Abstract
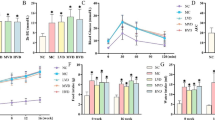
Testicular impairment is a serious complication of diabetes that is mediated by oxidative stress and inflammation. Physalis has antioxidative and anti-inflammatory actions. Thus, the present study investigated the ameliorative role of Physalis juice (PJ) prepared from the fruits against testicular damages in streptozotocin (STZ)-induced diabetic rats. Adult male Wistar rats were divided randomly into five groups (n=6): control, orally administered 5 mL PJ/kg daily (PJ), injected intraperitoneally with a single dose of 55 mg STZ/kg without treatment (STZ), or treated daily with PJ (STZ+PJ) or with 500 mg metformin/kg (STZ+Met), for 28 days. The STZ group showed a marked elevation in the blood glucose level by 230%, whereas remarkable declines in the serum levels of testosterone (44%), follicle-stimulating hormone (FSH) (48%), and luteinizing hormone (LH) (36%), as compared to controls. In comparison to controls, the testis of the STZ group showed remarkable declines in the testis weight (15%), the glutathione (GSH) content (45%), mRNA and protein levels of B-cell lymphoma-2 (Bcl-2) (48 and 35%), mRNA and activities of superoxide dismutase (SOD) (63 and 40%), catalase (CAT) (56 and 31%), glutathione peroxidase (GPx) (51 and 44%), and glutathione reductase (GR) (62 and 43%), whereas marked elevations in the levels of interleukin-1 beta (IL-1β (169%), tumor necrosis factor-alfa (TNFα) (85%), nitric oxide (NO) (96%), malondialdehyde (MDA) (83%), mRNA and protein levels of Bcl-2-associated X protein (Bax) (400 and 61%), and mRNA level of caspase-3 (Cas-3) (370%). Some histopathological alterations were observed in the testicular tissue of the STZ group. In contrast, PJ markedly alleviated all the abovementioned disturbances. In conclusion, PJ at a dose of 5 mL/kg attenuated the diabetes-associated testicular impairments, which may be due to its antioxidative, anti-inflammatory, and antiapoptotic actions.


Similar content being viewed by others
References
Abdel-Daim M, Shaaban A, Madkour F, Elgendy H (2020) Oral spirulina platensis attenuates hyperglycemia and exhibits antinociceptive effect in streptozotocin-induced diabetic neuropathy Rat model. J Pain Res 13:2289–2296. https://doi.org/10.2147/JPR.S267347
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Alanazi IS, Emam M, Elsabagh M et al (2021) The protective effects of 18β-glycyrrhetinic acid against acrylamide-induced cellular damage in diabetic rats. Environ Sci Pollut Res 2021:1–9. https://doi.org/10.1007/S11356-021-14742-4
Albasher G, Alsaleh AS, Alkubaisi N et al (2020) Red beetroot extract abrogates chlorpyrifos-induced cortical damage in rats. Oxid Med Cell Longev 2020. https://doi.org/10.1155/2020/2963020
Aleissa MS, Alkahtani S, Abd Eldaim MA et al (2020) Fucoidan ameliorates oxidative stress, inflammation, DNA damage, and hepatorenal injuries in diabetic rats intoxicated with aflatoxin B 1. Oxid Med Cell Longev 2020. https://doi.org/10.1155/2020/9316751
Ali AA-M, Essawy EA-R, Hamed HSE-DF et al (2021) The ameliorative role of Physalis pubescens L. against neurological impairment associated with streptozotocin induced diabetes in rats. Metab Brain Dis 36(6):1191–1200 https://doi.org/10.1007/s11011-021-00730-7
Al-Olayan EM, El-Khadragy MF, Aref AM et al (2014) The potential protective effect of physalis peruviana L. against carbon tetrachloride-induced hepatotoxicity in rats is mediated by suppression of oxidative stress and downregulation of MMP-9 expression. Oxid Med Cell Longev 2014:1–12. https://doi.org/10.1155/2014/381413
American Diabetes Association (2015) 2. Classification and diagnosis of diabetes. Diabetes Care 38:S8–S16. https://doi.org/10.2337/dc15-S005
Atta M, El-Far A, Farrag F et al (2018) Thymoquinone attenuates cardiomyopathy in streptozotocin-treated diabetic rats. Oxid Med Cell Longev:2018. https://doi.org/10.1155/2018/7845681
Ballester J, Muñoz MC, Domínguez J et al (2004)Insulin-dependent diabetes affects testicular function by FSH- and LH-linked mechanisms. J Androl 25:706–719. https://doi.org/10.1002/j.1939-4640.2004.tb02845.x
Barham D, Trinder P (1972) An improved colour reagent for the determination of blood glucose by the oxidase system. Analyst 97:142–145. https://doi.org/10.1039/an9729700142
Baty RS, Hassan KE, Alsharif KF et al (2020) Neuroprotective role of luteolin against lead acetate-induced cortical damage in rats. Hum Exp Toxicol 39:1200–1212. https://doi.org/10.1177/0960327120913094
Beutler E, Duron O, Kelly B (1963) Improved method for the determination of blood glutathione - PubMed. J Lab Clin Med 61:882–888
Castro A, Rodriguez L, Vargas E (2008) Dry gooseberry (Physalis peruviana L) with pretreatment of osmotic dehydration. Vitae Rev Fac Quim Farm 15:226–231
Chen X, Li X, Zhang X et al (2019) Antihyperglycemic and antihyperlipidemic activities of a polysaccharide from Physalis pubescens L. In streptozotocin (STZ)-induced diabetic mice. Food Funct 10:4868–4876. https://doi.org/10.1039/c9fo00687g
El Husseny M, Mamdouh M, Shaban S et al (2017) Adipokines: potential therapeutic targets for vascular dysfunction in type II diabetes mellitus and obesity. J Diabetes Res 2017:1–11. https://doi.org/10.1155/2017/8095926
El Sheikha AF, Piombo G, Goli T, Montet D (2010) Main composition of Physalis (Physalis pubescens L.) fruit juice from Egypt. Fruits 65:255–265. https://doi.org/10.1051/FRUITS/2010021
El-Beltagi HS, Mohamed HI, Safwat G et al (2019) Chemical composition and biological activity of Physalis peruviana L. Gesunde Pflanz. 71:113–122. https://doi.org/10.1007/S10343-019-00456-8
Gadelkarim M, Abushouk A, Ghanem E et al (2018) Adipose-derived stem cells: effectiveness and advances in delivery in diabetic wound healing. Biomed Pharmacother 107:625–633. https://doi.org/10.1016/J.BIOPHA.2018.08.013
Goldberg DM, Spooner RJ (1983) Method for the determination of glutathione reductase. Methods Enzym Anal 3:258–265
Green LC, Wagner DA, Glogowski J et al (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138. https://doi.org/10.1016/0003-2697(82)90118-X
Hakkarainen J, Zhang FP, Jokela H et al (2018) Hydroxysteroid (17β) dehydrogenase 1 expressed by Sertoli cells contributes to steroid synthesis and is required for male fertility. Fed Am Soc Exp Biol J 32:3229–3241. https://doi.org/10.1096/fj.201700921R
Hasnan J, Yusof MI, Damitri TD et al (2010) Relationship between apoptotic markers (Bax and Bcl-2) and biochemical markers in type 2 diabetes mellitus. Singapore Med J 51:50–55
Hassan AI, Ghoneim MAM (2013) A possible inhibitory effect of physalis (Physalis pubescens L.) On Diabetes in Male Rats. World Appl Sci J 21:681–688. https://doi.org/10.5829/idosi.wasj.2013.21.5.7220
Inih OS, Esther YE, Adetola FO et al (2018) Testicular dysfunction is a common feature in Men with type 2 diabetes mellitus in a Nigerian tertiary hospital. Curr Diabetes Rev 14:298–306. https://doi.org/10.2174/1573399813666170425152046
Kim HJ, Jung BH, Yoo KY et al (2017) Determination of the critical diabetes duration in a streptozotocin-induced diabetic rat calvarial defect model for experimentation regarding bone regeneration. J Periodontal Implant Sci 47:339–350. https://doi.org/10.5051/jpis.2017.47.5.339
Livak K, Schmittgen T (2001) Analysis of relative gene expression data using real-time quantitative pCR and the 2(-Delta delta c(T)) method. Methods San Diego Calif 25:402–408
Long L, Qiu H, Cai B et al (2018) Hyperglycemia induced testicular damage in type 2 diabetes mellitus rats exhibiting microcirculation impairments associated with vascular endothelial growth factor decreased via PI3K/Akt pathway. Oncotarget 9:5321–5336. https://doi.org/10.18632/oncotarget.23915
Maresch CC, Stute DC, Ludlow H et al (2017) Hyperglycemia is associated with reduced testicular function and activin dysregulation in the Ins2Akita+/− mouse model of type 1 diabetes. Mol Cell Endocrinol 446:91–101. https://doi.org/10.1016/j.mce.2017.02.020
Marinova D, Ribarova F, Atanassova M (2005) Total phenolic and total flavonoids in Bulgarian fruits and vegetables. J Univ Chem Technol Metall 40:255–260
Martin LJ, Touaibia M (2020) Improvement of testicular steroidogenesis using flavonoids and isoflavonoids for prevention of late-onset male hypogonadism. Antioxidants 9:237
Mohamed AA, Ali MM, Dorrah MA, Bassal TT (2018) Mediation of inducible nitric oxide and immune-reactive lysozymes biosynthesis by eicosanoid and biogenic amines in flesh flies. Int J Trop Insect Sci 38(1):93–104. https://doi.org/10.1017/S1742758417000315
Morsy GM, Abou el-Ala KS, Ali AA (2016) Studies on fate and toxicity of nanoalumina in male albino rats: oxidative stress in the brain, liver and kidney. Toxicol Ind Health 32:200–214. https://doi.org/10.1177/0748233713498462
Murphy CJ, Richburg JH (2014) Implications of Sertoli cell induced germ cell apoptosis to testicular pathology. Spermatogenesis 4:e979110. https://doi.org/10.4161/21565562.2014.979110
Nishikimi M, Appaji N, Yagi K (1972) The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem Biophys Res Commun 46:849–854. https://doi.org/10.1016/S0006-291X(72)80218-3
Nna VU, Bakar ABA, Ahmad A et al (2019) Oxidative stress, NF-κB-mediated inflammation and apoptosis in the testes of streptozotocin–induced diabetic rats: combined protective effects of Malaysian propolis and metformin. Antioxidants 8:465. https://doi.org/10.3390/antiox8100465
Nna VU, Bakar ABA, Ahmad A, Mohamed M (2020) Diabetes-induced testicular oxidative stress, inflammation, and caspase-dependent apoptosis: the protective role of metformin. Arch Physiol Biochem 126:377–388. https://doi.org/10.1080/13813455.2018.1543329
O’Donnell L, Stanton P, de Kretser DM (2000) Endocrinology of the male reproductive system and spermatogenesis. MDText.com, Inc
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Othman MS, Nada A, Zaki HS, Abdel Moneim AE (2014) Effect of Physalis peruviana L. on cadmium-induced testicular toxicity in rats. Biol Trace Elem Res 159:278–287. https://doi.org/10.1007/s12011-014-9955-1
Paglia D, Valentine W (1967) Studies on the quantitative and quali¬tative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Pathak A, Rathore AS, Bhutani V, Pathak R (2012) Role of zinc on antioxidative enzymes and lipid peroxidation in brain of diabetic rats. 3. https://doi.org/10.4172/2157-7609.1000122
Pérez-Panero A, Ruiz-Muñoz M, Cuesta-Vargas A, Gónzalez-Sánchez M (2019) Prevention, assessment, diagnosis and management of diabetic foot based on clinical practice guidelines: a systematic review. Medicine (Baltimore) 98:e16877. https://doi.org/10.1097/MD.0000000000016877
Preston BT, Stevenson IR, Lincoln GA et al (2012) Testes size, testosterone production and reproductive behaviour in a natural mammalian mating system. J Anim Ecol 81:296–305. https://doi.org/10.1111/J.1365-2656.2011.01907.X
Redza-Dutordoir M, Averill-Bates DA (2016) Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys Acta Mol Cell Res 1863:2977–2992
Saeedi P, Petersohn I, Salpea P, et al (2019) Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. 157:107843. https://doi.org/10.1016/j.diabres.2019.107843
Sayed AA, Ali AA, Mohamed HRH (2018) Fertility enhancing efficacy of Cicer arietinum in male albino mice. Cell Mol Biol 64:29–38
Schmidt AM (2018) Highlighting diabetes mellitus. Arterioscler Thromb Vasc Biol 38:e1–e8
Shenstone E, Lippman Z, Van Eck J (2020) A review of nutritional properties and health benefits of Physalis species. Plant Foods Hum Nutr 75:316–325
Shrilatha B, Muralidhara (2007) Early oxidative stress in testis and epididymal sperm in streptozotocin-induced diabetic mice: Its progression and genotoxic consequences. Reprod Toxicol 23:578–587. https://doi.org/10.1016/j.reprotox.2007.02.001
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16
Soliman GZA, Soliman AS, Abbas MS, Abdallah SMM (2019) Egyptian Physalis peruviana L. as anti-diabetic and hypolipidemic natural herb: a promising treatment of alloxan-induced diabetic rats. Case Stud J 8:62–73
Subramanian S (2014) Physalis peruviana Linn. fruit extract improves insulin sensitivity and ameliorates hyperglycemia in high-fat diet low dose STZ-induced type 2 diabetic rats. J Pharm Res 8:625–632
Tian Y, Song W, Xu D et al (2020) Autophagy induced by ROS aggravates testis oxidative damage in diabetes via breaking the feedforward loop linking p62 and Nrf2. Oxid Med Cell Longev 2020. https://doi.org/10.1155/2020/7156579
Tuhin R, Begum M, Rahman M et al (2017) Wound healing effect of Euphorbia hirta linn. (Euphorbiaceae) in alloxan induced diabetic rats. BMC Complement Altern Med 17. https://doi.org/10.1186/S12906-017-1930-X
Waisundara VY, Hsu A, Huang D, Tan BKH (2008) Scutellaria baicalensis: enhances the anti-diabetic activity of metformin in streptozotocin-induced diabetic wistar rats. Am J Chin Med 36:517–540. https://doi.org/10.1142/S0192415X08005953
Wang C, Liu Y, Wang Y et al (2019a) Low-frequency pulsed electromagnetic field promotes functional recovery, reduces inflammation and oxidative stress, and enhances HSP70 expression following spinal cord injury. Mol Med Rep 19:1687–1693. https://doi.org/10.3892/mmr.2019.9820
Wang Y, Wang D, Jin Z (2019b) MiR-27a suppresses TLR4-induced renal ischemia-reperfusion injury. Mol Med Rep 20:967–976. https://doi.org/10.3892/mmr.2019.10333
Ward D, Bousfield G, Moore K (1991) Gonadotropins. In: Cupps PT, ed. Reproduction in domestic animals. San Diego, Calif
Wei M, Ong L, Smith MT et al (2003) The streptozotocin-diabetic rat as a model of the chronic complications of human diabetes. Heart Lung Circ 12:44–50. https://doi.org/10.1046/J.1444-2892.2003.00160.X
Zhang D, Yu YJ, Sen XF et al (2019) Recombinant betatrophin (Angptl-8/lipasin) ameliorates streptozotocin-induced hyperglycemia and β-cell destruction in neonatal rats. Mol Med Rep 20:4523–4532. https://doi.org/10.3892/mmr.2019.10719
Acknowledgements
The authors would like to acknowledge Prof. Dr. Ramy Basheer Ibrahim, Faculty of Science, Helwan University, for his helpful contributions in the molecular analysis.
Availability of data and materials
All data are available upon request.
Author information
Authors and Affiliations
Contributions
AAA, EAE, and FAA contributed to the planning of the experimental design. NSM contributed to the animal handling and experimental procedures. AEA and EAE contributed to the molecular analysis and experimental procedures. AAA contributed to the statistical analysis and interpretation of data. AAA contributed to the manuscript writing and editing.
Corresponding author
Ethics declarations
Ethics approval
All the handling and sampling procedures, in the present study, were approved by Cairo University – Institutional Animal Care and Use Committee (CU-IACUC), Giza, Egypt, under the approval number of CU/I/F/C/22/19.
Consent to participate
It is not applicable.
Consent for publication
The data provided here is original.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ali, A.AM., Essawy, E.AR., Mohamed, N.S. et al. Physalis pubescens L. alleviates testicular disruptions associated with streptozotocin-induced diabetes in male Wistar rats, Rattus norvegicus. Environ Sci Pollut Res 29, 12300–12312 (2022). https://doi.org/10.1007/s11356-021-16616-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16616-1