Abstract
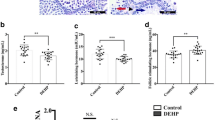
The toxic effect of di(2-ethylhexyl) phthalate (DEHP) on prepubertal testes was examined in this study. We treated 3-week-old male mice with 4.8 mg/kg/day (milligram/kilogram/day) (no observed adverse effect level), 30 mg/kg/day (high exposure dose relative to humans), 100 mg/kg/day (level causing a reproductive system disorder), and 500 mg/kg/day (dose causing a multigenerational reproductive system disorder) of DEHP via gavage. Obvious abnormalities in the testicular organ coefficient, spermatogenic epithelium, and testosterone levels occurred in the 500 mg/kg DEHP group. Ribonucleic acid sequencing (RNA-seq) showed that differentially expressed genes (DEGs) in each group could enrich reproduction and reproductive process terms according to the gene ontology (GO) results, and coenrichment of metabolism pathway was observed by the Reactome pathway analysis. Through the analysis of common genes in the metabolism pathway, we discovered that DEHP exposure at 4.8 to 500 mg/kg or 100 mg/kg caused the same damages to the prepubertal testis. In general, we identified two key transcriptional biomarkers (fatty acid binding protein 3 (Fabp3) and carboxylesterase (Ces) 1d), which provided new insight into the gene regulatory mechanism associated with DEHP exposure and will contribute to the prediction and diagnosis of prepuberty testis injury caused by DEHP.







Similar content being viewed by others
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request.
References
(U.S. Food and Drug Administration 2002) "Safety assessment of di(2-ethylhexyl)phthalate (DEHP) released from PVC medical devices." Available at: http://www.fda.gov/downloads/MedicalDevices/. . ./UCM080457.pdf.Accessed March 8,2018.
Aksglaede L, Juul A, Leffers H, Skakkebaek NE, Andersson AM (2006) The sensitivity of the child to sex steroids: possible impact of exogenous estrogens. Hum Reprod Update 12(4):341–349. https://doi.org/10.1093/humupd/dml018
Albert O, Nardelli TC, Lalancette C, Hales BF, Robaire B (2018) Effects of in utero and lactational exposure to new generation green plasticizers on adult male rats: a comparative study with Di(2-Ethylhexyl) phthalate. Toxicol Sci 164(1):129–141. https://doi.org/10.1093/toxsci/kfy072
Balci A, Ozkemahli G, Erkekoglu P, Zeybek N, Yersal N, Kocer-Gumusel B (2020) Histopathologic, apoptotic and autophagic, effects of prenatal bisphenol A and/or di(2-ethylhexyl) phthalate exposure on prepubertal rat testis. Environ Sci Pollut Res Int 27(16):20104–20116. https://doi.org/10.1007/s11356-020-08274-6
Beg MA, Sheikh IA (2020) Endocrine disruption: structural interactions of androgen receptor against Di(2-ethylhexyl) phthalate and its metabolites. Toxics 8(4). https://doi.org/10.3390/toxics8040115
Bensaad K, Favaro E, Lewis CA, Peck B, Lord S, Collins JM, Pinnick KE, Wigfield S, Buffa FM, Li JL, Zhang Q, Wakelam MJO, Karpe F, Schulze A, Harris AL (2014) Fatty acid uptake and lipid storage induced by HIF-1α contribute to cell growth and survival after hypoxia-reoxygenation. Cell Rep 9(1):349–365. https://doi.org/10.1016/j.celrep.2014.08.056
Botelho GG, Golin M, Bufalo AC, Morais RN, Dalsenter PR, Martino-Andrade AJ (2009) Reproductive effects of di(2-ethylhexyl)phthalate in immature male rats and its relation to cholesterol, testosterone, and thyroxin levels. Arch Environ Contam Toxicol 57(4):777–784. https://doi.org/10.1007/s00244-009-9317-8
Bourdeaux D, Sautou-Miranda V, Bagel-Boithias S, Boyer A, Chopineau J (2004) Analysis by liquid chromatography and infrared spectrometry of di(2-ethylhexyl)phthalate released by multilayer infusion tubing. J Pharm Biomed Anal 35(1):57–64. https://doi.org/10.1016/j.jpba.2003.12.018
Camacho L, Latendresse JR, Muskhelishvili L, Law CD, Delclos KB (2020) Effects of intravenous and oral di(2-ethylhexyl) phthalate (DEHP) and 20% Intralipid vehicle on neonatal rat testis, lung, liver, and kidney. Food Chem Toxicol 144:111497. https://doi.org/10.1016/j.fct.2020.111497
Capela D, Mhaouty-Kodja S (2021) Effects of pubertal exposure to low doses of di-(2-ethylexyl)phthalate on reproductive behaviors in male mice. Chemosphere 263:128191. https://doi.org/10.1016/j.chemosphere.2020.128191
Chen S, Zhou Y, Chen Y, Gu J (2018) fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics (Oxford, England) 34(17):i884–i890. https://doi.org/10.1093/bioinformatics/bty560
Cheng H, Huang C, Tang G, Qiu H, Gao L, Zhang W, Wang J, Yang J, Chen L (2019) Emerging role of EPHX1 in chemoresistance of acute myeloid leukemia by regurlating drug-metabolizing enzymes and apoptotic signaling. Mol Carcinog 58(5):808–819. https://doi.org/10.1002/mc.22973
Cheong AW, Lee YL, Liu WM, Yeung WS, Lee KF (2009) Oviductal microsomal epoxide hydrolase (EPHX1) reduces reactive oxygen species (ROS) level and enhances preimplantation mouse embryo development. Biol Reprod 81(1):126–132. https://doi.org/10.1095/biolreprod.108.071449
Doyle T, Bowman J, Windell V, McLean D, Kim K (2013) Transgenerational effects of di-(2-ethylhexyl) phthalate on testicular germ cell associations and spermatogonial stem cells in mice. Biol Reprod 88(5):112. https://doi.org/10.1095/biolreprod.112.106104
ECHA ECA (2010) "Review of new available information for bis(2-ethylhexyl)phthalate(DEHP)." European Chemical Agency.
Gautheron J, Jéru I (2020) The multifaceted role of epoxide hydrolases in human health and disease. Int J Mol Sci 22(1). https://doi.org/10.3390/ijms22010013
Gill S, Kaur S (1987) Hepatic epoxide hydrolase activities and their induction by clofibrate and diethylhexylphthalate in various strains of mice. Biochem Pharmacol 36(24):4221–4227. https://doi.org/10.1016/0006-2952(87)90662-9
Hackett N, Butler M, Shaykhiev R, Salit J, Omberg L, Rodriguez-Flores J, Mezey J, Strulovici-Barel Y, Wang G, Didon L, Crystal R (2012) RNA-seq quantification of the human small airway epithelium transcriptome. BMC Genomics 13:82. https://doi.org/10.1186/1471-2164-13-82
Han L, Wang J, Zhao T, Wu Y, Wei Y, Chen J, Kang L, Shen L, Long C, Yang Z, Wu S, Wei G (2021) Stereological analysis and transcriptome profiling of testicular injury induced by di-(2-ethylhexyl) phthalate in prepubertal rats. Ecotoxicol Environ Saf 220:112326. https://doi.org/10.1016/j.ecoenv.2021.112326
Ho S, Cheong A, Adgent M, Veevers J, Suen A, Tam N, Leung Y, Jefferson W, Williams C (2017) Environmental factors, epigenetics, and developmental origin of reproductive disorders. Reprod Toxicol (Elmsford, NY) 68:85–104. https://doi.org/10.1016/j.reprotox.2016.07.011
Hossain MS, Tareq KMA, Hammano KI, Tsujii H (2007) Effect of fatty acids on boar sperm motility, viability and acrosome reaction. Reprod Med Biol 6(4):235–239. https://doi.org/10.1111/j.1447-0578.2007.00191.x
Ito Y, Kamijima M, Nakajima T (2019) Di(2-ethylhexyl) phthalate-induced toxicity and peroxisome proliferator-activated receptor alpha: a review. Environ Health Prev Med 24(1):47. https://doi.org/10.1186/s12199-019-0802-z
Jones S, Boisvert A, Naghi A, Hullin-Matsuda F, Greimel P, Kobayashi T, Papadopoulos V, Culty M (2016) Stimulatory effects of combined endocrine disruptors on MA-10 Leydig cell steroid production and lipid homeostasis. Toxicology 355-356:21–30. https://doi.org/10.1016/j.tox.2016.05.008
Karabulut G, Barlas N (2018) Genotoxic, histologic, immunohistochemical, morphometric and hormonal effects of di-(2-ethylhexyl)-phthalate (DEHP) on reproductive systems in pre-pubertal male rats. Toxicol Res (Camb) 7(5):859–873. https://doi.org/10.1039/c8tx00045j
Kavlock R, Boekelheide K, Chapin R, Cunningham M, Faustman E, Foster P, Golub M, Henderson R, Hinberg I, Little R, Seed J, Shea K, Tabacova S, Tyl R, Williams P, Zacharewski T (2002) NTP center for the evaluation of risks to human reproduction: phthalates expert panel report on the reproductive and developmental toxicity of di-isodecyl phthalate. Reprod Toxicol 16(5):655–678. https://doi.org/10.1016/s0890-6238(02)00068-0
Kim D, Langmead B, Salzberg S (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360. https://doi.org/10.1038/nmeth.3317
Kim DH, Park CG, Kim SH, Kim YJ (2019) The effects of mono-(2-ethylhexyl) phthalate (MEHP) on human estrogen receptor (hER) and androgen receptor (hAR) by YES/YAS in vitro assay. Molecules 24(8). https://doi.org/10.3390/molecules24081558
Langmead B, Salzberg S (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9(4):357–359. https://doi.org/10.1038/nmeth.1923
Latini G (2005) Monitoring phthalate exposure in humans. Clin Chim Acta 361(1-2):20–29. https://doi.org/10.1016/j.cccn.2005.05.003
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12:323. https://doi.org/10.1186/1471-2105-12-323
Li N, Wang D, Zhou Y, Ma M, Li J, Wang Z (2010) Dibutyl phthalate contributes to the thyroid receptor antagonistic activity in drinking water processes. Environ Sci Technol 44(17):6863–6868. https://doi.org/10.1021/es101254c
Li X, Sun H, Yao Y, Zhao Z, Qin X, Duan Y, Wang L (2018) Distribution of phthalate metabolites between paired maternal-fetal samples. Environ Sci Technol 52(11):6626–6635. https://doi.org/10.1021/acs.est.8b00838
Li Y, Ma F, Li Z, Yu Y, Yan H, Tahir A, Zheng W, Li X, Huang T, Ge RS (2021) Exposure to 4-bromodiphenyl ether during pregnancy blocks testis development in male rat fetuses. Toxicol Lett 342:38–49. https://doi.org/10.1016/j.toxlet.2021.02.004
Liu G, Cai W, Liu H, Jiang H, Bi Y, Wang H (2021) The association of bisphenol A and phthalates with risk of breast cancer: a meta-analysis. Int J Environ Res Public Health 18(5). https://doi.org/10.3390/ijerph18052375
Liu X, Niu X, Qiu Z (2020) A five-gene signature based on stromal/immune scores in the tumor microenvironment and its clinical implications for liver cancer. DNA Cell Biol 39(9):1621–1638. https://doi.org/10.1089/dna.2020.5512
Long X, Li Q, Zhi LJ, Li JM, Wang ZY (2020) LINC00205 modulates the expression of EPHX1 through the inhibition of miR-184 in hepatocellular carcinoma as a ceRNA. J Cell Physiol 235(3):3013–3021. https://doi.org/10.1002/jcp.29206
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15(12):550. https://doi.org/10.1186/s13059-014-0550-8
Ma TT, Wu LH, Chen L, Zhang HB, Teng Y, Luo YM (2015) Phthalate esters contamination in soils and vegetables of plastic film greenhouses of suburb Nanjing, China and the potential human health risk. Environ Sci Pollut Res Int 22(16):12018–12028. https://doi.org/10.1007/s11356-015-4401-2
Mehrotra K, Morgenstern R, Ahlberg MB, Georgellis A (1999) Hypophysectomy and/or peroxisome proliferators strongly influence the levels of phase II xenobiotic metabolizing enzymes in rat testis. Chem Biol Interact 122(2):73–87. https://doi.org/10.1016/s0009-2797(99)00110-6
Mehrotra K, Morgenstern R, Lundqvist G, Becedas L, Bengtsson Ahlberg M, Georgellis A (1997) Effects of peroxisome proliferators and/or hypothyroidism on xenobiotic-metabolizing enzymes in rat testis. Chem Biol Interact 104(2-3):131–145. https://doi.org/10.1016/s0009-2797(97)00020-3
Monroe AK, Dobs AS (2013) The effect of androgens on lipids. Curr Opin Endocrinol Diabetes Obes 20(2):132–139. https://doi.org/10.1097/MED.0b013e32835edb71
Morris SM Jr (2006) Arginine: beyond protein. Am J Clin Nutr 83(2):508s–512s. https://doi.org/10.1093/ajcn/83.2.508S
Nisa H, Budhathoki S, Morita M, Toyomura K, Nagano J, Ohnaka K, Kono S, Ueki T, Tanaka M, Kakeji Y, Maehara Y, Okamura T, Ikejiri K, Futami K, Maekawa T, Yasunami Y, Takenaka K, Ichimiya H, Terasaka R (2013) Microsomal epoxide hydrolase polymorphisms, cigarette smoking, and risk of colorectal cancer: the Fukuoka Colorectal Cancer Study. Mol Carcinog 52(8):619–626. https://doi.org/10.1002/mc.21897
Ozaki H, Sugihara K, Watanabe Y, Moriguchi K, Uramaru N, Sone T, Ohta S, Kitamura S (2017) Comparative study of hydrolytic metabolism of dimethyl phthalate, dibutyl phthalate and di(2-ethylhexyl) phthalate by microsomes of various rat tissues. Food Chem Toxicol 100:217–224. https://doi.org/10.1016/j.fct.2016.12.019
Pan J, Yao Y, Guo X, Kong F, Zhou J, Meng X (2019) Endoplasmic reticulum stress, a novel significant mechanism responsible for DEHP-induced increased distance between seminiferous tubule of mouse testis. J Cell Physiol 234(11):19807–19823. https://doi.org/10.1002/jcp.28580
Peck CC, Albro PW (1982) Toxic potential of the plasticizer Di(2-ethylhexyl) phthalate in the context of its disposition and metabolism in primates and man. Environ Health Perspect 45:11–17. https://doi.org/10.1289/ehp.824511
Perobelli, J. E. (2014). "The male peripubertal phase as a developmental window for reproductive toxicology studies." Curr Pharm Des 20(34): 5398-5415 10.2174/1381612820666140205150059.
Pertea M, Kim D, Pertea G, Leek J, Salzberg S (2016) Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat Protoc 11(9):1650–1667. https://doi.org/10.1038/nprot.2016.095
Pertea M, Pertea G, Antonescu C, Chang T, Mendell J, Salzberg S (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33(3):290–295. https://doi.org/10.1038/nbt.3122
Qin Y, Han X, Peng Y, Shen R, Guo X, Cao L, Song L, Sha J, Xia Y, Wang X (2012) Genetic variants in epoxide hydrolases modify the risk of oligozoospermia and asthenospermia in Han-Chinese population. Gene 510(2):171–174. https://doi.org/10.1016/j.gene.2012.09.016
Rowland A, Miners JO, Mackenzie PI (2013) The UDP-glucuronosyltransferases: their role in drug metabolism and detoxification. Int J Biochem Cell Biol 45(6):1121–1132. https://doi.org/10.1016/j.biocel.2013.02.019
Saadeldin IM, Hussein MA, Suleiman AH, Abohassan MG, Ahmed MM, Moustafa AA, Moumen AF, Swelum AA-A (2018) Ameliorative effect of ginseng extract on phthalate and bisphenol A reprotoxicity during pregnancy in rats. Environ Sci Pollut Res Int 25(21):21205–21215. https://doi.org/10.1007/s11356-018-2299-1
Shen G, Zhou L, Liu W, Cui Y, Xie W, Chen H, Yu W, Li W, Li H (2017) Di(2-ethylhexyl)phthalate alters the synthesis and β-oxidation of fatty acids and hinders ATP supply in mouse testes via UPLC-Q-exactive orbitrap MS-based metabonomics study. J Agric Food Chem 65(24):5056–5063. https://doi.org/10.1021/acs.jafc.7b01015
Shen L, Tang X, Wei Y, Long C, Tan B, Wu S, Sun M, Zhou Y, Cao X, Wei G (2018) Vitamin E and vitamin C attenuate Di-(2-ethylhexyl) phthalate-induced blood-testis barrier disruption by p38 MAPK in immature SD rats. Reprod Toxicol 81:17–27. https://doi.org/10.1016/j.reprotox.2018.06.015
Silva M, Barr D, Reidy J, Kato K, Malek N, Hodge C, Hurtz D, Calafat A, Needham L, Brock J (2003) Glucuronidation patterns of common urinary and serum monoester phthalate metabolites. Arch Toxicol 77(10):561–567. https://doi.org/10.1007/s00204-003-0486-3
Singh S, Singh SK (2019) Chronic exposure to perfluorononanoic acid impairs spermatogenesis, steroidogenesis and fertility in male mice. J Appl Toxicol 39(3):420–431. https://doi.org/10.1002/jat.3733
Sipilä P, Junnila A, Hakkarainen J, Huhtaniemi R, Mairinoja L, Zhang FP, Strauss L, Ohlsson C, Kotaja N, Huhtaniemi I, Poutanen M (2020) The lack of HSD17B3 in male mice results in disturbed Leydig cell maturation and endocrine imbalance akin to humans with HSD17B3 deficiency. FASEB J 34(5):6111–6128. https://doi.org/10.1096/fj.201902384R
Sosa C, Guillén N, Lucea S, Sorribas V (2020) Effects of oral exposure to arsenite on arsenic metabolism and transport in rat kidney. Toxicol Lett 333:4–12. https://doi.org/10.1016/j.toxlet.2020.07.029
Sun R, Dong C, Li R, Chu H, Liu J, Hao D, Zhang L, Zhao B, Wang L, Zhang Y (2020) Proteomic analysis reveals that EPHX1 contributes to 5-fluorouracil resistance in a human hepatocellular carcinoma cell line. Proteomics Clin Appl 14(4):e1900080. https://doi.org/10.1002/prca.201900080
Tassinari R, Tait S, Busani L, Martinelli A, Narciso L, Valeri M, Gastaldelli A, Deodati A, La Rocca C, Maranghi F (2021) Metabolic, reproductive and thyroid effects of bis(2-ethylhexyl) phthalate (DEHP) orally administered to male and female juvenile rats at dose levels derived from children biomonitoring study. Toxicology 449:152653. https://doi.org/10.1016/j.tox.2020.152653
Tavilani H, Doosti M, Nourmohammadi I, Mahjub H, Vaisiraygani A, Salimi S, Hosseinipanah SM (2007) Lipid composition of spermatozoa in normozoospermic and asthenozoospermic males. Prostaglandins Leukot Essent Fat Acids 77(1):45–50. https://doi.org/10.1016/j.plefa.2007.07.001
Walker C, Ghazisaeidi S, Collet B, Boisvert A, Culty M (2020) In utero exposure to low doses of genistein and di-(2-ethylhexyl) phthalate (DEHP) alters innate immune cells in neonatal and adult rat testes. Andrology 8(4):943–964. https://doi.org/10.1111/andr.12840
Wang C, Huang X (2012) "Lipid metabolism and Drosophila sperm development." Science China. Life Sci 55(1):35–40. https://doi.org/10.1007/s11427-012-4274-2
Wang J, Zhao T, Chen J, Kang L, Wei Y, Wu Y, Han L, Shen L, Long C, Wu S, Wei G (2020) Multiple transcriptomic profiling: p53 signaling pathway is involved in DEHP-induced prepubertal testicular injury via promoting cell apoptosis and inhibiting cell proliferation of Leydig cells. J Hazard Mater:124316. https://doi.org/10.1016/j.jhazmat.2020.124316
Wang K, Sun Y, Guo C, Liu T, Fei X, Chang C (2019) Androgen receptor regulates ASS1P3/miR-34a-5p/ASS1 signaling to promote renal cell carcinoma cell growth. Cell Death Dis 10(5):339. https://doi.org/10.1038/s41419-019-1330-x
Wang YX, You L, Zeng Q, Sun Y, Huang YH, Wang C, Wang P, Cao WC, Yang P, Li YF, Lu WQ (2015) Phthalate exposure and human semen quality: Results from an infertility clinic in China. Environ Res 142:1–9. https://doi.org/10.1016/j.envres.2015.06.010
Wu L, Hafiz MZ, Guan Y, He S, Xiong J, Liu W, Yan B, Li X, Yang J (2018) 17β-estradiol suppresses carboxylesterases by activating c-Jun/AP-1 pathway in primary human and mouse hepatocytes. Eur J Pharmacol 819:98–107. https://doi.org/10.1016/j.ejphar.2017.11.036
Xiao D, Chen YT, Yang D, Yan B (2012) Age-related inducibility of carboxylesterases by the antiepileptic agent phenobarbital and implications in drug metabolism and lipid accumulation. Biochem Pharmacol 84(2):232–239. https://doi.org/10.1016/j.bcp.2012.04.002
Ye L, Zhao B, Hu G, Chu Y, Ge RS (2011) Inhibition of human and rat testicular steroidogenic enzyme activities by bisphenol A. Toxicol Lett 207(2):137–142. https://doi.org/10.1016/j.toxlet.2011.09.001
Yi WEI, Xiang-Liang T, Yu Z, Bin L, Lian-Ju S, Chun-Lan L, Tao LIN, Da-Wei HE, Sheng-de WU, Guang-Hui WEI (2018) DEHP exposure destroys blood-testis barrier (BTB) integrity of immature testes through excessive ROS-mediated autophagy. Genes Dis 5(3):263–274. https://doi.org/10.1016/j.gendis.2018.06.004
Yu Z, Shi Z, Zheng Z, Han J, Yang W, Lu R, Lin W, Zheng Y, Nie D, Chen G (2021) DEHP induce cholesterol imbalance via disturbing bile acid metabolism by altering the composition of gut microbiota in rats. Chemosphere 263:127959. https://doi.org/10.1016/j.chemosphere.2020.127959
Zhang L, Li H, Gao M, Zhang T, Wu Z, Wang Z, Chong T (2018) Genistein attenuates di-(2-ethylhexyl) phthalate-induced testicular injuries via activation of Nrf2/HO-1 following prepubertal exposure. Int J Mol Med 41(3):1437–1446. https://doi.org/10.3892/ijmm.2018.3371
Zhao T, Wang J, Shen L, Long C, Liu B, Wei Y, Han L, Wei Y, Wu S, Wei G (2020a) Increased m6A RNA modification is related to the inhibition of the Nrf2-mediated antioxidant response in di-(2-ethylhexyl) phthalate-induced prepubertal testicular injury. Environ Pollut (Barking, Essex : 1987) 259:113911. https://doi.org/10.1016/j.envpol.2020.113911
Zhao T, Wang J, Wu Y, Han L, Chen J, Wei Y, Shen L, Long C, Wu S, Wei G (2021) Increased m6A modification of RNA methylation related to the inhibition of demethylase FTO contributes to MEHP-induced Leydig cell injury(☆). Environ Pollut 268(Pt A):115627. https://doi.org/10.1016/j.envpol.2020.115627
Zhao TX, Wei YX, Wang JK, Han LD, Sun M, Wu YH, Shen LJ, Long CL, Wu SD, Wei GH (2020b) The gut-microbiota-testis axis mediated by the activation of the Nrf2 antioxidant pathway is related to prepuberal steroidogenesis disorders induced by di-(2-ethylhexyl) phthalate. Environ Sci Pollut Res Int 27(28):35261–35271. https://doi.org/10.1007/s11356-020-09854-2
Zhu BB, Zhang ZC, Li J, Gao XX, Chen YH, Wang H, Gao L, Xu DX (2021) Di-(2-ethylhexyl) phthalate induces testicular endoplasmic reticulum stress and germ cell apoptosis in adolescent mice. Environ Sci Pollut Res Int 28(17):21696–21705. https://doi.org/10.1007/s11356-020-12210-z
Zhuang L, Li C, Chen Q, Jin Q, Wu L, Lu L, Yan X, Chen K (2019) Fatty acid-binding protein 3 contributes to ischemic heart injury by regulating cardiac myocyte apoptosis and MAPK pathways. Am J Physiol Heart Circ Physiol 316(5):H971–h984. https://doi.org/10.1152/ajpheart.00360.2018
Acknowledgements
I express my gratitude to all who helped me during the writing of this thesis. I gratefully acknowledge the help of my supervisor, Mr. Wsd, who has offered me valuable suggestions in my academic studies. Without his consistent and illuminating instruction, this thesis could not have reached its present form.
I am also grateful to Wjk. Under his patient guidance, I learned and completed a substantial amount of work. I am also deeply indebted to Cjd for his help.
I am indebted to my family for their continuous support and encouragement.
I thank AJE for polishing this article.
Funding
This study was supported by the National Natural Science Foundation of China (Grant number 82071632); the Innovation Program for Chongqing’s Overseas Returnees (Grant number cx 2019030); the Chongqing Municipal Health Commission (High-level Medical Reserved Personnel Training Project of Chongqing); and the Guangdong Basic and Applied Basic Research Foundation (No. 2020A1515110796).
Author information
Authors and Affiliations
Contributions
Kl designed the whole experiment, built the model, detected the results, summarized the data, and wrote the first draft. Cjd participated in the modeling. Wjl proposed constructive suggestions for the modification of the paper. Ztx proposed the idea for the experiment. Wyx and Wyh verified the experimental design. Hld prepared the experimental reagent materials. Zxq participated in the data processing. The visualization of the experimental results was guided by Lcl. Slj supervised the experiment, and Wgh reviewed the article and agreed to contribute. Wsd manages and supports the whole project.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experimental procedures in this study, including those involving animals, were approved by the Experimental Animal Committee of Chongqing Medical University.
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kang, L., Chen, J., Wang, J. et al. Multiple transcriptomic profiling: potential novel metabolism-related genes predict prepubertal testis damage caused by DEHP exposure. Environ Sci Pollut Res 29, 13478–13490 (2022). https://doi.org/10.1007/s11356-021-16701-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16701-5