Abstract
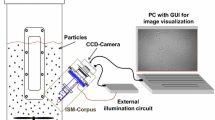
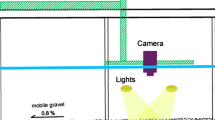
Microfaunal identification and analysis are very complex; thus, an image analysis method was utilized in this paper to overcome the shortcomings of using the number, dominant species, and diversity of population structure of microfauna as activated sludge indicators. Based on a classification of microfaunal movement, the quantitative processing and analysis of the micro-video frame image of microfaunal movement were carried out by using the Image J software. Background subtraction method was utilized to detect target microfauna by matching target area features to track microfaunal movement. Three parameters, namely, motion trajectory (L), consecutive frame of motion paths (Si), and average change rate of extent \( \left(\overline{A_r}\right) \), were selected to represent the motion trajectory and mass center of microfauna. Four motion-velocity parameters, namely, the left and right rotation angles of adjacent frames (∆αi), instantaneous velocity (Vi), average linear velocity (\( \overline{V} \)), and average angular velocity (\( \overline{W} \)), were selected to characterize the movement modes of microfauna. Finally, a motion analysis method based on the Image J software was established to demonstrate the different motion modes of microfauna in activated sludge. This study provides a methodological foundation for the establishment of a new method of microfauna as indicator. Based on this method, the correlation between the microfaunal motion velocity and activated sludge flocs was analyzed.









Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, Kun Wang, upon reasonable request.
References
Al-Shahwani SM, Horan NJ (1991) The use of protozoa to indicate changes in the performance of activated sludge plants. Water Res 25:633–638. https://doi.org/10.1016/0043-1354(91)90038-r
Amaral AL, Leal CS, Vaz AI, Vieira JC, Quinteiro AC, Costa ML, Castro LM (2018) Use of chemometric analyses to assess biological wastewater treatment plants by protozoa and metazoa monitoring. Environ Monit Assess 190:497–512. https://doi.org/10.1007/s10661-018-6882-1
Araujo dos Santos L, Ferreira V, Pereira MO, Nicolau A (2014) Relationship between protozoan and metazoan communities and operation and performance parameters in a textile sewage activated sludge system. Eur J Protistol 50:319–328. https://doi.org/10.1016/j.ejop.2014.03.005
Bouwmans T (2014) Traditional and recent approaches in background modeling for foreground detection: an overview. Comput Sci Rev 11-12:31–66. https://doi.org/10.1016/j.cosrev.2014.04.001
Bownik A (2017) Daphnia swimming behaviour as a biomarker in toxicity assessment: a review. Sci Total Environ 601-602:194–205. https://doi.org/10.1016/j.scitotenv.2017.05.199
Chen J, Wang Z, Li G, Guo R (2014) The swimming speed alteration of two freshwater rotifers Brachionus calyciflorus and Asplanchna brightwelli under dimethoate stress. Chemosphere 95:256–260. https://doi.org/10.1016/j.chemosphere.2013.08.086
Domínguez C, Heras J, Pascual V (2017) IJ-OpenCV: combining ImageJ and OpenCV for processing images in biomedicine. Comput Biol Med 84:189–194. https://doi.org/10.1016/j.compbiomed.2017.03.027
Foissner W (2016) Protists as bioindicators in activated sludge: identification, ecology and future needs. Eur J Protistol 55:75–94. https://doi.org/10.1016/j.ejop.2016.02.004
Guo R, Ren X, Ren H (2012) A new method for analysis of the toxicity of organophosphorus pesticide, dimethoate on rotifer based on response surface methodology. J Hazard Mater 237-238:270–276. https://doi.org/10.1016/j.jhazmat.2012.08.041
Jafarinejad S (2016) Cost estimation and economical evaluation of three configurations of activated sludge process for a wastewater treatment plant (WWTP) using simulation. Appl Water Sci 7:2513–2521. https://doi.org/10.1007/s13201-016-0446-8
Lee S, Basu S, Tyler CW, Wei IW (2004) Ciliate populations as bio-indicators at Deer Island Treatment Plant. Adv Environ Res 8:371–378. https://doi.org/10.1016/s1093-0191(02)00118-1
Lu X, Xu C, Wang L, Teng L (2018) Improved background subtraction method for detecting moving objects based on GMM. IEEJ Trans Electr Electron Eng 13:1540–1550. https://doi.org/10.1002/tee.22718
Madoni P (1994) A sludge biotic index (SBI) for the evaluation of the biological performance of activated sludge plants based on the microfauna analysis. Water Res 28:67–75. https://doi.org/10.1016/0043-1354(94)90120-1
Maini R, Aggarwal H (2010) A comprehensive review of image enhancement. Tech J Comput 2:8–13
Nielsen ME, Roslev P (2018) Behavioral responses and starvation survival of Daphnia magna exposed to fluoxetine and propranolol. Chemosphere 211:978–985. https://doi.org/10.1016/j.chemosphere.2018.08.027
Nikitin OV, Nasyrova EI, Nuriakhmetova VR, Stepanova NY, Danilova NV, Latypova VZ (2018) Toxicity assessment of polluted sediments using swimming behavior alteration test with Daphnia magna. IOP Conf Ser Earth Environ Sci 107. https://doi.org/10.1088/1755-1315/107/1/012068
Ntougias S, Tanasidis S, Melidis P (2011) Microfaunal indicators, Ciliophora phylogeny and protozoan population shifts in an intermittently aerated and fed bioreactor. J Hazard Mater 186:1862–1869. https://doi.org/10.1016/j.jhazmat.2010.12.099
Ogleni N, Damar Y (2011) Microbiological and performance evaluation of sequencing batch reactor for textile. Water Environ Res. https://doi.org/10.2175/106143011x13233670703323
Otsu N (1979) A Threshold selection method from gray level histogram [J]. IEEE Trans Syst Man Cybern 9(1):62–66. https://doi.org/10.1109/TSMC.1979.4310076
Pajdak-Stos A, Sobczyk M, Fialkowska E, Kocerba-Soroka W, Fyda J (2017) The effect of three different predatory ciliate species on activated sludge microfauna. Eur J Protistol 58:87–93. https://doi.org/10.1016/j.ejop.2017.01.001
Schindelin J, Rueden CT, Hiner MC, Eliceiri KW (2015) The ImageJ ecosystem: an open platform for biomedical image analysis. Mol Reprod Dev 82:518–529. https://doi.org/10.1002/mrd.22489
Shen W-F, Zhang Z-S, Gong X-J, Gu M-R, Shi Z-X, Wei Y-X (1999) New technology of micro biological monitoring. China Architecture & Building Press, Beijing
Stolze N, Bader C, Henning C, Mastin J, Holmes AE, Sutlief AL (2019) Automated image analysis with ImageJ of yeast colony forming units from cannabis flowers. J Microbiol Methods 164. https://doi.org/10.1016/j.mimet.2019.105681
Untersteiner H, Kahapka J, Kaiser H (2003) Behavioural response of the cladoceran Daphnia magna Straus to sublethal Copper stress—validation by image analysis. Aquat Toxicol 65:435–442. https://doi.org/10.1016/s0166-445x(03)00157-7
Villar SA, Torcida S, Acosta GG (2016) Median filtering: a new insight. J Math Imaging Vision 58:130–146. https://doi.org/10.1007/s10851-016-0694-0
Villarroel MJ, Ferrando MD, Sancho E, Andreu E (1999) Daphnia magna feeding behavior after exposure to tetradifon and recovery from intoxication. Ecotoxicol Environ Saf 44:40–46. https://doi.org/10.1006/eesa.1999.1817
Wang J, Chon K, Ren X, Kou Y, Chae K-J, Piao Y (2019) Effects of beneficial microorganisms on nutrient removal and excess sludge production in an anaerobic-anoxic/oxic (A(2)O) process for municipal wastewater treatment. Bioresour Technol 281:90–98. https://doi.org/10.1016/j.biortech.2019.02.047
Yilmaz A, Javed O, Shah M (2006) Object tracking. ACM Comput Surv 38:13. https://doi.org/10.1145/1177352.1177355
Zhou K, Xu M, Dai J, Cao H (2006) The microfauna communities and operational monitoring of an activated sludge plant in China. Eur J Protistol 42:291–295. https://doi.org/10.1016/j.ejop.2006.07.005
Zhou K, Xu M, Liu B, Cao H (2008) Characteristics of microfauna and their relationships with the performance of an activated sludge plant in China. J Environ Sci 20:482–486. https://doi.org/10.1016/s1001-0742(08)62083-5
Funding
This project was supported by “Key Research and Development Plan of Anhui Province” (202004h07020027).
Author information
Authors and Affiliations
Contributions
Xiaobing Hu: visualization/conceptualization, methodology, writing—review, and editing. Kun Wang: investigation, data curation, writing—original draft, writing—review, and editing. Jing Chang, Lin Zhang, and Meiying Zhong: data collection. Yong Nie: review and editing. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The authors acknowledge that the current research has been conducted ethically. They declared that this manuscript does not involve researching about humans or animals.
Consent to participate
The authors consent to participate in this research study.
Consent to publish
The authors consent to publish the current research in ESPR journal.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 226 kb)
Rights and permissions
About this article
Cite this article
Hu, X., Wang, K., Chang, J. et al. Establishment of a comprehensive analysis method for the microfaunal movement in activated sludge. Environ Sci Pollut Res 28, 17084–17097 (2021). https://doi.org/10.1007/s11356-020-12090-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-12090-3