Abstract
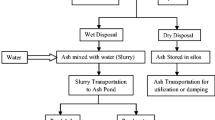
Incense sticks ash is one of the most unexplored by-products generated at religious places and houses obtained after the combustion of incense sticks. Every year, tonnes of incense sticks ash is produced at religious places in India which are disposed of into the rivers and water bodies. The presence of heavy metals and high content of alkali metals challenges a potential threat to the living organism after the disposal in the river. The leaching of heavy metals and alkali metals may lead to water pollution. Besides this, incense sticks also have a high amount of calcium, silica, alumina, and ferrous along with traces of rutile and other oxides either in crystalline or amorphous phases. The incense sticks ash, heavy metals, and alkali metals can be extracted by water, mineral acids, and alkali. Ferrous can be extracted by magnetic separation, while calcium by HCl, alumina by sulfuric acid treatment, and silica by strong hydroxides like NaOH. The recovery of such elements by using acids and bases will eliminate their toxic heavy metals at the same time recovering major value-added minerals from it. Here, in the present research work, the effect on the elemental composition, morphology, crystallinity, and size of incense sticks ash particles was observed by extracting ferrous, followed by extraction of calcium by HCl and alumina by H2SO4 at 90–95 °C for 90 min. The final residue was treated with 4 M NaOH, in order to extract leachable silica at 90 °C for 90 min along with continuous stirring. The transformation of various minerals phases and microstructures of incense sticks ash (ISA) and other residues during ferrous, extraction, calcium, and alumina and silica extraction was studied using Fourier transform infrared (FTIR), dynamic light scattering (DLS), X-ray fluorescence (XRF), X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), and inductively coupled plasma-optical emission spectroscopy (ICP-OES). DLS was used for analyzing the size during the experiments while FTIR helped in the confirmation of the formation of new products during the treatments. From the various instrumental analyses, it was found that the toxic metals present in the initial incense sticks ash got eliminated. Besides this, the major alkali metals, i.e., Ca and Mg, got reduced during these successive treatments. Initially, there were mainly irregular shaped, micron-sized particles that were dominant in the incense sticks ash particles. Besides this, there were plenty of carbon particles left unburned during combustion. In the final residue, nanosized flowers shaped along with cuboidal micron-sized particles were dominant. present in If, such sequential techniques will be applied by the industries based on recycling of incense sticks ash, then not only the solid waste pollution will be reduced but also numerous value-added minerals like ferrous, silica, alumina calcium oxides and carbonates can be recovered from such waste. The value-added minerals could act as an economical and sustainable source of adsorbent for wastewater treatment in future.







Similar content being viewed by others
Change history
05 November 2021
A Correction to this paper has been published: https://doi.org/10.1007/s11356-021-17310-y
Abbreviations
- CFAS:
-
calcium-ferro-aluminosilicate
- ISA:
-
incense sticks ash
- FE:
-
incense sticks ash residue after ferrous recovery
- FeCa:
-
incense sticks ash residue after ferrous and calcium recovery
- FeCaAl:
-
incense sticks ash residue after ferrous, calcium and aluminum recovery
- FeCaAlSi:
-
incense sticks ash residue after ferrous, calcium, aluminum and silica recovery
References
Alehyen S, Zerzouri M, ELalouani M, Achouri ME (2017) Porosity and fire resistance of fly ash based geopolymer. J Mater Environ Sci 8:3676–3689
Ali H, Khan E (2019) Trophic transfer, bioaccumulation, and biomagnification of non-essential hazardous heavy metals and metalloids in food chains/webs—concepts and implications for wildlife and human health. Hum Ecol Risk Assess 25:1353–1376. https://doi.org/10.1080/10807039.2018.1469398
Baby J, Raj JS, Biby ET, Sankarganesh P, Jeevitha MV, Ajisha SU, Rajan SS (2010) Toxic effect of heavy metals on aquatic environment. Int J Biol Chem Sci 4:939–952. https://doi.org/10.4314/ijbcs.v4i4.62976
Balaram V (2019) Rare earth elements: a review of applications, occurrence, exploration, analysis, recycling, and environmental impact. Geosci Front 10:1285–1303. https://doi.org/10.1016/j.gsf.2018.12.005
Bo J, Huifeng W, Yang L, Zhen L (2018) Preparation of iron-modified portland cement adsorbent and the investigation of its decolorization performance. IOP Conference Series: Earth and Environmental Science 121:022015. https://doi.org/10.1088/1755-1315/121/2/022015
Fang GC, Chang CN, Chu CC, Wu YS, Fu PPC, Chang SC, Yang IL (2003) Fine (PM2.5), coarse (PM2.5–10), and metallic elements of suspended particulates for incense burning at Tzu Yun Yen temple in central Taiwan. Chemosphere 51:983–991. https://doi.org/10.1016/S0045-6535(03)00124-3
Gomes S, François M, Abdelmoula M, Refait P, Pellissier C, Evrard O (1999) Characterization of magnetite in silico-aluminous fly ash by SEM, TEM, XRD, magnetic susceptibility, and Mössbauer spectroscopy. Cem Concr Res 29:1705–1711. https://doi.org/10.1016/S0008-8846(99)00133-7
Gong Y, Grant DJW, Brittain HG (2007) Principles of solubility. In: Augustijns P, Brewster ME (eds) Solvent systems and their selection in pharmaceutics and biopharmaceutics. Biotechnology: Pharmaceutical Aspects. vol VI. Springer, New York, NY, pp 1–27. https://doi.org/10.1007/978-0-387-69154-1_1
Gunasekaran S, Anbalagan G, Pandi S (2006) Raman and infrared spectra of carbonates of calcite structure. J Raman Spectrosc 37:892–899. https://doi.org/10.1002/jrs.1518
Hazarika P, Dutta NB, Biswas SC, Dutta RC, Jayaraj RSC (2018) Status of agarbatti industry in India with special reference to Northeast. Int J Adv Res Biol Sci 5:173–186
Hoang TA, Ang AM, Rohl AL (2007) Effects of temperature on the scaling of calcium sulphate in pipes. Powder Technol 179:31–37. https://doi.org/10.1016/j.powtec.2006.11.013
Hsueh HT, Ko TH, Chou WC, Hung WC (2011) Health risk of aerosols and toxic metals from incense and joss paper burning. Environ Chem Lett 10:79–87. https://doi.org/10.1007/s10311-011-0331-5
Jain SN, Tamboli SR, Sutar DS, Mawal VN, Shaikh AA, Prajapati AA (2020) Incense stick ash as a novel and sustainable adsorbent for sequestration of Victoria Blue from aqueous phase. Sustain Chem Pharm 15:100199. https://doi.org/10.1016/j.scp.2019.100199
Keawthun MS, Krachodnok S, Chaisena A (2014) Conversion of waste glasses into sodium silicate solutions. Int J Chem Sci 12:83–91
Khezri B, Chan YY, Tiong LYD, Webster RD (2015) Annual air pollution caused by the Hungry Ghost Festival. Environ Sci Process Impacts 17:1578–1586. https://doi.org/10.1039/c5em00312a
Kirubakaran D, Santhoshraja V (2017) Utilization of pelletized fly ash aggregate to replace the natural aggregate: A review. Int Res J Eng 4:148–151
Kumar S, Singh J, Mohapatra SK (2018) Role of particle size in assessment of physico-chemical properties and trace elements of Indian fly ash. Waste Manag Res 36:1016–1022. https://doi.org/10.1177/0734242X18804033
Lin T-C, Yang C-R, Chang F-H (2007) Burning characteristics and emission products related to metallic content in incense. J Hazard Mater 140:165–172. https://doi.org/10.1016/j.jhazmat.2006.06.052
Lin TC, Krishnaswamy G, Chi DS (2008) Incense smoke: clinical, structural and molecular effects on airway disease. Clin Mol Allergy 6:3–6. https://doi.org/10.1186/1476-7961-6-3
Liu X, Zhou J, Zhou S, Yue Y, Qiu J (2018) Transparent glass-ceramics functionalized by dispersed crystals. Prog Mater Sci 97:38–96. https://doi.org/10.1016/j.pmatsci.2018.02.006
Liu H, Wang Y, Zhao S, Hu H, Cao C, Li A, Yu Y, Yao H (2020) Review on the current status of the co-combustion technology of organic solid waste (OSW) and coal in China. Energy Fuel 34:15448–15487
Mishra S, Singh AL, Tiwary D (2018) Studies of physico-chemical status of the ponds at Varanasi Holy City under anthropogenic influences. Int J Environ Res Dev 4:261–268
Myszka B, Schüßler M, Hurle K, Demmert B, Detsch R, Boccaccini AR, Wolf SE (2019) Phase-specific bioactivity and altered Ostwald ripening pathways of calcium carbonate polymorphs in simulated body fluid. RSV Adv 9:18232–18244
Ni M, Ratner BD (2008) Differentiating calcium carbonate polymorphs by surface analysis techniques-an XPS and TOF-SIMS study. Surf Interface Anal 40(10):1356–1361. https://doi.org/10.1002/sia.2904
Ojha S (2020) Features of chemical properties of metal oxide glass nanocomposites. In: Metal Oxide Glass Nanocomposites. Elsevier, pp 147–157. https://doi.org/10.1016/b978-0-12-817458-6.00007-x
Pal MK, Gautam J (2012) Synthesis and characterization of polyacrylamide-calcium carbonate and polyacrylamide-calcium sulfate nanocomposites. Polym Compos 33(4):515–523. https://doi.org/10.1002/pc.22183
Ratshiedana R, Kuvarega AT, Mishra AK (2021) Titanium dioxide and graphitic carbon nitride–based nanocomposites and nanofibres for the degradation of organic pollutants in water: a review. Environ Sci Pollut Res 28:10357–10374. https://doi.org/10.1007/s11356-020-11987-3
Risdanareni P, Puspitasari P, Jaya EJ (2017) Chemical and physical characterization of fly ash as geopolymer material. In MATEC Web of Conferences, EDP Sciences 97:01031. https://doi.org/10.1051/matecconf/20179701031
Salah N, Al-Ghamdi AA, Memic A, Habib SS, Khan ZH (2016) Formation of carbon nanotubes from carbon-rich fly ash: growth parameters and mechanism. Mater Manuf Process 31(2):146–156. https://doi.org/10.1080/10426914.2015.1025975
Sharina AH, Lee YH, Musa A (2008) Effects of gold nanoparticles on the response of phenol biosensor containing photocurable membrane with tyrosinase. Sensors 8(10):6407–6416. https://doi.org/10.3390/s8106407
Shoumkova AS (2011) Magnetic separation of coal fly ash from Bulgarian power plants. Waste Manag Res 29(10):1078–1089. https://doi.org/10.1177/0734242x10379494
Stoppa F, Scordari F, Mesto E, Sharygin VV, Bortolozzi G (2010) Calcium-aluminum-silicate-hydrate “cement” phases and rare Ca-zeolite association at Colle Fabbri, Central Italy. Cent Eur J Geosci 2(2):175–187. https://doi.org/10.2478/v10085-010-0007-6
Weng CH (2020) Water pollution prevention and state of the art treatment technologies. Environ Sci Pollut Res 27:34583–34585. https://doi.org/10.1007/s11356-020-09994-5
Yadav VK, Yadav KK, Gnanamoorthy G, Choudhary N, Khan SH, Gupta N, Bach QV (2020) A novel synthesis and characterization of polyhedral shaped amorphous iron oxide nanoparticles from incense sticks ash waste. Environ Technol Innov 20:101089. https://doi.org/10.1016/j.eti.2020.101089
Yadav VK, Gnanamoorthy G, Cabral-Pinto MM, Alam J, Ahamed M, Gupta N, Yadav KK (2021) Variations and similarities in structural, chemical, and elemental properties on the ashes derived from the coal due to their combustion in open and controlled manner. Environ Sci Pollut Res:1–17. https://doi.org/10.1007/s11356-021-12989-5
Yadav VK, Fulekar MH (2014) Isolation and charcterization of iron nanoparticles from coal fly ash fromGandhinagar (Gujarat) thermal power plant (A mechanical method of isolation). Int J Eng Res Technol 3:471–479
Yin Y, Yin H, Wu Z, Qi C, Tian H, Zhang W, Hu Z, Feng L (2019) Characterization of coals and coal ashes with highSi content using combined second-derivative infrared spectroscopy and Raman spectroscopy. Crystals 9:513
Ylmén R, Jäglid U (2013) Carbonation of portland cement studied by diffuse reflection fourier transform infrared spectroscopy. Int J Concr Struct Mater 7:119–125
Acknowledgements
The authors extend their appreciation to the Deputyship for Research & Innovation, “Ministry of Education” in Saudi Arabia for funding this research work through the project number IFKSURG-1439-085. The authors are also thankful to Central Instrumental Facility (CIF), CUG, Gandhinagar, Jamia Millia Islamia, New Delhi, and SRM University Chennai for extending their characterization and instrument facilities.
Funding
The authors extend their appreciation to the Deputyship for Research & Innovation, “Ministry of Education” in Saudi Arabia for funding this research work through the project number IFKSURG-1439-085.
Author information
Authors and Affiliations
Contributions
VKY investigated the samples for ICP-AES, XRF, and EDS, and prepared an original draft of the manuscript. Material preparation, data collection, analysis and supervision were performed by KKY. JA and MMSCP, investigated and interpreted XRD, FTIR, and PSA results. GG and HK critically evaluated the manuscript. MA analyzed and interpreted FESEM micrographs. AAH analyzed and interpreted TEM micrographs. FAA prepared the original draft of the manuscript. AKS prepared the original draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The affiliations, Acknowledgment and Funding text is modified in the original proof.
Rights and permissions
About this article
Cite this article
Yadav, V.K., Yadav, K.K., Alam, J. et al. Transformation of hazardous sacred incense sticks ash waste into less toxic product by sequential approach prior to their disposal into the water bodies. Environ Sci Pollut Res 30, 71766–71778 (2023). https://doi.org/10.1007/s11356-021-15009-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15009-8