doi.org/10.1021/acs.analchem.7b01304
Tang Y, Allen BL, Kauffman DR, Star A (2009) Electrocatalytic activity of nitrogen-doped carbon nano tube cups. J Am Chem Soc. https://doi.org/10.1021/ja904595t
Tang S, Tong P, Lu W, Chen J, Yan Z, Zhang L (2014) A novel label-free electrochemical sensor for Hg2+ based on the catalytic formation of metal nanoparticle. Biosens Bioelectron. https://doi.org/10.1016/j.bios.2014.02.074
Toh HS, Jurkschat K, Compton RG (2015) The influence of the capping agent on the oxidation of silver nanoparticles: nano-impacts versus stripping voltammetry. Chem Eur J 21:2998–3004. https://doi.org/10.1002/chem.201406278
Tyagi CK, Jhade D, Shah SK (2016) Evaluation of anticoagulant activity of aqueous extract of Cestrum nocturnum. Int J Phytomed. https://doi.org/10.5138/09750185.1969
Wang J, Bonakdar M (1988) Preconcentration and voltammetric measurement of mercury with a crown-ether modified-carbon-paste electrode. Talanta. https://doi.org/10.1016/0039-9140(88)80085-7
Wei Y, Yang R, Liu JH, Huang XJ (2013) Selective detection toward Hg (II) and Pb (II) using polypyrrole/carbonaceous nanospheres modified screen-printed electrode. Electrochim Acta. https://doi.org/10.1016/j.electacta.2013.05.004
Wu L, Long Z, Liu L, Zhou Q, Lee YI, Zheng C (2012) Microwave-enhanced cold vapor generation for speciation analysis of mercury by atomic fluorescence spectrometry. Talanta. https://doi.org/10.1016/j.talanta.2012.03.009
Zaib M, Athar MM, Saeed A, Farooq U (2015) Electrochemical determination of inorganic mercury and arsenic-a review. Biosens Bioelectron. https://doi.org/10.1016/j.bios.2015.07.058
Zheng W, Aschner M, Ghersi-Egea JF (2003) Brain barrier systems: a new frontier in metal neurotoxicological research. Toxicol Appl Pharmacol. https://doi.org/10.1016/S0041-008X(03)00251-5
Zhou N, Li J, Chen H, Liao C, Chen Z, Chen L (2013) A functional graphene oxide-ionic liquid composites/gold nanoparticles sensing platform for ultrasensitive electrochemical detection of Hg2+. Analyst. https://doi.org/10.1039/C2AN36405K
Funding
This paper was supported in part of grant from UGC (F-1–17-1/2014–15/RGNF-2014–15-SC-UTT-58260) to New Delhi, Government of India, and is gratefully acknowledged. PKS acknowledges the University Grant Commission, New Delhi, India (UGC No.F.30–431/2018 (BSR), M-14–58), and Institute of Eminence (IoE) Seed Grant, Banaras Hindu University, India (Scheme No.6031), for the financial support. Author Kavindra Nath Tiwari also acknowledges to IoE, Banaras Hindu University, Varanasi, India, for the financial support for the research work (Scheme No.6031).
Author information
Authors and Affiliations
Contributions
P Kumar: optimization of AECN-AgNPs synthesis. KN Tiwari, SK Mishra: supervision, methodology. PK Sonkar and V Ganesan: electrochemical detection and validation of Hg2+ sensing. AK Singh, J Singh, JDixit: plant material collection and extract preparation.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, P., Sonkar, P., Tiwari, K.N. et al. Sensing of mercury ion using light induced aqueous leaf extract mediated green synthesized silver nanoparticles of Cestrum nocturnum L. Environ Sci Pollut Res 29, 79995–80004 (2022). https://doi.org/10.1007/s11356-022-19357-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-19357-x