Abstract
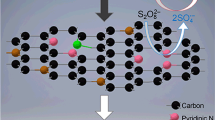
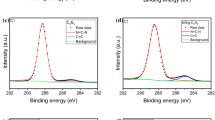
Metal-free carbonaceous composite membranes have been proven to effectively drive novel in situ catalytic oxidation for the degradation of organic pollutants via persulfates activation. In this study, nitrogen-doped graphene (NG) was employed as a modifier to enhance the catalytic activity of the carbon mats by assembly with reduced graphene oxide (rGO) and carbon nanotubes (CNTs) on the top of a nylon supporter. The morphology and performance of the NG/rGO/CNTs composite membrane were compared to those obtained without the addition of NG (rGO/CNTs). Owing to the larger nanochannels for water delivery and stronger hydrophobicity on the surface, the NG/rGO/CNTs composite membrane shows a superior low-pressure filtration performance in favor of energy-saving operation. For the in situ catalytic oxidation of the NG/rGO/CNTs composite membrane through the activation of peroxydisufate (PDS), the average removal rate of sulfamethoxazole (SMX), one of frequently detected sulfonamide antibiotics in water, can reach 21.7 mg·m−2·h−1 under continuous filtration mode, which was 17% more rapid than that of the rGO/CNTs, resulting in significant detoxifying of the oxidation intermediates. Owing to the addition of NG into the carbon mats, the reactive nitrogen-doped sites identified by X-Ray photoelectron spectroscopy (XPS), such as pyridinic and graphitic N, played important roles in PDS activation, while both the radical and non-radical pathways were involved in in situ catalytic oxidation. According to the experimental evidence of the effects that solution environment has on the SMX removal and transmembrane pressure, the NG/rGO/CNTs composite membrane shows a relatively high resistance to changes in the solution pH, chloride ion inhibition, and background organics fouling. These results suggest a new approach to the application of activated persulfate oxidation in water treatment, such that improvements to the reaction stability warrant further investigation.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Ahmad M, Teel AL, Watts RJ (2013) Mechanism of persulfate activation by phenols. Environ Sci Technol 47(11):5864–5871
American Public Health Association (1998) Standard methods for examination of water and wastewater, 20th edn. American Water Works Association, Washington DC
Bassin JP, Kleerebezem R, Dezotti M, van Loosdrecht MC (2012) Measuring biomass specific ammonium, nitrite and phosphate uptake rates in aerobic granular sludge. Chemosphere 89(10):1161–1168
Chen H, Carroll KC (2016) Metal-free catalysis of persulfate activation and organic-pollutant degradation by nitrogen-doped graphene and aminated graphene. Environ Pollut 215:96–102
Chen X, Oh WD, Lim TT (2018) Graphene- and CNTs-based carbocatalysts in persulfates activation: material design and catalytic mechanisms. Chem Eng J 354:941–976
Cheng X, Guo H, Zhang Y, Korshin GV, Yang B (2019) Insights into the mechanism of nonradical reactions of persulfate activated by carbon nanotubes: activation performance and structure-function relationship. Water Res 157:406–414
Devi P, Das U, Dalai AK (2016) In-situ chemical oxidation: principle and applications of peroxide and persulfate treatments in wastewater systems. Sci Total Environ 571:643–657
Duan X, Ao Z, Sun H, Indrawirawan S, Wang Y, Kang J, Wang S (2015a) Nitrogen-doped graphene for generation and evolution of reactive radicals by metal-free catalysis. ACS Appl Mater Interfaces 7(7):4169–4178
Duan X, O’Donnell K, Sun H, Wang Y, Wang S (2015b) Sulfur and nitrogen Co-doped graphene for metal-free catalytic oxidation reactions. Small 11(25):3036–3044
Fang G, Gao J, Dionysiou DD, Liu C, Zhou D (2013) Activation of persulfate by quinones: free radical reactions and implication for the degradation of PCBs. Environ Sci Technol 47(9):4605–4611
Frank B, Zhang J, Blume R, Schlogl R, Su DS (2009) Heteroatoms increase the selectivity in oxidative dehydrogenation reactions on nanocarbons. Angew Chem Int Ed Eng 48(37):6913–6917
Gao SJ, Qin H, Liu P, Jin J (2015) SWCNT-intercalated GO ultrathin films for ultrafast separation of molecules. J Chromatogr A 3(12):6649–6654
Gao PS, Guo Y, Li X, Wang X, Wang J, Qian F, Zhang Z (2018) Magnetic solid phase extraction of sulfonamides based on carboxylated magnetic graphene oxide nanoparticles in environmental waters. J Chromatogr A 1575:1–10
Gao J, Luo C, Gan L, Wu D, Tan F, Cheng X, Zhou W (2020) A comparative study of UV/H2O2 and UV/PDS for the degradation of micro-pollutants: kinetics and effect of water matrices. Environ Sci Pollut Res 27(19):24531–24541
Guan C, Jiang J, Pang S, Luo C, Ma J, Zhou Y, Yang Y (2017) Oxidation kinetics of bromophenols by nonradical activation of peroxydisulfate in the presence of carbon nanotube and formation of brominated polymeric products. Environ Sci Technol 51(18):10718–10728
Guan C, Jiang J, Pang S, Ma J, Chen X, Lim TT (2019) Nonradical transformation of sulfamethoxazole by carbon nanotube activated peroxydisulfate: kinetics, mechanism and product toxicity. Chem Eng J 378(15):122147
Huang H, Sheng J, Qian F, Zhou F, Gao S, Shen X (2018) Effects of graphene oxide incorporation on the mat structure and performance of carbon nanotube composite membranes. Res Chem Intermed 45:533–548
Isidori M, Lavorgna M, Nardelli A, Pascarella L, Parrella A (2005) Toxic and genotoxic evaluation of six antibiotics on non-target organisms. Sci Total Environ 346(13):87–98
Ji Y, Fan Y, Liu K, Kong D, Lu J (2015) Thermo activated persulfate oxidation of antibiotic sulfamethoxazole and structurally related compounds. Water Res 87:1–9
Lei Y, Chen CS, Tu YJ, Huang YH, Zhang H (2015) Heterogeneous degradation of organic pollutants by persulfate activated by CuO-Fe3O4: mechanism, stability, and effects of pH and bicarbonate ions. Environ Sci Technol 49(11):6838–6845
Li D, Duan X, Sun H, Kang J, Zhang H, Tade MO, Wang S (2017) Facile synthesis of nitrogen-doped graphene via low-temperature pyrolysis: the effects of precursors and annealing ambience on metal-free catalytic oxidation. Carbon 115:649–658
Lienert J, Gudel K, Escher BI (2007) Screening method for ecotoxicological hazard assessment of 42 pharmaceuticals considering human metabolism and excretory routes. Environ Sci Technol 41(12):4471–4478
Liu Y, Yu L, Ong CN, Xie J (2016) Nitrogen-doped graphene nanosheets as reactive water purification membranes. Nano Res 9:1983–1993
Liu H, Liu Y, Tang L, Wang J, Xie Q (2020) Egg shell biochar-based green catalysts for the removal of organic pollutants by activating persulfate. Sci Total Environ 745:141095
Matta R, Tlili S, Chiron S, Barbati S (2010) Removal of carbamazepine from urban wastewater by sulfate radical oxidation. Environ Chem Lett 9(3):347–353
Pan X, Chen J, Wu N, Qi Y, Xu X, Ge J, Wang X (2018) Degradation of aqueous 2,4,4′-Trihydroxybenzophenone by persulfate activated with nitrogen doped carbonaceous materials and the formation of dimer products. Water Res 143:176–187
Pedrosa M, Drazic G, Tavares PB, Figueiredo JL, Silva AMT (2019) Metal-free graphene-based catalytic membrane for degradation of organic contaminants by persulfate activation. Chem Eng J 369:223–232
Qi C, Liu X, Ma J, Lin C, Li X, Zhang H (2016) Activation of peroxymonosulfate by base: implications for the degradation of organic pollutants. Chemosphere 151:280–288
Qian F, Chen X, Wang J, Shen Y, Gao J, Mei J (2017) Differentiation in nitrogen-converting activity and microbial community structure between granular size fractions in a continuous autotrophic nitrogen removal reactor. J Microbiol Biotechnol 27(10):1798–1807
Ren W, Nie G, Zhou P, Zhang H, Duan X, Wang S (2020) The intrinsic nature of persulfate activation and N-doping in carbocatalysis. Environ Sci Technol 54(10):6438–6447
Sheng J, Yin H, Qian F, Huang H, Gao S, Wang J (2020) Reduced graphene oxide-based composite membranes for in-situ catalytic oxidation of sulfamethoxazole operated in membrane filtration. Sep Purif Technol 236:116275
Song T, Tian W, Qiao K, Zhao J, Chu M, Du Z, Wang L, Xie W (2021) Adsorption behaviors of polycyclic aromatic hydrocarbons and oxygen derivatives in wastewater on N-doped reduced graphene oxide. Sep Purif Technol 254:117565
Sun H, Wang Y, Liu S, Ge L, Wang L, Zhu Z, Wang S (2013) Facile synthesis of nitrogen doped reduced graphene oxide as a superior metal-free catalyst for oxidation. Chem Commun 49(85):9914–9916
Tang L, Liu Y, Wang J, Zeng G, Deng Y, Dong H, Peng B (2018) Enhanced activation process of persulfate by mesoporous carbon for degradation of aqueous organic pollutants: electron transfer mechanism. Appl Catal B Environ 231:1–10
Vieira O, Ribeiro RS, Pedrosa M, Lado Ribeiro AR, Silva AMT (2020) Nitrogen-doped reduced graphene oxide – PVDF nanocomposite membrane for persulfate activation and degradation of water organic micropollutants. Chem Eng J 402:126117
Wang F, Ma S, Si Y, Dong L, Wang X, Yao J, Xing B (2017) Interaction mechanisms of antibiotic sulfamethoxazole with various graphene-based materials and multiwall carbon nanotubes and the effect of humic acid in water. Carbon 114:671–678
Yang Y, Lu X, Jiang J, Ma J, Liu G, Cao Y, Luo C (2017) Degradation of sulfamethoxazole by UV, UV/H2O2 and UV/persulfate (PDS): formation of oxidation products and effect of bicarbonate. Water Res 118:196–207
Yu J, Feng H, Tang L, Pang Y, Zeng G, Lu Y, Wang J (2020a) Metal-free carbon materials for persulfate-based advanced oxidation process: microstructure, property and tailoring. Prog Mater Sci 111:100654
Yu J, Tang L, Pang Y, Zeng G, Feng H, Zou J, Tan J (2020b) Hierarchical porous biochar from shrimp shell for persulfate activation: a two-electron transfer path and key impact factors. Appl Catal B-Environ 260:118160
Zhang T, Chen Y, Wang Y, Le Roux J, Yang Y, Croue JP (2014) Efficient peroxydisulfate activation process not relying on sulfate radical generation for water pollutant degradation. Environ Sci Technol 48(10):5868–5875
Zhang M, Chen X, Zhou H, Murugananthan M, Zhang Y (2015) Degradation of p-nitrophenol by heat and metal ions co-activated persulfate. Chem Eng J 264:39–47
Acknowledgments
The authors acknowledge the support from the Qinglan Project for Jiangsu Colleges and Universities, China.
Funding
This study was supported by the National Natural Science Foundation of China (51608341), the Natural Science Foundation of Jiangsu Province, China (BK20150284), and the Open Project of National & Local Joint Engineering Laboratory for Municipal Sewage Resource Utilization Technology (No.2019KF02).
Author information
Authors and Affiliations
Contributions
Feiyue Qian developed the research proposal, and was a major contributor in writing the manuscript; Honggui Yin and Jiayi Sheng performed the fabrication of composite membranes and in situ catalytic oxidation for SMX degradation, analyzed the original experiment data; Shiqian Gao developed the quantitative analysis methods of SMX at low concentration, and was responsible for the extraction of oxidation intermediates from the filtrate. Feng Liu designed the system of in situ catalytic oxidation, and interpreted the results regarding the effects of solution environment; Yaoliang Shen gave important suggestions in the improvement of this manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ricardo Torres-Palma
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 743 kb).
Rights and permissions
About this article
Cite this article
Qian, F., Yin, H., Liu, F. et al. The in situ catalytic oxidation of sulfamethoxazole via peroxydisufate activation operated in a NG/rGO/CNTs composite membrane filtration. Environ Sci Pollut Res 28, 26828–26839 (2021). https://doi.org/10.1007/s11356-021-12545-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12545-1