Abstract
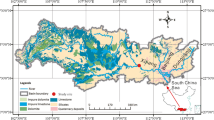
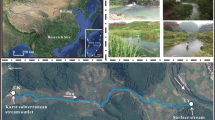
Karst processes play an important role in the global carbon cycle. Aquatic phototrophs can transform bicarbonate, which is mainly derived from the weathering of carbonates, into organic carbon. Carbonate mineral weathering coupled with aquatic photosynthesis can be considered a stable and durable carbon sink process. In this study, we addressed seasonal variations in water chemistry in the Lijiang River Basin, which is a typical karst basin, through a comprehensive geochemical study of the river water in four seasons. The parameters were measured in situ, including major ions and isotopes of inorganic and organic carbon. The results showed that (1) DIC was mainly derived from the weathering of carbonates; (2) the transformation from bicarbonate to organic carbon by aquatic phototrophs was evident, and the water chemistry changed, especially in spring and autumn and in the mainstream from Guilin to Yangshuo, which benefited the growth of aquatic phototrophs; and (3) the organic carbon derived from bicarbonates by aquatic phototrophs was nearly half the total organic carbon and 8% of the dissolved inorganic carbon. These results imply that aquatic phototrophs in karst basins can significantly stabilize carbon originating from carbonate rock weathering processes in karst areas.








Similar content being viewed by others
References
Berner EK, Berner RA (1987) The global water cycle: geochemistry and environment, vol 394. Prentice-Hall, New York
Bouillon S, Yambélé A, Spencer RGM, Gillikin DP, Hernes PJ, Six J, Merckx R, Borges AV (2012) Organic matter sources, fluxes and greenhouse gas exchange in the Oubangui River (Congo River basin). Biogeosciences 9:2045–2062
Boutton TW, Archer SR, Nordt LC (1994) Climate, CO2 and plant abundance. Nature 372:625–626
Cao J, Zhou L, Yang H, Lu Q, Kang Z (2011) Comparison of carbon transfer between forest soils in karst and clasolite areas and the karst carbon sink effect in Maocun village of Guilin. Quat Sci 31:431–437
Chen Q, Shen C, Sun Y, Peng S, Yi W, Li Z, Jiang M (2005) Spatial and temporal distribution of carbon isotopes in soil organic matter at the Dinghushan Biosphere Reserve, South China. Plant Soil 273:115–128
Chen B, Li W, Chen Q, Liu D (2014a) Effects of different bottom sediment and submerged macrophytes on macrofauna communites in the Lijiang River during the summer. Acta Sci Circumst 34:1758–1765
Chen B, Yang R, Liu Z, Yan H, Zhao M (2014b) Effects of aquatic phototrophs on diurnal hydrochemical and δ13CDIC variations in an epikarst spring and two spring-fed ponds of Laqiao, Maolan, SW China. Geochimica 43:375–385
Ciais P et al (2013) Carbon and other biogeochemical cycles. In: Stocker TF et al (eds) Climate Change 2013: The physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambrige and New York, pp 465–570
Clement R, Dimnet L, Maberly SC, Gontero B (2016) The nature of the CO2-concentrating mechanisms in a marine diatom, Thalassiosira pseudonana. New Phytol 209:1417–1427
Clement R, Jensen E, Prioretti L, Maberly SC, Gontero B (2017) Diversity of CO2-concentrating mechanisms and responses to CO2 concentration in marine and freshwater diatoms. J Exp Bot 68:3925–3935
Gaillardet J, Dupré B, Louvat P, Allègre CJ (1999) Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers. Chem Geol 159:3–30
Gandois L, Perrin AS, Probst A (2011) Impact of nitrogenous fertilizer-induced proton release on cultivated soils with contrasting carbonate contents: a column experiment. Geochim Cosmochim Acta 75:1185–1198
Gibbs RJ (1970) Mechanisms Controlling World Water Chemistry. Science 170:1088–1090
Guo Y, Yu S, Li Y, Sun P, He R (2016) Chemical characteristics and source of acid precipitation in Guilin. Environ Sci 37:2897–2905
Han G, Liu C (2004) Water geochemistry controlled by carbonate dissolution: a study of the river waters draining karst-dominated terrain, Guizhou Province, China. Chem Geol 204:1–21
Hindshaw RS, Tipper ET, Reynolds BC, Lemarchand E, Wiederhold JG, Magnusson J, Bernasconi SM, Kretzschmar R, Bourdon B (2011) Hydrological control of stream water chemistry in a glacial catchment (Damma Glacier,Switzerland). Chem Geol 285:215–230
Huang Q, Qin X, Liu P, Tang P (2015) Impact of acid rain to δ13CDIC of karst groundwater and carbon sink in dry season in Guilin. Earth Sci (J China Univ Geosci) 40:1237–1247
Huang S, Pu J, Cao J, Li J, Zhang T, Jiang F, Li L, Wu F, Pan M, Bai B (2018) Origin and effect factors of sedimentary organic carbon in a karst groundwater-fed reservoir, South China. Environ Sci Pollut Res 25:8497–8511
Humborg C, Ittekkot V, Cociasu A, Bodungen BV (1997) Effect of Danube River dam on Black Sea biogeochemistry and ecosystem structure. Nature 386:385–388
Lang SQ, Bernasconi SM, Früh-Green GL (2012) Stable isotope analysis of organic carbon in small (μg C) samples and dissolved organic matter using a GasBench preparation device. Rapid Commun Mass Spectrom 26:9–16
LaZerte BD (1983) Stable Carbon Isotope Ratios: Implications for the source of sediment carbon and for phytoplankton carbon assimilation in Lake Memphremagog Quebec. Can J Fish Aquat Sci 40(10):1658–1666
Li S, Calmels D, Han G, Gaillardet J, Liu C (2008) Sulfuric acid as an agent of carbonate weathering constrained by δ13CDIC: examples from Southwest China. Earth Planet Sci Lett 270:189–199
Li Q, He Y, Cao J, Liang J, Zhu M (2011a) The plant carbonic anhydrase at karst area and its ecological effects. Ecol Environ Sci 20:1867–1871
Li S, Lu X, He M, Zhou T, Bei R, Li L, Ziegler AD (2011b) Major element chemistry in the upper Yangtze River: a case study of the Longchuanjiang River. Geomorphology 129:29–42
Li R, Yu S, Sun P, He S, Yuan Y, Xiong Z (2015) Characteristics of δ13C in typical aquatic plants and carbon sequestration by plant photosynthesis in the Banzhai catchment, Maolan of Guizhou Province. Carsol Sin 34:9–16
Lian B, Yuan D, Liu Z (2011) Effect of microbes on karstification in karst ecosystems. Sci Bull 56:3743–3747
Liu Z, Dreybrod W (1997) Dissolution kinetics of calcium carbonate minerals in H2O-CO2 solutions in turbulent flow: the role of the diffusion boundary layer and the slow reaction H2O+CO2→H++HCO3 −. Geochim Cosmochim Acta 61:2879–2889
Liu Z, Li Q, Sun H, Wang J (2007) Seasonal, diurnal and storm-scale hydrochemical variations of typical epikarst springs in subtropical karst areas of SW China: Soil CO2 and dilution effects. J Hydrol 337:207–223
Liu Y, Zhang J, He Y, Sun H, Liu Z (2010) The utilization of dissolved inorganic carbon by Oocystis solitaria wittr and its influence on the precipitation of calcium carbonate. Geochimica 39:191–196
Liu Z, Dreybrodt W, Liu H (2011) Atmospheric CO2 sink: silicate weathering or carbonate weathering? Appl Geochem 26(Supplement):S292–S294
Liu Z, Macpherson GL, Groves C, Martin JB, Yuan D, Zeng S (2018) Large and active CO2 uptake by coupled carbonate weathering. Earth-Sci Rev 182:42–49
Maher K, Chamberlain CP (2014) Hydrologic regulation of chemical weathering and the geologic carbon cycle. Science 343:1502–1504
Martin JB (2017) Carbonate minerals in the global carbon cycle. Chem Geol 449:58–72
Mcconnaughey T (1998) Acid secretion, calcification, and photosynthetic carbon concentrating. Can J Bot 76:1119–1126
Négrel P, Allègre CJ, Dupré B, Lewin E (1993) Erosion sources determined by inversion of major and trace element ratios and strontium isotopic ratios in river water: the Congo Basin Case. Earth Planet Sci Lett 120:59–76
Pan G, He S, Cao J, Tao Y, Sun Y (2002) Variation of δ13C in karst soil in Yaji Karst Experiment Site, Guilin. Sci Bull 47:500–503
Peng J, Hu R (2001) Carbon and oxygen isotope systematics in the Xikuangshan giant antimony deposit, central Hunan. Geol Rev 47:34–41
Perrin AS, Probst A, Probst JL (2008) Impact of nitrogenous fertilizers on carbonate dissolution in small agricultural catchments: implications for weathering CO2 uptake at regional and global scales. Geochim Cosmochim Acta 72:3105–3123
Pu J, Li J, Khadka MB, Martin JB, Zhang T, Yu S, Yuan D (2017) Instream metabolism and atmospheric carbon sequestration in a groundwater-fed karst stream. Sci Total Environ 579:1343–1355
Pu J, Li J, Zhang T, Martin JB, Khadka MB, Yuan D (2019) Diel-scale variation of dissolved inorganic carbon during a rainfall event in a small karst stream in southern China. Environ Sci Pollut Res 26:11029–11041. https://doi.org/10.1007/s11356-019-04456-z
Qin X, Liu P, Huang Q, Zhang L (2013) Estimation of atmospheric/soil CO2 consumption by rock weathering in the Pearl River Valley. Acta Geosci Sin 34:455–462
Raymond PA, Oh NH, Turner RE, Broussard W (2008) Anthropogenically enhanced fluxes of water and carbon from the Mississippi River. Nature 451:449–452
Redfield AC (1958) The biological control of chemical factors in the environment. Am Sci 46:230A–2221A
Roy S, Gaillardet J, Allègre CJ (1999) Geochemistry of dissolved and suspended loads of the Seine river, France: anthropogenic impact, carbonate and silicate weathering. Geochim Cosmochim Acta 63:1277–1292
Sun H, Han J, Zhang S, Lu X (2007) The impacts of ‘05.6’ extreme flood event on riverine carbon fluxes in Xijiang River. Sci Bull 52:805–812
Sun H, Han J, Li D, Zhang S, Lu X (2010) Chemical weathering inferred from riverine water chemistry in the lower Xijiang basin, South China. Sci Total Environ 408:4749–4760
Sun H, Han J, Zhang S, Lu X (2015) Carbon isotopic evidence for transformation of DIC to POC in the lower Xijiang River, SE China. Quat Int 380–381:288–296
Tao Z, Gao Q, Yao G, Shen C, Wu Q, Wu Z, Liu G (2004) The sources, seasonal variation and transported fluxes of the riverine particulate organic carbon of the Zengjiang River, Southern China. Acta Sci Circumst 24:789–794
Torres MA, West AJ, Li G (2014) Sulphide oxidation and carbonate dissolution as a source of CO2 over geological timescales. Nature 507:346–349
Torres MA, West AJ, Clark KE (2015) Geomorphic regime modulates hydrologic control of chemical weathering in the Andes–Amazon. Geochim Cosmochim Acta 166:105–128
Turner RE, Rabalais NN (1991) Changes in Mississippi river water quality this century. Bioscience 41:140–147
Wang P, Cao J, Li L, Yang H, Li G (2013) Utilization of Ca2+ and HCO3 − in karst water by chlorella from different sources. Acta Hydrob Sin 37(4):626–631
Wang P, Hu G, Cao J (2017) Stable carbon isotopic composition of submerged plants living in karst water and its eco-environmental importance. Aquat Bot 140:78–83
Waterson EJ, Canuel EA (2008) Sources of sedimentary organic matter in the Mississippi River and adjacent Gulf of Mexico as revealed by lipid biomarker and δ13CTOC analyses. Org Geochem 39:422–439
Wu Y, Xing D, Liu Y (2011) The characteristics of bicarbonate used by plants. Earth Environ 39(2):273–277
Wu Y, Xu Y, Li H, Xing D (2012) Effect of acetazolamide on stable carbon isotope fractionation in Chlamydomonas reinhardtii and Chlorella vulgaris. Chin Sci Bull 57:786–789
Xu Z, Liu C (2007) Chemical weathering in the upper reaches of Xijiang River draining the Yunnan–Guizhou Plateau, Southwest China. Chem Geol 239:83–95
Yang M, Liu Z, Sun H, Yang R, Chen B (2016) Organic carbon source tracing and DIC fertilization effect in the Pearl River: Insights from lipid biomarker and geochemical analysis. Appl Geochem 73:132–141
Yu S, He S, Sun P, Pu J, Huang J, Luo H, Li Y, Li R, Yuan Y (2016) Impacts of anthropogenic activities on weathering and carbon fluxes: a case study in the Xijiang River basin, southwest China. Environ Earth Sci 75:589
Zhang J, Quay PD, Wilbur DO (1995) Carbon isotope fractionation during gas-water exchange and dissolution of CO2. Geochim Cosmochim Acta 59:107–114
Zhang S, Lu X, Higgitt DL, Chen C, Sun H, Han J (2007) Water chemistry of the Zhujiang (Pearl River): Natural processes and anthropogenic influences. J Geophys Res 112:F01011
Zhang C, Wang J, Pu J, Yan J (2012a) Bicarbonate daily variations in a Karst River: the Carbon sink effect of subaquatic vegetation photosynthesis. Acta Geol Sin Engl Ed 86:973–979
Zhang H, Yu S, He S, Liu Q, Li Y (2012b) Analysis on the chemical characteristics of the atmospheric precipitation in Guilin. Carsol Sin 31:289–295
Zhang C, Xie Y, Ning L, Yu H, Wang J, Li F (2013) Characteristics of δ13C in typical aquatic plants and carbon sequestration in the Huixian karst wetland, Guilin. Carsol Sin 32:247–252
Zhou J (2015) Climate, depositional environmental changes from middle Holocene to the present documented in Guilin karst wetland. Southwest University, Chongqing
Zhou Z, Chen C, Liu K, Li J, Chen M, Jiang Y, Yu F (2014) Phytoplankton community and water quality in Guilin city section of Lijiang River in summer, China. Ecol Environ Sci 23:649–656
Funding
This study was financially supported by the National Natural Science Foundation of China (Grant Nos. 41402238, 41402324), the Project of Natural Science Foundation of Guangxi (Grant Nos. 2016GXNSFBA380174, 2017GXNSFFA198006), the Project of Institute of Karst Geology, CAGS (2016001), and the Project of the China Geological Survey (DD20190022).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, P., He, S., Yuan, Y. et al. Effects of aquatic phototrophs on seasonal hydrochemical, inorganic, and organic carbon variations in a typical karst basin, Southwest China. Environ Sci Pollut Res 26, 32836–32851 (2019). https://doi.org/10.1007/s11356-019-06374-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06374-6