Abstract
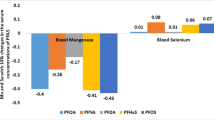
Data (N = 2552) from National Health and Nutrition Examination Survey for US adults aged ≥ 20 years for 2011–2016 were analyzed to estimate the associations between the concentrations of blood cadmium, lead, and total mercury and the concentrations of seven perfluoroalkyl acids (PFAA), namely, 2-(N-Methyl-perfluorooctane sulfonamido) acetic acid (MPAH), perfluorooctanoic acid (PFOA), perfluorononanoic acid (PFNA), perfluorodecanoic acid (PFDA), perfluoroundecanoic acid (PFUnDA), perfluorohexane sulfonic acid (PFHxS), and perfluorooctane sulfonic acid (PFOS). Concentrations of blood cadmium were negatively associated with the concentrations of PFHxS (β = − 0.05428, p < 0.01) and PFOS (β = − 0.0212, p = 0.02). Concentrations of blood lead were positively associated with the concentrations of MPAH (β = 0.03301, p < 0.01), PFOA (β = 0.04783, p = 0.01), PFNA (β = 0.11761, p < 0.01), PFDA (β = 0.08007, p < 0.01), PFUA (β = 0.11382, p < 0.01), and PFOS (β = 0.04996, p = 0.02). Percent increases in the concentration of blood lead were 0.32%, 0.46%, 1.13%, 0.77%, 1.09%, and 0.48% for 10% increases in the concentrations of MPAH, PFOA, PFNA, PFDA, PFUA, and PFOS, respectively. Concentrations of blood total mercury were positively associated with the concentrations of PFNA (β = 0.37105, p < 0.01), PFDA (β = 0.46875, p < 0.01), PFUA (β = 0.56934, p < 0.01), and PFOS (β = 0.17557, p < 0.01). Percent increases in the concentration of blood total mercury were 3.6%, 4.57%, 5.58%, and 1.69% for 10% increases in the concentrations of PFNA, PFDA, PFUA, and PFOS, respectively. Associations between the concentrations of PFAAs with blood total mercury were substantially stronger than the concentrations with blood lead. Higher the carbon chain length for PFAAs, stronger were the associations between PFAAs with lead and mercury.

Similar content being viewed by others
Data availability
All data used for analysis for this study are in public domain and available free of charge at www.cdc.gov/nchs/nhanes/index.htm
References
Banjabi AA, Li AJ, Kumosani TA, Yousef JM, Kannan K (2020) Serum concentrations of perfluoroalkyl substances and their association with osteoporosis in a population in Jeddah. Saudi Arabia Environ Res 187:109676. https://doi.org/10.1016/j.envres.2020.109676
Bao W-W, Qian ZM, Geiger SD, Liu E, Liu Y, Wang S-Q, Lawrence WR, Yang B-Y, Hu L-W, Zeng X-W, Dong G-H (2017) Gender-specific associations between serum isomers of perfluoroalkyl substances and blood pressure among Chinese: isomers of C8 health project in China. Sci Total Environ 607-608:1304–1312. https://doi.org/10.1016/j.scitotenv.2017.07.124
Buha A, Đukić-Ćosić D, Ćurčić M, Bulat Z, Antonijević B, Moulis JM, Goumenou M, Wallace D (2020) Emerging links between cadmium exposure and insulin resistance: human, animal, and cell study data. Toxics. 8(3):E63. https://doi.org/10.3390/toxics8030063
Buser MC, Ingber SZ, Raines N, Fowler DA, Scinicariello F (2016) Urinary and blood cadmium and lead and kidney function: NHANES 2007-2012. Int J Hyg Environ Health 219(3):261–267. https://doi.org/10.1016/j.ijheh.2016.01.005
Cao X, Fu M, Bi R, Zheng X, Fu B, Tian S, Liu C, Li Q, Liu J (2021) Cadmium induced BEAS-2B cells apoptosis and mitochondria damage via MAPK signaling pathway. Chemosphere. 263:128346. https://doi.org/10.1016/j.chemosphere.2020.128346
Cave M, Appana S, Patel M, Falkner KC, McClain CJ, Brock G (2010) Polychlorinated biphenyls, lead, and mercury are associated with liver disease in American adults: NHANES 2003–2004. Environ Health Perspect 118(12):1735–1742. https://doi.org/10.1289/ehp.1002720
Cecil KM, Brubaker CJ, Adler CM, Dietrich KN, Altaye M, Egelhoff JC, Wessel S, Elangovan I, Hornung R, Jarvis K, Lanphear BP (2008) Decreased brain volume in adults with childhood lead exposure. PLoS Med 5(5):e112. https://doi.org/10.1371/journal.pmed.0050112
Chang ET, Adami HO, Boffetta P, Cole P, Starr TB, Mandel JS (2014) A critical review of perfluorooctanoate and perfluorooctanesulfonate exposure and cancer risk in humans. Crit Rev Toxicol 44(Suppl 1):1–81. https://doi.org/10.3109/10408444.2014.905767
Chen A, Kim SS, Chung E, Dietrich KN (2013) Thyroid hormones in relation to lead, mercury, and cadmium exposure in the National Health and Nutrition Examination Survey, 2007–2008. Environ Health Perspect 121(2):181–186. https://doi.org/10.1289/ehp.1205239
Dasgupta P, Kulkarni P, Bhat NS, Majid S, Shiina M, Shahryari V, Yamamura S, Tanaka Y, Gupta RK, Dahiya R, Hashimoto Y (2020) Activation of the Erk/MAPK signaling pathway is a driver for cadmium induced prostate cancer. Toxicol Appl Pharmacol 401:115102. https://doi.org/10.1016/j.taap.2020.115102
Deubler EL, Gapstur SM, Diver WR et al (2020) Erythrocyte levels of cadmium and lead and risk of B-cell non-Hodgkin lymphoma and multiple myeloma. Int J Cancer. https://doi.org/10.1002/ijc.33136
Djordjevic VR, Wallace DR, Schweitzer A, Boricic N, Knezevic D, Matic S, Grubor N, Kerkez M, Radenkovic D, Bulat Z, Antonijevic B, Matovic V, Buha A (2019) Environmental cadmium exposure and pancreatic cancer: evidence from case control, animal and in vitro studies. Environ Int 128:353–361. https://doi.org/10.1016/j.envint.2019.04.048
Edwards J, Ackerman C (2016) A review of diabetes mellitus and exposure to the environmental toxicant cadmium with an emphasis on likely mechanisms of action. Curr Diabetes Rev 12(3):252–258. https://doi.org/10.2174/1573399811666150812142922
Filippini T, Torres D, Lopes C, Carvalho C, Moreira P, Naska A, Kasdagli MI, Malavolti M, Orsini N, Vinceti M (2020) Cadmium exposure and risk of breast cancer: a dose-response meta-analysis of cohort studies. Environ Int 142:105879. https://doi.org/10.1016/j.envint.2020.105879
Geier DA, Kern JK, Geier MR (2017) Blood lead levels and learning disabilities: a cross-sectional study of the 2003-2004 National Health and Nutrition Examination Survey (NHANES). Int J Environ Res Public Health 14(10):1202. https://doi.org/10.3390/ijerph14101202
Geier DA, Kern JK, Geier MR (2018) A cross-sectional study of the relationship between blood lead levels and reported attention deficit disorder: an assessment of the economic impact on the United States. Metab Brain Dis 33(1):201–208. https://doi.org/10.1007/s11011-017-0146-6
Genchi G, Sinicropi MS, Lauria G, Carocci A, Catalano A (2020) The effects of cadmium toxicity. Int J Environ Res Public Health 17(11):3782. https://doi.org/10.3390/ijerph17113782
Girardi P, Merler E (2019) A mortality study on male subjects exposed to polyfluoroalkyl acids with high internal dose of perfluorooctanoic acid. Environ Res 179(Pt A):108743. https://doi.org/10.1016/j.envres.2019.108743
Gomis MI, Vestergren R, MacLeod M, Muller JF, Cousins IT (2017) Historical human exposure to perfluoroalkyl acids in the United States and Australia reconstructed from biomonitoring data using population-based pharmacokinetic modelling. Environ Int 108:92–102
Harari F, Sallsten G, Christensson A, Petkovic M, Hedblad B, Forsgard N, Melander O, Nilsson PM, Borné Y, Engström G, Barregard L (2018) Blood lead levels and decreased kidney function in a population-based cohort. Am J Kidney Dis 72(3):381–389
He X, Liu Y, Xu B, Gu L, Tang W (2018) PFOA is associated with diabetes and metabolic alteration in US men: National Health and Nutrition Examination Survey 2003-2012. Sci Total Environ 625:566–574. https://doi.org/10.1016/j.scitotenv.2017.12.186
Hodgson S, Nieuwenhuijsen MJ, Elliott P, Jarup L (2007) Kidney disease mortality and environmental exposure to mercury. Am J Epidemiol 165(1):72–77
Hurley S, Goldberg D, Wang M, Park JS, Petreas M, Bernstein L, Anton-Culver H, Nelson DO, Reynolds P (2018) Breast cancer risk and serum levels of per- and poly-fluoroalkyl substances: a case-control study nested in the California Teachers Study. Environ Health 17(1):83. https://doi.org/10.1186/s12940-018-0426-6
Hyder O, Chung M, Cosgrove D, Herman JM, Li Z, Firoozmand A, Gurakar A, Koteish A, Pawlik TM (2013) Cadmium exposure and liver disease among US adults. J Gastrointest Surg 17(7):1265–1273. https://doi.org/10.1007/s11605-013-2210-9
Innes KE, Wimsatt JH, Frisbee S, Ducatman AM (2014) Inverse association of colorectal cancer prevalence to serum levels of perfluorooctane sulfonate (PFOS) and perfluorooctanoate (PFOA) in a large Appalachian population. BMC Cancer 14:45. Published 2014 Jan 27. https://doi.org/10.1186/1471-2407-14-45
Jain RB (2018) Revised and extended serum cotinine cut-offs to classify smokers and nonsmokers. Biomarkers. 23:502–507. https://doi.org/10.1080/1354750X.2018.1443516
Jain RB (2019a) Co-exposures to toxic metals cadmium, lead, and mercury and their impact on unhealthy kidney function. Environ Sci Pollut Res Int 26(29):30112–30118. https://doi.org/10.1007/s11356-019-06182-y
Jain RB (2019b) Synergistic impact of co-exposures to toxic metals cadmium, lead, and mercury along with perfluoroalkyl substances on the healthy kidney function. Environ Res 169:342–347. https://doi.org/10.1016/j.envres.2018.11.037
Jain RB (2020) Variabilities in the concentrations of selected perfluoroalkyl acids among normotensives and hypertensives across various stages of glomerular function. Arch Environ Occup Health. https://doi.org/10.1080/19338244.2020.1732856
Jain RB, Ducatman A (2019) Roles of gender and obesity in defining correlations between perfluoroalkyl substances and lipid/lipoproteins. Sci Total Environ 653:74–81. https://doi.org/10.1016/j.scitotenv.2018.10.362
Kumar S, Sharma A (2019) Cadmium toxicity: effects on human reproduction and fertility. Rev Environ Health 34(4):327–338. https://doi.org/10.1515/reveh-2019-0016
Lanphear BP, Hornung R, Khoury J, Yolton K, Baghurst P, Bellinger DC, Canfield RL, Dietrich KN, Bornschein R, Greene T, Rothenberg SJ, Needleman HL, Schnaas L, Wasserman G, Graziano J, Roberts R (2005) Low-level environmental lead exposure and children’s intellectual function: an international pooled analysis [published correction appears in Environ Health Perspect. 2019 Sep;127(9):99001]. Environ Health Perspect 113(7):894–899. https://doi.org/10.1289/ehp.7688
Lin YS, Ginsberg G, Caffrey JL, Xue J, Vulimiri SV, Nath RG, Sonawane B (2014) Association of body burden of mercury with liver function test status in the U.S. population. Environ Int 70:88–94. https://doi.org/10.1016/j.envint.2014.05.010
Lubin JH, Colt JS, Camann D, Davis S, Cerhan JR, Severson RK, Bernstein L, Hartage P (2004) Epidemiologic evaluation of measurement data in the presence of detection limits. Environ Health Perspect 112(17):1691–1696
Mancini FR, Rajaobelina K, Praud D, Dow C, Antignac JP, Kvaskoff M, Severi G, Bonnet F, Boutron-Ruault MC, Fagherazzi G (2018) Nonlinear associations between dietary exposures to perfluorooctanoic acid (PFOA) or perfluorooctane sulfonate (PFOS) and type 2 diabetes risk in women: Findings from the E3N cohort study. Int J Hyg Environ Health 221(7):1054–1060. https://doi.org/10.1016/j.ijheh.2018.07.007
Mancini FR, Cano-Sancho G, Gambaretti J, Marchand P, Boutron-Ruault MC, Severi G, Arveux P, Antignac JP, Kvaskoff M (2020) Perfluorinated alkylated substances serum concentration and breast cancer risk: Evidence from a nested case-control study in the French E3N cohort. Int J Cancer 146(4):917–928. https://doi.org/10.1002/ijc.32357
Mannino DM, Homa DM, Matte T, Hernandez-Avila M (2005) Active and passive smoking and blood lead levels in U.S. adults: data from the Third National Health and Nutrition Examination Survey. Nicotine Tob Res 7(4):557–564. https://doi.org/10.1080/14622200500185264
Marshall AT, Betts S, Kan EC, McConnell R, Lanphear BP, Sowell ER (2020) Association of lead-exposure risk and family income with childhood brain outcomes. Nat Med 26(1):91–97. https://doi.org/10.1038/s41591-019-0713-y
Obeng-Gyasi E (2018) Lead exposure and oxidative stress-a life course approach in U.S. Adults. Toxics 6(3):42. https://doi.org/10.3390/toxics6030042
Obeng-Gyasi E (2019) Cholesterol and oxidative stress in U.S. pregnant women exposed to lead. Med Sci (Basel) 7(3):42. https://doi.org/10.3390/medsci7030042
Orr SE, Bridges CC (2017) Chronic kidney disease and exposure to nephrotoxic metals. Int J Mol Sci 18(5):1039. https://doi.org/10.3390/ijms18051039
Razzaghi H, Tinker SC, Crider K (2014) Blood mercury concentrations in pregnant and nonpregnant women in the United States: National Health and Nutrition Examination Survey 1999-2006. Am J Obstet Gynecol 210(4):357.e1–357.e9. https://doi.org/10.1016/j.ajog.2013.10.884
Shi H, Sun X, Kong A, Ma H, Xie Y, Cheng D, Wong CKC, Zhou Y, Gu J (2021) Cadmium induces epithelial-mesenchymal transition and migration of renal cancer cells by increasing PGE2 through a cAMP/PKA-COX2 dependent mechanism. Ecotoxicol Environ Saf 207:111480. https://doi.org/10.1016/j.ecoenv.2020.111480
Somers EC, Ganser MA, Warren JS, Basu N, Wang L, Zick SM, Park SK (2015) Mercury exposure and antinuclear antibodies among females of reproductive age in the United States: NHANES. Environ Health Perspect 123(8):792–798. https://doi.org/10.1289/ehp.1408751
Taha MM, Mahdy-Abdallah H, Shahy EM, Ibrahim KS, Elserougy S (2018) Impact of occupational cadmium exposure on bone in sewage workers. Int J Occup Environ Health 24(3-4):101–108. https://doi.org/10.1080/10773525.2018.1518745
Tinkov AA, Gritsenko VA, Skalnaya MG, Cherkasov SV, Aaseth J, Skalny AV (2018) Gut as a target for cadmium toxicity. Environ Pollut 235:429–434. https://doi.org/10.1016/j.envpol.2017.12.114
Yang J, Guo T, Liang X, Zhai Y, Cheng Y, Sun H, Cui Y, Cheng X (2020) Cadmium inhibits apoptosis of human esophageal epithelial cells by upregulating CDK6. Ecotoxicol Environ Saf 205:111146. https://doi.org/10.1016/j.ecoenv.2020.111146
Zalups RK (1998) Basolateral uptake of inorganic mercury in the kidney. Toxicol Appl Pharmacol 151:192–199. https://doi.org/10.1006/taap.1998.8416
Zhang W, Wang F, Xu P, Miao C, Zeng X, Cui X, Lu C, Xie H, Yin H, Chen F, Ma J, Gao S, Fu Z (2014) Perfluorooctanoic acid stimulates breast cancer cells invasion and up-regulates matrix metalloproteinase-2/-9 expression mediated by activating NF-κB. Toxicol Lett 229(1):118–125. https://doi.org/10.1016/j.toxlet.2014.06.004
Author information
Authors and Affiliations
Contributions
The responsibility of executing every aspect of this study was borne by Ram B. Jain. This included conceptualizing and designing the study; generating study database; deciding the methods of statistical analysis; conducting the data analysis; generating, tabulating, and interpreting the study results; and ultimately, writing, reviewing, revising, and finalizing the study manuscript.
Corresponding author
Ethics declarations
Ethical approval
Since this study did not recruit any human and/or animal subjects, this section does not apply.
Consent to participate
Since this study did not recruit any human subjects, this section does not apply.
Consent to publish
Since this study is not attempting to re-publish/publish any third party or author’s previously published material, this section does not apply.
Competing interests
The author declares that he has no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jain, R.B. Associations between concentrations of selected perfluoroalkyl acids and concentrations of blood cadmium, lead, and total mercury. Environ Sci Pollut Res 28, 26537–26544 (2021). https://doi.org/10.1007/s11356-021-12493-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12493-w