Abstract
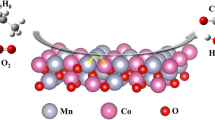
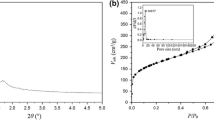
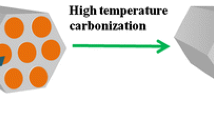
Co-Fe, Cu-Cr, and Co-Mn mixed oxide catalysts were prepared using a one-pot hard template synthesis method, and their catalytic performance was investigated before and after the rearrangement of the template. To evaluate the structural properties of the catalysts, various analyses were employed, including the BET, XRD, H2-TPR, FE-SEM, EDX, and X-ray digital mapping of the elements. The results indicated that the rearrangement of the catalyst structure had a profound effect on the structural and catalytic properties, so that in all three synthesized catalysts, the specific surface and the reducibility increased significantly, and the crystalline structure and morphology of the catalysts changed remarkably. The specific surface area of the CoFe, CuCr, and CoMn catalysts increased from 3.5, 1.1, and 72.9 m2/g to 151.3, 52.8, and 108.0 m2/g, respectively. These structural changes significantly increased the catalytic performance. The results indicated that the 100% conversion temperature of the CoMn catalyst as the optimal sample after rearrangement was reduced from 250 to 125 °C. Also, the stability of the CoMn catalyst in dry and wet conditions was investigated and the results indicated that the presence of water vapor reduced the activity and stability of the catalyst. The activation energy was also calculated on Co-Mn catalyst (59.5 kJ/mol) and the results confirmed that the most probable mechanism for this reaction was the MVK mechanism.













Similar content being viewed by others
References
Arandiyan H, Dai H, Deng J, Liu Y, Bai B, Wang Y, Li X, Xie S, Li J (2013) Three-dimensionally ordered macroporous La0.6Sr0.4MnO3 with high surface areas: active catalysts for the combustion of methane. J Catal 307:327–339. https://doi.org/10.1016/j.jcat.2013.07.013
Bai B, Arandiyan H, Li J (2013) Comparison of the performance for oxidation of formaldehyde on nano-Co3O4, 2D-Co3O4, and 3D-Co3O4 catalysts. Appl Catal B Environ 142–143:677–683. https://doi.org/10.1016/j.apcatb.2013.05.056
Bai B, Qiao Q, Arandiyan H, Li J, Hao J (2016) Three-dimensional ordered mesoporous MnO2-supported Ag nanoparticles for catalytic removal of formaldehyde. Environ Sci Technol 50:2635–2640. https://doi.org/10.1021/acs.est.5b03342
Biabani-Ravandi A, Rezaei M (2012) Low temperature CO oxidation over Fe-co mixed oxide nanocatalysts. Chem Eng J 184:141–146. https://doi.org/10.1016/j.cej.2012.01.017
Bulavchenko OA, Gerasimov EY, Afonasenko TN (2018) Reduction of double manganese–cobalt oxides: in situ XRD and TPR study †. Dalt Trans 47:17153–17159. https://doi.org/10.1039/C8DT04137G
Chang LH, Sasirekha N, Rajesh B, Chen YW (2007) CO oxidation on ceria- and manganese oxide-supported gold catalysts. Sep Purif Technol 58:211–218. https://doi.org/10.1016/j.seppur.2007.07.031
Deng Y, Wang T, Zhu L, Jia AP, Lu JQ, Luo MF (2018) Enhanced performance of CO oxidation over Pt/CuCrOx catalyst in the presence of CO2 and H2O. Appl Surf Sci 442:613–621. https://doi.org/10.1016/j.apsusc.2018.02.099
Doi Y, Takai A, Sakamoto Y, Terasaki O, Yamauchi Y, Kuroda K (2010) Tailored synthesis of mesoporous platinum replicas using double gyroid mesoporous silica (KIT-6) with different pore diameters via vapor infiltration of a reducing agent. Chem Commun 46:6365–6367. https://doi.org/10.1039/c0cc01196g
Fan S, Wang W, Ke H, Rao JC, Zhou Y (2016) Bimagnetic urchin-like Co3O4/CoFe2O4 nanocomposites: synthesis and magnetic properties. RSC Adv 6:97055–97062. https://doi.org/10.1039/C6RA18846J
Faure B, Alphonse P (2015) Co-Mn-oxide spinel catalysts for CO and propane oxidation at mild temperature. Appl Catal B Environ 180:715–725. https://doi.org/10.1016/j.apcatb.2015.07.019
Hamidreza A, Wang Y, Sun H et al (2018) Ordered meso- and macroporous perovskite oxide catalysts for emerging applications. Chem Commun 54:6484–6502. https://doi.org/10.1039/C8CC01239C
Hammoudeh A, Naschitzki M (1999a) Field ion microscopy study of CO oxidation on palladium field emitter: field effects and imaging mechanism. Mater Sci Eng A 270:89–93. https://doi.org/10.1016/S0921-5093(99)00246-4
Hammoudeh A, Naschitzki M (1999b) CO oxidation on palladium field emitter: kinetic oscillations and bistability. Mater Sci Eng A 270:83–88. https://doi.org/10.1016/S0921-5093(99)00245-2
Han SW, Kim DH, Jeong MG, Park KJ, Kim YD (2016) CO oxidation catalyzed by NiO supported on mesoporous Al2O3 at room temperature. Chem Eng J 283:992–998. https://doi.org/10.1016/j.cej.2015.08.021
Jozwiak WK, Kaczmarek E, Maniecki TP, Ignaczak W, Maniukiewicz W (2007) Reduction behavior of iron oxides in hydrogen and carbon monoxide atmospheres. Appl Catal A Gen 326:17–27. https://doi.org/10.1016/j.apcata.2007.03.021
Khedr MH, Halim KSA, Nasr MI, El-Mansy AM (2006) Effect of temperature on the catalytic oxidation of CO over nano-sized iron oxide. Mater Sci Eng A 430:40–45. https://doi.org/10.1016/j.msea.2006.05.119
Leofanti G, Padovan M, Tozzola G, Venturelli B (1998) Surface area and pore texture of catalysts. Catal Today 41:207–219. https://doi.org/10.1016/S0920-5861(98)00050-9
Li M, Wang DH, Shi XC, Zhang ZT, Dong TX (2007) Kinetics of catalytic oxidation of CO over copper-manganese oxide catalyst. Sep Purif Technol 57:147–151. https://doi.org/10.1016/j.seppur.2007.03.016
Liang C, Ma Z, Ding L, Qiu J (2009) Template preparation of highly active and selective cu-Cr catalysts with high surface area for glycerol hydrogenolysis. Catal Letters 130:169–176. https://doi.org/10.1007/s10562-009-9844-y
Liu W, Flytzani-stephanopoulos M (1995) Total oxidation of carbon-monoxide and methane over transition metal fluorite oxide composite catalysts: II. Catalyst characterization and reaction-kinetics. J Catal 153:317–332
Liu Y, Dai H, Deng J, Li X, Wang Y, Arandiyan H, Xie S, Yang H, Guo G (2013) Au/3DOM La0.6Sr0.4MnO3: highly active nanocatalysts for the oxidation of carbon monoxide and toluene. J Catal 305:146–153. https://doi.org/10.1016/j.jcat.2013.04.025
Lv L, Xu Q, Ding R, Qi L, Wang H (2013) Chemical synthesis of mesoporous CoFe2O4 nanoparticles as promising bifunctional electrode materials for supercapacitors. Mater Lett 111:35–38. https://doi.org/10.1016/j.matlet.2013.08.055
Ma L, Seo CY, Chen X, Sun K, Schwank JW (2018) Indium-doped Co3O4 nanorods for catalytic oxidation of CO and C3H6 towards diesel exhaust. Appl Catal B Environ 222:44–58. https://doi.org/10.1016/j.apcatb.2017.10.001
Mobini S, Meshkani F, Rezaei M (2017a) Synthesis and characterization of nanocrystalline copper–chromium catalyst and its application in the oxidation of carbon monoxide. Process Saf Environ Prot 107:181–189. https://doi.org/10.1016/j.psep.2017.02.009
Mobini S, Meshkani F, Rezaei M (2017b) Surfactant-assisted hydrothermal synthesis of CuCr2O4 spinel catalyst and its application in CO oxidation process. J Environ Chem Eng 5:4906–4916. https://doi.org/10.1016/j.jece.2017.09.027
Mobini S, Meshkani F, Rezaei M (2019) Supported Mn catalysts and the role of different supports in the catalytic oxidation of carbon monoxide Chem Eng Sci 37–51. https://doi.org/10.1016/j.ces.2018.12.006
Natile MM, Glisenti A (2003) New NiO/Co3O4 and Fe2O3/Co3O4 nanocomposite catalysts: synthesis and characterization. Chem Mater 15:2502–2510. https://doi.org/10.1021/cm031019e
Roy S, Marimuthu A, Hegde MS, Madras G (2007) High rates of NO and N2O reduction by CO, CO and hydrocarbon oxidation by O2 over nano crystalline Ce0.98Pd0.02O2-δ: catalytic and kinetic studies. Appl Catal B Environ 71:23–31. https://doi.org/10.1016/j.apcatb.2006.08.005
Royer S, Duprez D (2011) Catalytic oxidation of carbon monoxide over transition metal oxides. ChemCatChem 3:24–65. https://doi.org/10.1002/cctc.201000378
Schwartz WR, Pfefferle LD (2012) Combustion of methane over palladium-based catalysts: support interactions. J Phys Chem C 116:8571–8578. https://doi.org/10.1021/jp2119668
Sedmak G, Hocevar S, Levec J (2003) Kinetics of selective CO oxidation in excess of H2 over the nanostructured Cu0.1Ce0.9O2−y catalyst. J Catal 213:135–150
Shinde VM, Madras G (2012) Kinetics of carbon monoxide oxidation with Sn0.95M0.05O2-δ (M = Cu, Fe, Mn, Co) catalysts. Catal Sci Technol 2:437–446. https://doi.org/10.1039/c1cy00421b
Sing KSW, Everett DH, Haul RAW et al (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Tüysüz H, Salabaş EL, Bill E, Bongard H, Spliethoff B, Lehmann CW, Schüth F (2012) Synthesis of hard magnetic ordered mesoporous Co3O4/CoFe2O4 nanocomposites. Chem Mater 24:2493–2500. https://doi.org/10.1021/cm3005166
Valencia J, Arias NP, Giraldo O, Rosales-Rivera A (2012) Synthesis and characterization of cobalt-manganese oxides. Phys B Condens Matter 407:3155–3157. https://doi.org/10.1016/j.physb.2011.12.051
Wang C-B, Tang C-W, Tsai H-C, Chien S-H (2006) Characterization and catalytic oxidation of carbon monoxide over supported cobalt catalysts. Catal Letters 107:223–230. https://doi.org/10.1007/s10562-005-0002-x
Wang Y, Arandiyan H, Scott JA, Akia M, Dai H, Deng J, Aguey-Zinsou KF, Amal R (2016) High performance Au-Pd supported on 3D hybrid strontium-substituted lanthanum manganite perovskite catalyst for methane combustion high performance Au − Pd supported on 3D hybrid strontium-substituted lanthanum manganite perovskite catalyst for methane Co. ACS Catal 6:6935–6947. https://doi.org/10.1021/acscatal.6b01685
Wang Y, Arandiyan H, Scott J, Bagheri A, Dai H, Amal R (2017) Recent advances in ordered meso/macroporous metal oxides for heterogeneous catalysis: a review. J Mater Chem A 5:8825–8846. https://doi.org/10.1039/C6TA10896B
Wang Y, Yang D, Li S, Zhang L, Zheng G, Guo L (2019) Layered copper manganese oxide for the efficient catalytic CO and VOCs oxidation. Chem Eng J 357:258–268. https://doi.org/10.1016/j.cej.2018.09.156
Xie X, Li Y, Liu Z-Q, Haruta M, Shen W (2009) Low-temperature oxidation of CO catalysed by Co3O4 nanorods. Nature 458:746–749. https://doi.org/10.1038/nature07877
Xie S, Liu Y, Deng J, Zhao X, Yang J, Zhang K, Han Z, Arandiyan H, Dai H (2017) Effect of transition metal doping on the catalytic performance of Au–Pd/3DOM Mn2O3for the oxidation of methane and o-xylene. Appl Catal B Environ 206:221–232. https://doi.org/10.1016/j.apcatb.2017.01.030
Zhang X, Qu Z, Li X, Wen M, Quan X, Ma D, Wu J (2010) Studies of silver species for low-temperature CO oxidation on Ag/SiO2 catalysts. Sep Purif Technol 72:395–400. https://doi.org/10.1016/j.seppur.2010.03.012
Zhou M, Cai L, Bajdich M, García-Melchor M, Li H, He J, Wilcox J, Wu W, Vojvodic A, Zheng X (2015) Enhancing catalytic CO oxidation over Co3O4 nanowires by substituting Co2+ with Cu2+. ACS Catal 5:4485–4491. https://doi.org/10.1021/acscatal.5b00488
Funding
The authors received financial support from the Iran National Science Foundation (INSF) under the grant numbers of 97017638.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Santiago V. Luis
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mobini, S., Rezaei, M. & Meshkani, F. One-pot hard template synthesis of mesoporous spinel nanoparticles as efficient catalysts for low temperature CO oxidation. Environ Sci Pollut Res 28, 547–563 (2021). https://doi.org/10.1007/s11356-020-10398-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-10398-8