Abstract
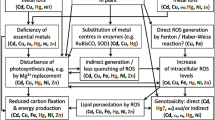
Phragmites australis, which is widely distributed throughout the world, is often used in the phytoremediation of acid mine drainage (AMD) due to its various mechanisms for survival under extremely harsh conditions. To explore the different responses of different aerial organs of P. australis to stress, soil and plant samples were collected from the AMD-polluted area of the Tongling mining area. The contents of manganese (Mn), iron (Fe), copper (Cu), zinc (Zn), arsenic (As), cadmium (Cd), and lead (Pb) in the soil and the leaf blades, leaf sheaths, and stems of P. australis as well as the contents/activities of cysteine synthase (CSase), superoxide dismutase (SOD), peroxidase (POD), glutathione (GSH), malondialdehyde (MDA), and proline (Pro) in the organs were determined. Our results revealed that the leaf sheath had the highest potential to store metals of all the organs. The highest translocation factor (TF) for Fe was observed from the stems to the leaf sheaths. A higher bioconcentration factor (BCF) for Mn was found in the leaf blades and leaf sheaths, while higher BCFs for Cd and Zn were observed in the stems. The content/activity of enzymatic and non-enzymatic stress-resistance substances varied from organ to organ. In general, the leaf sheaths remained almost as or slightly less stress-resistant than the leaf blades. It can be concluded that different plant organs play different roles in stress resistance, and understanding the tolerance mechanism of leaf sheaths to metals is essential for the application of phytoremediation procedures.

Similar content being viewed by others
References
Afshan S, Ali S, Bharwana SA, Rizwan M, Farid M, Abbas M, Ibrahim F, Mehmood MA, Abbasi GH (2015) Citric acid enhances the phytoextraction of chromium, plant growth and photosynthesis by alleviating the oxidative damages in Brassica napus L. Environ Sci Pollut Res 22:11679–11689. https://doi.org/10.1007/s11356-015-4396-8
Akcil A, Koldas S (2006) Acid Mine Drainage (AMD): Causes, treatment and case studies. J Clean Prod 14:1139–1145. https://doi.org/10.1016/j.jclepro.2004.09.006
Alfadul SMS, Al-Fredan MAA (2013) Effects of Cd, Cu, Pb, and Zn combinations on Phragmites australis metabolism, metal accumulation and distribution. Arab J Sci Eng 38:11–19. https://doi.org/10.1007/s13369-012-0393-0
Ashfaque F, Khan MIR, Khan NA (2014) Exogenously applied H2O2 promotes proline accumulation, water relations, photosynthetic efficiency and growth of wheat (Triticum aestivum L.) under salt stress. Annu Res Rev Biol 4:105–120. https://doi.org/10.9734/ARRB/2014/5629
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59(2):206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Baycu G, Tolunay D, Özden H, Guenenebakan S (2006) Ecophysiological and seasonal variations in Cd, Pb, Zn and Ni concentrations in the leaves of urban deciduous trees in Istanbul. Environ Pollut 143:545–554. https://doi.org/10.1016/j.envpol.2005.10.050
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bernardini A, Salvatori E, Guerrini V, Fusaro L, Canepari S, Manes F (2016) Effects of high Zn and Pb concentrations on Phragmites australis (Cav.) Trin. Ex. Steudel: Photosynthetic performance and metal accumulation capacity under controlled conditions. Int J Phytoremediat 18(1):16–24. https://doi.org/10.1080/15226514.2015.1058327
Borland AM, Farrar JF (1989) The partitioning of photosynthetically fixed carbon in the leaf blade and leaf sheath of Poa pratensis L. J Exp Bot 40(11):1247–1254. https://doi.org/10.1093/jxb/40.11.1247
Bragato C, Schiavon M, Polese R, Ertani A, Pittarello M, Malagoli M (2009) Seasonal variations of Cu, Zn, Ni and Cr concentration in Phragmites australis (Cav.) Trin ex steudel in a constructed wetland of North Italy. Desalination 246:35–44. https://doi.org/10.1016/j.desal.2008.02.036
Chauhan DK, Tripathi DK, Rai NK (2011) Detection of biogenic silica in leaf blade, leaf sheath, and stem of bermuda grass (Cynodon dactylon) using LIBS and phytolith analysis. Food Biophys 6(3):416–423. https://doi.org/10.1007/s11483-011-9219-y
Chen Y, Cheng JJ, Creamer KS (2008) Inhibition of anaerobic digestion process: a review. Bioresour Technol 99:4044–4064. https://doi.org/10.1007/s11483-011-9219-y
Chen F, Wang S, Mou S, Azimuddin I, Zhang D, Pan X (2015) Physiological responses and accumulation of heavy metals and arsenic of Medicago sativa L. growing on acidic copper mine tailings in arid lands. J. Geochem Explor 157:27–35. https://doi.org/10.1016/j.gexplo.2015.05.011
Cicero-Fernández D, Peña-Fernández M, Expósito-Camargo JA, Antizar-Ladislao B (2017) Long-term (two annual cycles) phytoremediation of heavy metal-contaminated estuarine sediments by Phragmites australis. New Biotechnol 38:56–64. https://doi.org/10.1016/j.nbt.2016.07.011
Civello PM, Martinez GA, Chaves AR, Anon MC (1995) Peroxidase from strawberry fruit (Fragaria ananassa Duch.): partial purification and determination of some properties. J Agric Food Chem 43:2596–2601. https://doi.org/10.1021/jf00058a008
Duman F, Cicek M, Sezen G (2007) Seasonal changes of metal accumulation and distribution in common club rush (Schoenoplectus lacustris) and common reed (Phragmites australis). Ecotoxicol. 16:457–463. https://doi.org/10.1007/s10646-007-0150-4
Esmaeilzadeh M, Karbassi A, Bastami KD (2017) Antioxidant response to metal pollution in Phragmites australis from Anzali wetland. Mar Pollut Bull 119:376–380. https://doi.org/10.1016/j.marpolbul.2017.03.030
Estebana JP, Escolásticoa C, Fernándezb JR, Masaguerc A, Molinerc A (2013) Bioavailability and extraction of heavy metals from contaminated soil by Atriplex halimus. Environ Exp Bot 88:53–59. https://doi.org/10.1016/j.envexpbot.2011.12.003
Ford KL (2003) Passive treatment systems for acid mine drainage. U.S. Department of the Interior, Bureau of Land Management, Technical (April), 20. BLM/ST/ST-02/001 + 3596
Habiba U, Ali S, Farid M, Shakoor MB, Rizwan M, Ibrahim M, Abbasi GH, Hayat T, Ali B (2015) EDTA enhanced plant growth, antioxidant defence system and phytoextraction of copper by Brassica napus L. Environ Sci Pollut Res 22:1534–1544. https://doi.org/10.1007/s11356-014-3431-5
Hansel CM, Fendorf S, Sutton S, Newville M (2001) Characterization of Fe plaque and associated metals on the roots of mine-waste impacted aquatic plants. Environ Sci Technol 35:3863–3868. https://doi.org/10.1021/es0105459
Hladun KR, Parker DR, Trumble JT (2015) Cadmium, copper, and lead accumulation and bioconcentration in the vegetative and reproductive organs of raphanus sativus: implications for plant performance and pollination. J Chem Ecol 41(4):386–395. https://doi.org/10.1007/s10886-015-0569-7
Iannelli MA, Pietrini F, Fiorea L, Petrillib L, Massacci A (2002) Antioxidant response to cadmium in Phragmites australis plants. Plant Physiol Biochem 40:977–982. https://doi.org/10.1016/S0981-9428(02)01455-9
Jiang ZF, Huang SZ, Han YL, Zhao JZ, Fu JJ (2012) Physiological response of Cu and Cu mine tailing remediation of Paulownia fortunei (Seem) Hemsl. Ecotoxicology. 21:759–767. https://doi.org/10.1007/s10646-011-0836-5
Kandziora-Ciupa M, Nadgórska-Socha A, Barczyk G, Ciepal R (2017) Bioaccumulation of heavy metals and ecophysiological responses to heavy metal stress in selected populations of Vaccinium myrtillus L. and Vaccinium vitis-idaea L. Ecotoxicology. 26:966–980. https://doi.org/10.1007/s10646-017-1825-0
Lal N (2010) Molecular mechanisms and genetic basis of heavy metal toxicity and tolerance in plants. In: Ashraf M, Ozturk M, Ahmad M (eds) Plant Adaptation and Phytoremediation. Springer, Dordrecht
Lou Y, Zhao P, Wang D, Amombo E, Sun X, Wang HH (2017) Germination, physiological responses and gene expression of tall fescue (Festuca arundinacea Schreb.) growing under Pb and Cd. PLoS One 12(1):e0169495. https://doi.org/10.1371/journal.pone.0169495
Mittler R, Vanderauwera S, Gollery M, Breusegem FV (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498. https://doi.org/10.1016/j.tplants.2004.08.009
Nadgórska-Socha A, Ptasiński B, Kita A (2013) Heavy metal bioaccumulation and antioxidative responses in Cardaminopsis arenosa and Plantago lanceolata leaves from metalliferous and non-metalliferous sites: a field study. Ecotoxicology 22(9):1422–1434. https://doi.org/10.1007/s10646-013-1129-y
Nelson DW, Sommers LE (1982) Part 2. Total carbon, organic carbon, and organic matter. In: Methods of soil analysis. American Society of Agronomy, Madison, pp 539–579
Parlak KU, Yilmaz DD (2012) Response of antioxidant defences to Zn stress in three duckweed species. Ecotoxicol Environ Saf 85:52–58. https://doi.org/10.1016/j.ecoenv.2012.08.023
Parzych A, Sobisz Z, Cymer M (2016) Preliminary research of heavy metals content in aquatic plants taken from surface water (Northern Poland). Desalin Water Treat 57:1451–1461. https://doi.org/10.1080/19443994.2014.1002275
Pietrini F, Iannelli MA, Pasqualini S, Massacci A (2003) Interaction of cadmium with glutathione and photosynthesis in developing leaves and chloroplasts of Phragmites australis (Cav.) Trin. ExSteudel. Plant Physiol 133(2):829–837. https://doi.org/10.1104/pp.103.026518
Qiao XQ, Wang PH, Shi GX, Yang HY (2015) Zinc conferred cadmium tolerance in Lemna minor L. via modulating polyamines and proline metabolism. Plant Growth Regul 77(1):1–9. https://doi.org/10.1007/s10725-015-0027-0
Qin F, Liu G, Huang GQ, Dong TF, Liao YM, Xu X (2017) Zinc application alleviates the adverse effects of lead stress more in female Morus alba than in males. Environ Exp Bot 146:68–76. https://doi.org/10.1016/j.envexpbot.2017.10.003
Rajinder SD, Dhindsa PP, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32:93–101. https://doi.org/10.1093/jxb/32.1.93
Rocha AS, Marisa R, Almeidab M, Clara P, Bastoa M, Teresa SD (2014) Antioxidant response of Phragmites australis to Cu and Cd contamination. Ecotoxicol Environ Saf 109:152–160. https://doi.org/10.1016/j.ecoenv.2014.06.027
Shen X, Li R, Chai M, Cheng S, Niu Z (2018) Interactive effects of single, binary and trinary trace metals (lead, zinc and copper) on the physiological responses of Kandelia obovata seedlings. Environ Geochem Health 41:135–148. https://doi.org/10.1007/s10653-018-0142-8
Sheoran AS, Sheoran V, Choudhary RP (2010) Bioremediation of acid-rock drainage by sulphate-reducing prokaryotes: a review. Min Eng 23(14):1073–1100. https://doi.org/10.1016/j.mineng.2010.07.001
Simate GS, Ndlovu S (2014) Acid mine drainage: challenges and opportunities. J Environ Chem Eng 2(3):1785–1803. https://doi.org/10.1016/j.jece.2014.07.021
Singha LP, Sinha N, Pandey P (2018) Rhizoremediation prospects of polyaromatic hydrocarbon degrading rhizobacteria, that facilitate glutathione and glutathione-S-transferase mediated stress response, and enhance growth of rice plants in pyrene contaminated soil. Ecotoxicol Environ Saf 164:579–588. https://doi.org/10.1016/j.ecoenv.2018.08.069
Torre-González AD, Montesinos-pereira D, Blasco B, Ruiz JM (2018) Analysis of metabolic and nutritional biomarkers in Brassica oleracea L. cv. Bronco plants under alkaline stress. J Hortic Sci Biotechnol:279–288. https://doi.org/10.1080/14620316.2017.1364979
Ünyayar S, Çelik A, Çekiç F, Gözel A (2006) Cadmium-induced genotoxicity, cytotoxicity and lipid peroxidation in Allium sativum and Vicia faba. Mutagenesis. 21:77–81. https://doi.org/10.1093/mutage/gel001
Verma S, Dubey RS (2003) Lead toxicity induces lipid peroxidation and alters the activities of antioxidant enzymes in growing rice plants. Plant Sci (Oxford) 164(4):645–655. https://doi.org/10.1016/S0168-9452(03)00022-0
Vymazal J (2016) Concentration is not enough to evaluate accumulation of heavy metals and nutrients in plants. Sci Total Environ 544:495–498. https://doi.org/10.1016/j.scitotenv.2015.12.011
Vymazal J, Březinová T (2016) Accumulation of heavy metals in aboveground biomass of Phragmites australis in horizontal flow constructed wetlands for wastewater treatment: a review. Chem Eng J 290:232–242. https://doi.org/10.1016/j.cej.2015.12.108
Warrilow AG, Hawkesford MJ (1998) Separation, subcellular location and influence of sulphur nutrition on isoforms of cysteine synthase in spinach. J Exp Bot 49:1625–1636. https://doi.org/10.1093/jxb/49.327.1625
Warrilow AG, Hawkesford MJ (2000) Cys synthase (O-acetylserine (thiol) lyase) substrate specificities classify the mitochondrial isoform as a cyanoalanine synthase. J Exp Bot 51:985–993. https://doi.org/10.1093/jexbot/51.347.985
Weis JS, Glover T, Weis P (2004) Interactions of metals affect their distribution in tissues of Phragmites australis. Environ Pollut 131:409–415. https://doi.org/10.1016/j.envpol.2004.03.006
Whitehead PG, Prior H (2005) Bioremediation of acid mine drainage: an introduction to the Wheal Jane wetlands project. Sci Total Environ 338(1-2):15–21. https://doi.org/10.1016/j.scitotenv.2004.09.016
Xu J, Zhang J, Xie H, Li C, Bao N, Zhang C, Shi Q (2010) Physiological responses of Phragmites australis to wastewater with different chemical oxygen demands. Ecol Eng 36:1341–1347. https://doi.org/10.1016/j.ecoleng.2010.06.010
Zouari M, Ben AC, Zorrig W, Elloumi N, Rabhi M, Delmail D, Ben RB, Labrousse P, Ben AF (2016) Exogenous proline mediates alleviation of cadmium stress by promoting photosynthetic activity, water status and antioxidative enzymes activities of young date palm (Phoenix dactylifera L.). Ecotoxicol Environ Saf 128:100–108. https://doi.org/10.1016/j.ecoenv.2016.02.015
Acknowledgments
The first author thanks Haojie Zhang for her great help in sampling sites map.
Funding
This study was funded by the Key Research and Development Projects in Anhui Province (Grant No. 201904a020027), and the Major Program for Anhui Agricultural Ecological Environmental Protection and Quality Safety Industrial Technology System (Grant No. AHCYJSTX-15). The authors received subsidization from the Personnel Training Project of the Mining Environmental Restoration and Wetlands Ecological Security Collaborative Innovation Center, Anhui University.
Author information
Authors and Affiliations
Contributions
Ziwei Ding made substantive contributions to the experimental processing and data interpreting and wrote the paper; Umar Daraz wrote part of the paper and provided language help. Qingye Sun planned the study and finalized the final draft of manuscript; Qing Fang performed part of the experiments. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 38 kb)
Rights and permissions
About this article
Cite this article
Ding, Z., Fang, Q., Daraz, U. et al. Physiological responses and metal distributions of different organs of Phragmites australis shoots under acid mine drainage stress. Environ Sci Pollut Res 28, 3375–3385 (2021). https://doi.org/10.1007/s11356-020-10700-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-10700-8