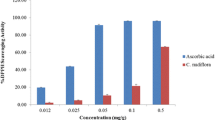
Abstract
Carbon nanotubes (CNTs) are extensively used in nanotechnology due to their unique physico-chemical properties. CNTs were implicated in many disorders connected with human health. So, we aimed in this study to provide new insight into the role of aqueous C. burmannii in treating the possible hepatotoxic effects of multi-walled carbon nanotube (MWCNTs) exposure. A total of 32 male albino rats were divided into 4 groups: control group, cinnamon-treated group, MWCNT-treated, and cinnamon- and MWCNT-treated group. To achieve the aim of this study, evaluation of percentage change of body weight, oxidant, and antioxidant status including lipid peroxidation (LPO), nitrite, total thiols, glutathione contents (GSH), the activity of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and glutathione-S transferase (GST) was done. Histopathological examination and the rate of pro-inflammatory cytokines including interleukin-6 (IL-6), interleukin-1β (IL-1β), cyclooxygenase-1 (COX-1), and tumor necrotic factor-α were performed. Oral administration of aqueous C. burmannii to those MWCNT-treated rats resulted in a significant reduction in LPO and total thiol contents with a significant elevation in the activities of SOD, CAT, and GPX, while GSH content and GST activity were not significantly affected. We observed a significant downregulation in the rate of previous pro-inflammatory cytokines. All this improvement in these examined markers resulted in a significant modulation in the hepatic histopathological lesions caused by MWCNTs. Aqueous C. burmannii extract exhibited a potential defensive effect on the hepatic injury triggered by MWCNTs through upgrading the antioxidant system and downregulating the rate of pro-inflammatory cytokines.





Similar content being viewed by others
References
Afifi NA, Ibrahim MA, Galal MK (2018) Hepatoprotective influence of quercetin and ellagic acid on thioacetamide-induced hepatotoxicity in rats. Can J Physiol Pharmacol 96(6):624–629
Albini A, Pagani A, Pulze L, Bruno A, Principi B, Congiu T, Gini E, Grimaldi A, Bassani B, De Flora S, de Eguileor M, Noonan DM (2015) Environmental impact of multi-wall carbon nanotubes in a novel model of exposure: systemic distribution, macrophage accumulation, and amyloid deposition. Int J Nanomedicine 10:6133–6145
Al-Dhubaib BE (2012) Pharmaceutical applications and phytochemical profile of Cinnamomum burmannii. Pharmacogn Rev 6(12):125–131
Ali GE, Ibrahim MA, El-Deeb AH, Amer H, Zaki SM (2019) Pulmonary deregulation of expression of miR-155 and two of its putative target genes; PROS1and TP53INP1 associated with gold nanoparticles (AuNPs) administration in rat. Int J Nanomedicine 14:5569–5579
Awasthi KK1, John PJ, Awasthi A, Awasthi K (2013) Multi walled carbon nano tubes induced hepatotoxicity in Swiss albino mice. Micron 44:359–64
Bai YH, Zhang Y, Zhang JP, Mu QX, Zhang WD, Butch ER, Snyder SE, Yan B (2010) Repeated administrations of carbon nanotubes in male mice cause reversible testis damage without affecting fertility. Nat Nanotechnol 5:683–689
Bancroft JD, Gamble M (2002) Theory and practice of histological techniques, 5th edn. Churchill Living stone Pub., Edinburgh
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Carmo ES, Lima ED, de Souza EL, de Sousa FB (2008) Effect of Cinnamomum zeylanicumblume essential oil on the growth and morphogenesis of some potentially pathogenic Aspergillus species. Braz J Microbiol 39:91–97
Clichici S, Biris AR, Tabaran F, Filip A (2012) Transient oxidative stress and inflammation after intra-peritoneal administration of multiwalled carbon nanotubes functionalized with single strand DNA in rats. Toxicol Appl Pharmacol 259(3):281–292
Cohen G, Dembiec D, Marcus J (1970) Measurement of catalase activity in tissue extracts. Anal Biochem 34:30–38
Colombo G, Reggiani F, Podestà MA, Garavaglia ML, Portinaro NM, Milzani A, Badalamenti S, Dalle-Donne I (2015) Plasma protein thiolation index (PTI) as a biomarker of thiol-specific oxidative stress in haemodialyzed patients. Free Radic Biol Med 89:443–451
Costello RB, Dwyer JT, Saldanha L, Bailey RL, Merkel J, Wambogo E (2016) Do cinnamon supplements have a role in glycemic control in type 2 diabetes? A narrative review. J Acad Nutr Diet 116(11):1794–1802
Cveticanin J, Joksic G, Leskovac A, Petrovic S, Sobot AV, Neskovic O (2010) Using carbon nanotubes to induce micronuclei and double strand breaks of the DNA in human cells. Nanotechnology 21 (1):015102.
Eidi A, Mortazavi P, Bazargan M, Zaringhalam J (2012) Activity of cinnamon ethanolic Extract against ccl4-induced liver injury in rats. EXCLI J 11:495–507
Hagenes, E. (2014) Genotoxic effects of exposure to carbon nanotubes in human lung cells. Master’s thesis in Toxicology, Faculty of Mathematics and Natural Sciences, Department of Biosciences. University of Oslo
Hamilton RFJ, Wu Z, Mitra S, Shaw PK, Holian A (2013) Effect of MWCNT size, carboxylation, and purification on in vitro and in vivo toxicity, inflammation and lung pathology. Part Fibre Toxicol 10(1):57
Hariri M, Ghiasvand R (2016) Cinnamon and chronic diseases. Adv Exp Med Biol 929:1–24
James SL, Glaven J (1989) Macrophage cytotoxicity against schistosomula of Schistosomamansoni involves arginine dependent production or reactive nitrogen intermediates. J Immunol 143:4208–4212
Kania N, Widowati W, Dewi F, Christianto A, Setiawan B, Budhiparama N, Noor Z (2018) Cinnamomum burmanini Blume increases bone turnover marker and induces tibia’s granule formation in ovariectomized rats. J Ayurveda Integr Med 9(1):20–26
Kermanizadeh A, Gaiser BK, Johnston H, Brown DM, Stone V (2014) Toxicological impact of engineered nanomaterials on the liver – a review. Br J Pharmacol 171:3980–3987
Khaki A (2015) Effect of Cinnamomum zeylanicumon on Spermatogenesis. Iran Red Crescent Med J 17(2):18668
Kido T, Tsunoda M, Kasai T, Sasaki T, Umeda Y, Senoh H, Yanagisawa H, Asakura M, Aizawa Y, Fukushima S (2014) The increases in relative mRNA expressions of inflammatory cytokines and chemokines in splenic macrophages from rats exposed to multi-walled carbon nanotubes by whole-body inhalation for 13 weeks. Inhal Toxicol 26(12):750–758
Koster JF, Biemond P, Swaak AJ (1986) Intra-cellular and extracellular sulphydryl levels in rheumatoid arthritis. Ann Rheum Dis 45(1):44–46
Li X, Mazaleuskaya LL, Ballantyne LL, Meng H, FitzGerald GA (2018) Funk CD Genomic and lipidomic analyses differentiate the compensatory roles of two COX isoforms during systemic inflammation in mice. J Lipid Res 59(1):102–112
Liang G, Yin L, Zhang J, Liu R, Zhang T, Ye B, Pu Y (2010) Effects of sub chronic exposure to multi-walled carbon nanotubes on mice. J Toxicol Environ Health A 73:463–470
Lim JH, Kim SH, Shin IS, Park NH, Moon C, Kang SS, Kim SH, Park SC, Kim JC (2011) Maternal exposure to multi-wall carbon nanotubes does not induce embryo–fetal developmental toxicity in rats. Birth Defects Res B Dev Reprod Toxicol 92:69–76
Mannervik B, Guthenberg C (1981) Glutathione transferase (human placenta). Methods Enzymol 77:231–235
Marklund S, Marklund G (1974) Involvement of the super-oxide anion radical in the autoxidation of pyrogallol and convenient assay for super-oxide dismutase. Eur J Biochem 47:469–474
Matkovics B, Kotorman M, Varga IS, Hai DQ, Varga C (1998) Oxidative stress in experimental diabetes induced by streptozotocin. Acta Physiol Hung 85:29–38
May PW (1995) CVD diamond—a new technology for the future. Endeavour 19(3):101–106
Mitchell LA, Lauer FT, Burchiel SW, McDonald JD (2009) Mechanisms for how inhaled multi-walled carbon nanotubes suppress systemic immune function in mice. Nat Nanotechnol 4:451–456
Morgan AM, Ibrahim MA, Hussien AM (2017) The potential protective role of akropower against atrazine induced humoral immunotoxicity in rabbits. Biomed Pharmacother 96:710–715
Paget GE, Barnes JM (1969) In: Lawrence DR, Bacharach AL (eds) Evaluation of drug activities, vol 1. Academic press, New York, p 161
Ranasinghe P, Galappaththy P, Constantine GR, Jayawardena R, Weeratunga HD, Premakumara S, Katulanda P (2017) Cinnamomum zeylanicum (Ceylon cinnamon) as a potential pharmaceutical agent for type-2 diabetes mellitus: study protocol for a randomized controlled trial. Trials 18(1):446
SPSS (1998) SPSS Base 8.0 for windows user’s guide. SPSS Inc., Chicago
Sharma M, Nikota J, Halappanavar S, Castranova V, Rothen-Rutishauser B, Clippinger A (2016) Predicting pulmonary fibrosis in humans after exposure to multi-walled carbon nanotubes (MWCNTs). Arch Toxicol 90(7):1605–1622
Sheng X, Zhang Y, Gong Z, Huang C, Zang YQ (2008) Improved insulin resistance and Lipid metabolism by cinnamon extract through activation of peroxisome proliferation activated receptors. PPAR Res. https://doi.org/10.1155/2008/581348
Srivastava RK, Pant AB, Kashyap MP, Kumar V, Lohani M, Jonas L, Rahman Q (2011) Multi-walled carbon nanotubes induce oxidative stress and apoptosis in human lung cancer cell line-A549. Nanotoxicology 5:195–207
Tabei Y, Fukui H, Nishioka A, Hagiwara Y, Sato K, Yoneda T, Koyama T, Horie M (2019) Effect of iron overload from multi walled carbon nanotubes on neutrophil-like differentiated HL-60 cells. Sci Rep 9(1):2224
Tung YT, Chua MT, Wang SY, Chang ST (2008) Antiinflammation activities of essential oil and its constituents from indigenous cinnamon (Cinnamomum osmophloeum) twigs. Bioresour Technol 99:3908–3913
Tuzcu Z, Orhan C, Sahin N, Juturu V, Sahin K (2017) Cinnamon polyphenol extract inhibits hyperlipidemia and inflammation by modulation of transcription factors in high-fat diet-fed rat. Oxid Med Cell Longev 2017:1583098
Vinches L, Hallé S (2017) Resistance of Type 5 chemical protective clothing against nanometric airborne particles: Behavior of seams and zipper. J Occup Environ Hyg 14(12):939–946
Wadley AJ, Veldhuijzen van Zanten JJCS, Aldred S (2012) The interactions of oxidative stress and inflammation with vascular dysfunction in ageing: the vascular health triad. Age 35:705–718
Yadav SK, Bera T, Saxena PS, Maurya AK, Garbyal RS, Vajtai R, Ramachandrarao p, Srivastava A (2009) MWCNTS as a reinforcing agent to the Hap-Gel nano composite. J Biomed Mater Res A 93:886–896
Yagi K (1987) Lipid peroxides and human disease. Chem Phys Lipids 45:337–351
Yang S, Wang X, Jia G, Gu Y, Wang T, Nie H, Ge C, Wang H, Liu Y (2008) Long-term accumulation and low toxicity of single-walled carbon nanotubes in intra-venously exposed mice. Toxicol Lett 181(3):182–189
Zhang T, Tang M, Zhang S, Hu Y, Li H, Zhang T, Xue Y, Pu Y (2017) Systemic and immunotoxicity of pristine and PEGylated multi-walled carbon nanotubes in an intra-venous 28 days repeated dose toxicity study. Int J Nanomedicine 12:1539–1554
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
HelmyAbdou, K.A., Ahmed, R.R., Ibrahim, M.A. et al. The anti-inflammatory influence of Cinnamomum burmannii against multi-walled carbon nanotube-induced liver injury in rats. Environ Sci Pollut Res 26, 36063–36072 (2019). https://doi.org/10.1007/s11356-019-06707-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06707-5