Abstract
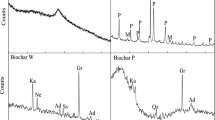
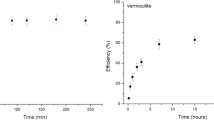
In this study, we report for the first time a novel type of sorbent that can be used for mercury adsorption from the air-based off-gasses—vermiculite impregnated with alkali polysulfides and thiosulfates. In contrast to other sorbents, vermiculite exhibits superior thermal stability in air and low adsorption capacity for organic vapors. This allows for a more favorable design of the soil remediation unit—direct coupling of thermal desorber with catalytic oxidizer using air as a carrier gas. In the bench-scale test at 180 °C, the sulfur/vermiculite sorbent exhibited significantly higher efficiency for the adsorption of mercury vapor from the off-gasses than the commercial sulfur/activated carbon sorbent at its highest operating temperature (120 °C). The average mercury concentration in the adsorber off-gas decreased from 1.634 mg/m3 for the sulfur/activated carbon to 0.008 mg/m3 achieved with impregnated vermiculite. The total concentration of organic compounds in the soil after thermal desorption was below the detection limit of the employed analytical method.







Similar content being viewed by others
References
Balasundaram K, Sharma M (2018) Concurrent removal of elemental mercury and SO2 from flue gas using a thiol-impregnated CaCO3-based adsorbent: a full factorial design study. Environ Sci Pollut Res Int 25:15518–15528. https://doi.org/10.1007/s11356-018-1672-4
Belozovski AB, Balandinal EA (1978) Removing mercury from industrial gases-by sorption on activated carbon impregnated with ferric chloride for high capacity and safety. US Patent No. 625752
Bertinchamps F, Attianese A, Mestdagh MM, Gaigneaux EM (2006) Catalysts for chlorinated VOCs abatement: multiple effects of water on the activity of VOX based catalysts for the combustion of chlorobenzene. Catal Today 112:165–168. https://doi.org/10.1016/j.cattod.2005.11.043
Gan S, Lau EV, Ng HK (2009) Remediation of soils contaminated with polycyclic aromatic hydrocarbons (PAHs). J Hazard Mater 172:532–549. https://doi.org/10.1016/j.jhazmat.2009.07.118
Ghorishi B, Gullett BK (1998) Sorption of mercury species by activated carbons and calcium-based sorbents: effect of temperature, mercury concentration and acid gases. Waste Manag Res 16:582–593. https://doi.org/10.1177/0734242X9801600609
Hou D, Gu Q, Ma F, O'Connell S (2016) Life cycle assessment comparison of thermal desorption and stabilization/solidification of mercury contaminated soil on agricultural land. J Clean Prod 139:949–956. https://doi.org/10.1016/j.jclepro.2016.08.108
Hsi HC, Rood MJ, Asce M, Rostam-Abadi M, Chen S, Chang R (2002) Mercury adsorption properties of sulfur-impregnated adsorbents. J Environ Eng 128:1080–1089. https://doi.org/10.1061/(ASCE)0733-9372(2002)128:11(1080)
Ie IR, Hung CH, Jen YS, Yuan CS, Chen WH (2013) Adsorption of vapor-phase elemental mercury (Hg0) and mercury chloride (HgCl2) with innovative composite activated carbons impregnated with Na2S and S0 in different sequences. Chem Eng J 229:469–476. https://doi.org/10.1016/j.cej.2013.06.059
Karatza D, Lancia A, Musmarra D, Zucchini C (2000) Study of mercury absorption and desorption on sulfur impregnated carbon. Exp Therm Fluid Sci 21:150–155. https://doi.org/10.1016/S0894-1777(99)00065-5
Kaštánek F, Topka P, Soukup K, Maléterová Y, Demnerová K, Kaštánek P, Šolcová O (2016) Remediation of contaminated soils by thermal desorption; effect of benzoyl peroxide addition. J Clean Prod 125:309–313. https://doi.org/10.1016/j.jclepro.2016.03.134
Khan FI, Husain T, Hejazi R (2004) An overview and analysis of site remediation technologies. J Environ Manag 71:95–122. https://doi.org/10.1016/j.jenvman.2004.02.003
Korpiel JA, Vidic RD (1997) Effect of sulphur impregnation method on activated carbon uptake of gas-phase mercury. Environ Sci Technol 31:2319–2325. https://doi.org/10.1021/es9609260
Lee SH, Park YO (2003) Gas-phase mercury removal by carbon-based sorbents. Fuel Process Technol 84:197–206. https://doi.org/10.1016/S0378-3820(03)00055-9
Li C, Tang H, Duan Y, Zhu C, Zheng Y, Huang T (2018) Synthetic calcium-based adsorbents for gaseous mercury(II) adsorption from flue gas and study on their mercury adsorption mechanism. Fuel 234:384–391. https://doi.org/10.1016/j.fuel.2018.06.135
Liu W, Vidic RD, Brown TD (2000) Optimization of high temperature sulphur impregnation on activated carbon for permanent separation of elementary mercury vapours. Environ Sci Technol 34:483–488. https://doi.org/10.1021/es9813008
Mokhatab S, Poe WA (2012) Handbook of natural gas transmission and processing. Gulf Professional Publishing, Waltham. https://doi.org/10.1016/C2013-0-15625-5
Nippon Soda Co. Ltd (1974) Activated carbon contg. halides for mercury extn.-from air or gases, with excellent adsorptivity. JP Patent No. 49053590
Ossai IC, Ahmed A, Hassan A, Fauziah SH (2020) Remediation of soil and water contaminated with petroleum hydrocarbon: a review. Environ Technol Innov 17:100526. https://doi.org/10.1016/j.eti.2019.100526
Prasad R, Kennedy LA, Ruckenstein E (1984) Catalytic combustion. Catal Rev 26:1–58. https://doi.org/10.1080/01614948408078059
Sano A, Takaoka M, Shiota K (2017) Vapor-phase elemental mercury adsorption by activated carbon co-impregnated with sulfur and chlorine. Chem Eng J 315:598–607. https://doi.org/10.1016/j.cej.2017.01.035
Šolcová O, Matějová L, Topka P, Musilová Z, Schneider P (2011) Comparison of textural information from argon (87 K) and nitrogen (77 K) physisorption. J Porous Mater 18:557–565. https://doi.org/10.1007/s10934-010-9409-x
Šolcová O, Topka P, Soukup K, Jirátová K, Váňová H, Kaštánek F (2014) Solid waste decontamination by thermal desorption and catalytic oxidation methods. Chem Pap 68:1279–1282. https://doi.org/10.2478/s11696-013-0455-0
Soukup K, Petráš D, Topka P, Slobodian P, Šolcová O (2012) Preparation and characterization of electrospun poly(p-phenylene oxide) membranes. Catal Today 193:165–171. https://doi.org/10.1016/j.cattod.2012.03.019
Soukup K, Topka P, Hejtmánek V, Petráš D, Valeš V, Šolcová O (2014) Noble metal catalysts supported on nanofibrous polymeric membranes for environmental applications. Catal Today 236:3–11. https://doi.org/10.1016/j.cattod.2014.03.040
Sumitomo Chemical Co. (1974) Carbon absorbent for removing mercury vapor from air-carrying at least one chloride of alkali or transition metal. JP Patent No. 49066592
Svoboda K, Hartman M, Šyc M, Pohořelý M, Kameníková P, Jeremiáš M, Durda T (2016) Possibilities of mercury removal in the dry flue gas cleaning lines of solid waste incineration units. J Environ Manag 166:499–511. https://doi.org/10.1016/j.jenvman.2015.11.001
Thornton G, Franz M, Edwards D, Pahlen G, Nathanail P (2007) The challenge of sustainability: incentives for brownfield regeneration in Europe. Environ Sci Policy 10:116–134. https://doi.org/10.1016/j.envsci.2006.08.008
Tomatis M, Moreira MT, Xu H, Deng W, He J, Parvez AM (2019) Removal of VOCs from waste gases using various thermal oxidizers: a comparative study based on life cycle assessment and cost analysis in China. J Clean Prod 233:808–818. https://doi.org/10.1016/j.jclepro.2019.06.131
Tomlin CDS (ed.) (1997) The pesticide manual-world compendium, 11th, British Crop Protection Council, Surrey, England. ISBN 1901396118
Topka P, Klementová M (2016) Total oxidation of ethanol over Au/Ce0.5Zr0.5O2 cordierite monolithic catalysts. Appl Catal A 522:130–137. https://doi.org/10.1016/j.apcata.2016.05.004
Topka P, Kaluža L, Gaálová J (2016) Total oxidation of ethanol and toluene over ceria-zirconia supported platinum catalysts. Chem Pap 70:898–906. https://doi.org/10.1515/chempap-2016-0028
Topka P, Hejtmánek V, Cruz GJF, Šolcová O, Soukup K (2019) Activated carbon from renewable material as an efficient support for palladium oxidation catalysts. Chem Eng Technol 42:851–858. https://doi.org/10.1002/ceat.201800611
Topka P, Dvořáková M, Kšírová P, Perekrestov R, Čada M, Balabánová J, Kostejn M, Jirátová K, Kovanda F (2020) Structured cobalt oxide catalysts for VOC abatement: the effect of preparation method. Environ Sci Pollut Res 27:7608–7617. https://doi.org/10.1007/s11356-019-06974-2
Vidonish JE, Zygourakis K, Masiello CA, Sabadell G, Alvarez PJJ (2016) Thermal treatment of hydrocarbon-impacted soils: a review of technology innovation for sustainable remediation. Engineering 2:426–437. https://doi.org/10.1016/J.ENG.2016.04.005
Xie Y, Li C, Zhao L, Zhang J, Zeng G, Zhang X, Zhang W, Tao S (2015) Experimental study on Hg0 removal from flue gas over columnar MnOx–CeO2/activated coke. Appl Surf Sci 333:59–67. https://doi.org/10.1016/j.apsusc.2015.01.234
Xu J, Bravo AG, Lagerkvist A, Bertilsson S, Sjöblom R, Kumpiene J (2015) Sources and remediation techniques for mercury contaminated soil. Environ Int 74:42–53. https://doi.org/10.1016/j.envint.2014.09.007
Yan R, Liang DT, Tsen L, Wong YP, Lee YK (2004) Bench-scale experimental evaluation of carbon performance on mercury vapour adsorption. Fuel 83:2401–2409. https://doi.org/10.1016/j.fuel.2004.06.031
Zhao C, Dong Y, Feng Y, Li Y, Dong Y (2019) Thermal desorption for remediation of contaminated soil: a review. Chemosphere 221:841–855. https://doi.org/10.1016/j.chemosphere.2019.01.079
Zheng Y, Jensen AD, Windelin C, Jensen F (2012) Dynamic measurement of mercury adsorption and oxidation on activated carbon in simulated cement kiln flue gas. Fuel 93:649–657. https://doi.org/10.1016/j.fuel.2011.09.053
Funding
The financial support of the Technology Agency of the Czech Republic (project No. TA04020700) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Paper dedicated to Professor František Kaštánek on the occasion of his 85th birthday
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Topka, P., Soukup, K., Hejtmánek, V. et al. Remediation of brownfields contaminated by organic compounds and heavy metals: a bench-scale test of a sulfur/vermiculite sorbent for mercury vapor removal. Environ Sci Pollut Res 27, 42182–42188 (2020). https://doi.org/10.1007/s11356-020-10696-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-10696-1