Abstract
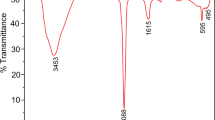
As persistent and ubiquitous contaminants in water, iodinated X-ray contrast media (ICM) pose a non-negligible risk to the environment and human health. In this study, we investigated the adsorption behavior of two typical ICM compounds, iohexol (IOH) and amidotrizoic acid (DTZ), on magnetic activated carbon. Theoretical investigations, using density functional theory, identified the molecule structures and calculated the molecular diameters of IOH (1.68 nm) and DTZ (1.16 nm), which revealed that ICM could be adsorbed by mesopores and larger micropores. Therefore, magnetic activated carbon with a porous structure was prepared by the co-precipitation method to investigate the adsorption mechanism of IOH and DTZ. MAC-5 (magnetic activated carbon with a theoretical iron oxide content of 37%) showed the best adsorption ability for both IOH and DTZ, with maximum adsorption capacities of 86.05 and 43.00 mg g−1, respectively. Adsorption kinetics and isotherm models were applied to explore the mechanisms involved, and the effects of solution pH, initial concentration, temperature, ionic strength, and natural organic matter were also investigated. The pore filling effect, π–π stacking, hydrogen bonding, and electrostatic interaction, were found to be the main adsorption mechanisms. The co-adsorption data showed that competition may occur in ICM coexisting environments. Interestingly, the used MAC-5 could be successfully regenerated and its adsorption efficiency did not decrease significantly after five cycles, indicating that it is a promising adsorbent for ICM. The results from this study provide some new insights for the treatment of water containing ICM.









Similar content being viewed by others
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aljeboree AM, Alshirifi AN, Alkaim AF (2017) Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arab J Chem 10:S3381–S3393. https://doi.org/10.1016/j.arabjc.2014.01.020
Arnnok P, Singh RR, Burakham R et al (2017) Selective uptake and bioaccumulation of antidepressants in fish from effluent-impacted Niagara River. Environ Sci Technol 51:10652–10662. https://doi.org/10.1021/acs.est.7b02912
Baghdadi M, Ghaffari E, Aminzadeh B (2016) Removal of carbamazepine from municipal wastewater effluent using optimally synthesized magnetic activated carbon: adsorption and sedimentation kinetic studies. J Environ Chem Eng 4:3309–3321. https://doi.org/10.1016/j.jece.2016.06.034
Bourin M, Jolliet P, Ballereau F (1997) An overview of the clinical pharmacokinetics of x-ray contrast media. Clin Pharmacokinet 32:180–193. https://doi.org/10.2165/00003088-199732030-00002
Cermakova L, Kopecka I, Pivokonsky M et al (2017) Removal of cyanobacterial amino acids in water treatment by activated carbon adsorption. Sep Purif Technol 173:330–338. https://doi.org/10.1016/j.seppur.2016.09.043
Dai J, Meng X, Zhang Y et al (2020) Effects of modification and magnetization of rice straw derived biochar on adsorption of tetracycline from water. Bioresour Technol 311:123455. https://doi.org/10.1016/j.biortech.2020.123455
De Gusseme B, Hennebel T, Vanhaecke L et al (2011) Biogenic palladium enhances diatrizoate removal from hospital wastewater in a microbial electrolysis cell. Environ Sci Technol 45:5737–5745. https://doi.org/10.1021/es200702m
Fabbri D, Calza P, Dalmasso D et al (2016) Iodinated X-ray contrast agents: photoinduced transformation and monitoring in surface water. Sci Total Environ 572:340–351. https://doi.org/10.1016/j.scitotenv.2016.08.003
Feng Z, Chen H, Li H et al (2020) Preparation, characterization, and application of magnetic activated carbon for treatment of biologically treated papermaking wastewater. Sci Total Environ 713:136423. https://doi.org/10.1016/j.scitotenv.2019.136423
Foroutan R, Mohammadi R, Razeghi J et al (2019) Performance of algal activated carbon/Fe3O4 magnetic composite for cationic dyes removal from aqueous solutions. Algal Res 40:101509. https://doi.org/10.1016/j.algal.2019.101509
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–470
Gao ZC, Lin YL, Xu B et al (2019) Evaluating iopamidol degradation performance and potential dual-wavelength synergy by UV-LED irradiation and UV-LED/chlorine treatment. Chem Eng J 360:806–816. https://doi.org/10.1016/j.cej.2018.12.022
Ge X, Wu Z, Manzoli M et al (2019) Adsorptive recovery of iopamidol from aqueous solution and parallel reuse of activated carbon: batch and flow study. Ind Eng Chem Res 58:7284–7295. https://doi.org/10.1021/acs.iecr.9b00516
Geng H, Wang F, Yan C et al (2020) Leaching behavior of metals from iron tailings under varying pH and low-molecular-weight organic acids. J Hazard Mater 383:121136. https://doi.org/10.1016/j.jhazmat.2019.121136
Guo H, Li Z, Zhang Y et al (2020) Degradation of chloramphenicol by pulsed discharge plasma with heterogeneous Fenton process using Fe3O4 nanocomposites. Sep Purif Technol 253:117540. https://doi.org/10.1016/j.seppur.2020.117540
Hao Z, Wang C, Yan Z et al (2018) Magnetic particles modification of coconut shell-derived activated carbon and biochar for effective removal of phenol from water. Chemosphere 211:962–969. https://doi.org/10.1016/j.chemosphere.2018.08.038
Hapeshi E, Lambrianides A, Koutsoftas P et al (2013) Investigating the fate of iodinated X-ray contrast media iohexol and diatrizoate during microbial degradation in an MBBR system treating urban wastewater. Environ Sci Pollut Res 20:3592–3606. https://doi.org/10.1007/s11356-013-1605-1
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Hu CY, Hou YZ, Lin YL et al (2019) Kinetics and model development of iohexol degradation during UV/H2O2 and UV/S2O82− oxidation. Chemosphere 229:602–610. https://doi.org/10.1016/j.chemosphere.2019.05.012
Hu CY, Du YF, Lin YL et al (2020) Kinetics of iohexol degradation by ozonation and formation of DBPs during post-chlorination. J Water Process Eng 35:101200. https://doi.org/10.1016/j.jwpe.2020.101200
Islam MA, Ahmed MJ, Khanday WA et al (2017) Mesoporous activated coconut shell-derived hydrochar prepared via hydrothermal carbonization-NaOH activation for methylene blue adsorption. J Environ Manage 203:237–244. https://doi.org/10.1016/j.jenvman.2017.07.029
Jeong CH, Machek EJ, Shakeri M et al (2017) The impact of iodinated X-ray contrast agents on formation and toxicity of disinfection by-products in drinking water. J Environ Sci (china) 58:173–182. https://doi.org/10.1016/j.jes.2017.03.032
Kamran U, Park SJ (2020) MnO2-decorated biochar composites of coconut shell and rice husk: an efficient lithium ions adsorption-desorption performance in aqueous media. Chemosphere 260:127500. https://doi.org/10.1016/j.chemosphere.2020.127500
Kataria N, Garg VK (2017) Removal of Congo red and brilliant green dyes from aqueous solution using flower shaped ZnO nanoparticles. J Environ Chem Eng 5:5420–5428. https://doi.org/10.1016/j.jece.2017.10.035
Kormos JL, Schulz M, Ternes TA (2011) Occurrence of iodinated X-ray contrast media and their biotransformation products in the urban water cycle. Environ Sci Technol 45:8723–8732. https://doi.org/10.1021/es2018187
Lagergren S (1898) About the theory of so-called adsorption of soluble substance. Kung Sven Veten Hand 24:1–39
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403. https://doi.org/10.1021/ja02242a004
Li X, Hu J, Yin D et al (2015) Solid-phase extraction coupled with ultra high performance liquid chromatography and electrospray tandem mass spectrometry for the highly sensitive determination of five iodinated X-ray contrast media in environmental water samples. J Sep Sci 38:1813–2006. https://doi.org/10.1002/jssc.201401296
Li J, Jiang J, Pang S et al (2020) Transformation of X-ray contrast media by conventional and advanced oxidation processes during water treatment: efficiency, oxidation intermediates, and formation of iodinated byproducts. Water Res 185:116234. https://doi.org/10.1016/j.watres.2020.116234
Low MJD (1960) Kinetics of chemisorption of gases on solids. Chem Rev 60:267–312. https://doi.org/10.1021/cr60205a003
Lu T, Chen F (2012) Multiwfn: a multifunctional wavefunction analyzer. J Comput Chem 33:580–592. https://doi.org/10.1002/jcc.22885
Lu J, Zhou Y, Lei J et al (2020) Fe3O4/graphene aerogels: A stable and efficient persulfate activator for the rapid degradation of malachite green. Chemosphere 251:126402. https://doi.org/10.1016/j.chemosphere.2020.126402
Mahouachi L, Rastogi T, Palm WU et al (2020) Natural clay as a sorbent to remove pharmaceutical micropollutants from wastewater. Chemosphere 258:127213. https://doi.org/10.1016/j.chemosphere.2020.127213
Maleki A, Hayati B, Naghizadeh M et al (2015) Adsorption of hexavalent chromium by metal organic frameworks from aqueous solution. J Ind Eng Chem 28:211–216. https://doi.org/10.1016/j.jiec.2015.02.016
Matsushita T, Kobayashi N, Hashizuka M et al (2015) Changes in mutagenicity and acute toxicity of solutions of iodinated X-ray contrast media during chlorination. Chemosphere 135:101–107. https://doi.org/10.1016/j.chemosphere.2015.03.082
Meng L, Yang S, Sun C et al (2017) A novel method for photo-oxidative degradation of diatrizoate in water via electromagnetic induction electrodeless lamp. J Hazard Mater 337:34–46. https://doi.org/10.1016/j.jhazmat.2017.05.005
Mestre AS, Machuqueiro M, Silva M et al (2014) Influence of activated carbons porous structure on iopamidol adsorption. Carbon 77:607–615. https://doi.org/10.1016/j.carbon.2014.05.065
Mohan D, Sarswat A, Singh VK et al (2011) Development of magnetic activated carbon from almond shells for trinitrophenol removal from water. Chem Eng J 172:1111–1125. https://doi.org/10.1016/j.cej.2011.06.054
Moura de Salles Pupo M, Albahaca Oliva JM, Barrios Eguiluz KI et al (2020) Characterization and comparison of Ti/TiO2-NT/SnO2–SbBi, Ti/SnO2–SbBi and BDD anode for the removal of persistent iodinated contrast media (ICM). Chemosphere 253:126701. https://doi.org/10.1016/j.chemosphere.2020.126701
Nguyen TD, Phan NH, Do MH et al (2011) Magnetic Fe2MO4 (M:Fe, Mn) activated carbons: Fabrication, characterization and heterogeneous Fenton oxidation of methyl orange. J Hazard Mater 185:653–661. https://doi.org/10.1016/j.jhazmat.2010.09.068
Nödler K, Licha T, Bester K et al (2010) Development of a multi-residue analytical method, based on liquid chromatography-tandem mass spectrometry, for the simultaneous determination of 46 micro-contaminants in aqueous samples. J Chromatogr A 1217:6511–6521. https://doi.org/10.1016/j.chroma.2010.08.048
Nowicki P, Pietrzak R, Wachowska H (2010) X-ray photoelectron spectroscopy study of nitrogen-enriched active carbons obtained by ammoxidation and chemical activation of brown and bituminous coals. Energy Fuels 24:1197–1206. https://doi.org/10.1021/ef900932g
Oh WD, Lua SK, Dong Z, Lim TT (2015) Performance of magnetic activated carbon composite as peroxymonosulfate activator and regenerable adsorbent via sulfate radical-mediated oxidation processes. J Hazard Mater 284:1–9. https://doi.org/10.1016/j.jhazmat.2014.10.042
Panaritis C, Zgheib J, Ebrahim SAH et al (2020) Electrochemical in-situ activation of Fe-oxide nanowires for the reverse water gas shift reaction. Appl Catal B 269:118826. https://doi.org/10.1016/j.apcatb.2020.118826
Postigo C, Demarini DM, Armstrong MD et al (2018) Chlorination of source water containing iodinated X-ray contrast media: mutagenicity and identification of new iodinated disinfection byproducts. Environ Sc Technol 52:13047–13056. https://doi.org/10.1021/acs.est.8b04625
Radjenovic J, Petrovic M (2016) Sulfate-mediated electrooxidation of X-ray contrast media on boron-doped diamond anode. Water Res 94:128–135. https://doi.org/10.1016/j.watres.2016.02.045
Raj KG, Joy PA (2015) Coconut shell based activated carbon–iron oxide magnetic nanocomposite for fast and efficient removal of oil spills. J Environ Chem Eng 3:2068–2075. https://doi.org/10.1016/j.jece.2015.04.028
Rao MM, Ramana DK, Seshaiah K et al (2009) Removal of some metal ions by activated carbon prepared from Phaseolus aureus hulls. J Hazard Mater 166:1006–1013. https://doi.org/10.1016/j.jhazmat.2008.12.002
Redeker M, Wick A, Meermann B et al (2018) Anaerobic transformation of the iodinated X-ray contrast medium iopromide, its aerobic transformation products, and transfer to further iodinated X-ray contrast media. Environ Sci Technol 52:8309–8320. https://doi.org/10.1021/acs.est.8b01140
Sarswat A, Mohan D (2016) Sustainable development of coconut shell activated carbon (CSAC) & a magnetic coconut shell activated carbon (MCSAC) for phenol (2-nitrophenol) removal. RSC Adv 6:85390–85410. https://doi.org/10.1039/c6ra19756f
Shen Y, Jiang B, Xing Y (2021) Recent advances in the application of magnetic Fe3O4 nanomaterials for the removal of emerging contaminants. Environ Sci Pollut R 28:7599–7620. https://doi.org/10.1007/s11356-020-11877-8
Shimizu Y, Ateia M, Yoshimura C (2018) Natural organic matter undergoes different molecular sieving by adsorption on activated carbon and carbon nanotubes. Chemosphere 203:345–352. https://doi.org/10.1016/j.chemosphere.2018.03.197
Singh RR, Rajnarayanan R, Aga DS (2019) Binding of iodinated contrast media (ICM) and their transformation products with hormone receptors: are ICM the new EDCs? Sci Total Environ 692:32–36. https://doi.org/10.1016/j.scitotenv.2019.07.159
Spessato L, Cazetta AL, Melo S et al (2020) Synthesis of superparamagnetic activated carbon for paracetamol removal from aqueous solution. J Mol Liq 300:112282. https://doi.org/10.1016/j.molliq.2019.112282
Tang L, Yu J, Pang Y et al (2018) Sustainable efficient adsorbent: alkali-acid modified magnetic biochar derived from sewage sludge for aqueous organic contaminant removal. Chem Eng J 336:160–169. https://doi.org/10.1016/j.cej.2017.11.048
Tang Y, Xie Y, Lu G et al (2020) Arsenic behavior during gallic acid-induced redox transformation of jarosite under acidic conditions. Chemosphere 255:126938. https://doi.org/10.1016/j.chemosphere.2020.126938
Tempkin MJ, Pyzhev V (1940) Recent modification to Langmuir isotherms. Acta Physiochem USSR 12:217–222
Ternes TA, Hirsch R (2000) Occurrence and behavior of X-ray contrast media in sewage facilities and the aquatic environment. Environ Sci Technol 34:2741–2748. https://doi.org/10.1021/es991118m
Thines KR, Abdullah EC, Mubarak NM et al (2017) Synthesis of magnetic biochar from agricultural waste biomass to enhancing route for waste water and polymer application: a review. Renew Sust Energ Rev 67:257–276. https://doi.org/10.1016/j.rser.2016.09.057
Wang X, Shu L, Wang Y et al (2011) Sorption of peat humic acids to multi-walled carbon nanotubes. Environ Sci Technol 45:9276–9283. https://doi.org/10.1021/es202258q
Wang F, Sun H, Ren X et al (2017) Effects of humic acid and heavy metals on the sorption of polar and apolar organic pollutants onto biochars. Environ Pollut 231:229–236. https://doi.org/10.1016/j.envpol.2017.08.023
Wang W, Deng S, Li D et al (2018) Adsorptive removal of organophosphate flame retardants from water by non-ionic resins. Chem Eng J 354:105–112. https://doi.org/10.1016/j.cej.2018.08.002
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Engng Div 89:31–59. https://doi.org/10.1061/jsedai.0000430
Wu G, Ma J, Li S et al (2018) Magnetic copper-based metal organic framework as an effective and recyclable adsorbent for removal of two fluoroquinolone antibiotics from aqueous solutions. J Colloid Interface Sci 528:360–371. https://doi.org/10.1016/j.jcis.2018.05.105
Xia NN, Zhang HY, Hu ZH et al (2021) A functionalized bio-based material with abundant mesopores and catechol groups for efficient removal of boron. Chemosphere 263:128202. https://doi.org/10.1016/j.chemosphere.2020.128202
Zhang Y, Zhang TA, Dreisinger D et al (2019) Recovery of vanadium from calcification roasted-acid leaching tailing by enhanced acid leaching. J Hazard Mater 369:632–641. https://doi.org/10.1016/j.jhazmat.2019.02.081
Zhang J, Chen H, He H et al (2020) Adsorption behavior and mechanism of 9-nitroanthracene on typical microplastics in aqueous solutions. Chemosphere 245:125628. https://doi.org/10.1016/j.chemosphere.2019.125628
Zhou Y, Lu J, Zhou Y et al (2019) Recent advances for dyes removal using novel adsorbents: a review. Environ Pollut 252:352–365. https://doi.org/10.1016/j.envpol.2019.05.072
Funding
This research was supported by the Natural Science Foundation of Jiangsu Province (No. BK20191372), the Postgraduate Research & Practice Innovation Program of Jiangsu Province (No. KYCX19_0819), and the National Natural Science Foundation of China (No. 21777067).
Natural Science Foundation of Jiangsu Province, BK20191372, Huan He; Postgraduate Research & Practice Innovation Program of Jiangsu Province, KYCX19_0819, Xinying Cheng; National Natural Science Foundation of China, 21777067, Shaogui Yang.
Author information
Authors and Affiliations
Contributions
Xinying Cheng performed the experiments for her doctoral thesis, analyzed data, interpreted results of experiments, and drafted this manuscript. Professor Limin Zhang and Huan He were the advisers of the doctoral thesis, contributed to the conception and design of this research, and edited and approved the final manuscript. Qiuyi Ji, Dunyu Sun, Jinghua Zhang, Xianxian Chen, Shiyin Li, and Shaogui Yang contributed with the material preparation, data collection, and data analysis, edited previous versions of the manuscript, and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, X., Ji, Q., Sun, D. et al. A comparative study on adsorption behavior of iodinated X-ray contrast media iohexol and amidotrizoic acid by magnetic-activated carbon. Environ Sci Pollut Res 29, 45404–45420 (2022). https://doi.org/10.1007/s11356-022-19127-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-19127-9