Abstract
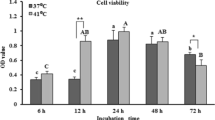
Researchers dealing with heat stress experiments use different cell kinds and use trypsin that has been reported to affect the cellular proteins of cultured cells. Therefore, we compared the effects of acute and chronic exposures to high temperature (45 °C) on camel skin fibroblast and granulosa cells. Primary culture of fibroblasts and granulosa cells tolerated the acute heat shock for 2 h; however, granulosa cells cultured for long duration (20 h) showed thermotolerance when compared with the fibroblasts. Moreover, the effect of cell dispersion method (trypsin and mechanical dissociation) on the thermotolerance of sub-cultured cells was examined. Trypsin altered the morphology of fibroblasts and granulosa cells exposed to 45 °C for 4 h. Moreover, trypsin significantly reduced the fibroblast and granulosa cell migration in the wound healing assay. The current results demonstrate that cell passaging and cell type can affect the thermotolerance of the cells; it also revealed that trypsin could alter the cellular response to the heat shock. We raise the demand for another alternative method for cell dispersion in experiments dealing with cellular responses to the heat shock.




Similar content being viewed by others
References
Abdel-Hameed A, El-Zeiny W, Zaakoug S (2011) Thermoregulation in the one-humped she camel (Camelus dromedarius): diurnal and seasonal effects on core and surface temperatures. J Anim Poult Prod Mans Univ 2:427–437
Baumann H, Doyle D (1979) Effect of trypsin on the cell surface proteins of hepatoma tissue culture cells. Characterization of a carbohydrate-rich glycopeptide released from a calcium binding membrane glycoprotein. J Biol Chem 254:3935–3946
Canavan HE, Cheng X, Graham DJ, Ratner BD, Castner DG (2005) Cell sheet detachment affects the extracellular matrix: a surface science study comparing thermal liftoff, enzymatic, and mechanical methods. J Biomed Mater Res A 75:1–13
Corver WE, Cornelisse CJ, Hermans J, Fleuren GJ (1995) Limited loss of nine tumor-associated surface antigenic determinants after tryptic cell dissociation. Cytometry 19:267–272
Danhier P, Copetti T, De Preter G, Leveque P, Feron O, Jordan BF, Sonveaux P, Gallez B (2013) Influence of cell detachment on the respiration rate of tumor and endothelial cells. PLoS One 8:e53324
Gong YN, Crawford JC, Heckmann BL, Green DR (2019) To the edge of cell death and back. FEBS J 286:430–440
Grigg G, Beard L, Dorges B, Heucke J, Coventry J, Coppock A, Blomberg S (2009) Strategic (adaptive) hypothermia in bull dromedary camels during rut; could it increase reproductive success? Biol Lett 5:853–856
Highfield DP, Holahan EV, Holahan PK, Dewey WC (1984) Hyperthermic survival of Chinese-hamster ovary cells as a function of cellular-population density at the time of plating. Radiat Res 97:139–153
Hoter A, Rizk S, Naim HY (2019) Cellular and molecular adaptation of Arabian camel to heat stress. Front Genet 10:588
Huang HL, Hsing HW, Lai TC, Chen YW, Lee TR, Chan HT, Lyu PC, Wu CL, Lu YC, Lin ST, Lin CW, Lai CH, Chang HT, Chou HC, Chan HL (2010) Trypsin-induced proteome alteration during cell subculture in mammalian cells. J Biomed Sci 17:36 https://doi.org/10.1186/1423-0127-17-36
Liang C-C, Park AY, Guan J-L (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2:329–333
Penna V, Lipay MV, Duailibi MT, Duailibi SE (2015) The likely role of proteolytic enzymes in unwanted differentiation of stem cells in culture. Future Sci OA 1:FSO28
Saadeldin IM, Swelum AA-A, Elsafadi M, Mahmood A, Alfayez M, Alowaimer AN (2018) Differences between the tolerance of camel oocytes and cumulus cells to acute and chronic hyperthermia. J Therm Biol 74:47–54
Saadeldin IM, Swelum AA-A, Noreldin AE, Tukur HA, Abdelazim AM, Abomughaid MM, Alowaimer AN (2019) Isolation and culture of skin-derived differentiated and stem-like cells obtained from the Arabian camel (Camelus dromedarius). Animals 9:378
Samali A, Holmberg CI, Sistonen L, Orrenius S (1999) Thermotolerance and cell death are distinct cellular responses to stress: dependence on heat shock proteins. FEBS Lett 461:306–310
Strober W (2001) Trypan blue exclusion test of cell viability. Curr Protoc Immunol Appendix 3:Appendix 3B. https://doi.org/10.1002/0471142735.ima03bs21
Sun G, Guzman E, Balasanyan V, Conner CM, Wong K, Zhou HR, Kosik KS, Montell DJ (2017) A molecular signature for anastasis, recovery from the brink of apoptotic cell death. J Cell Biol 216:3355–3368
Tang HM, Tang HL (2018) Anastasis: recovery from the brink of cell death. R Soc Open Sci 5:180442
Tang HL, Tang HM, Mak KH, Hu S, Wang SS, Wong KM, Wong CST, Wu HY, Law HT, Liu K, Talbot CC, Lau WK, Montell DJ, Fung MC, Luo K (2012) Cell survival, DNA damage, and oncogenic transformation after a transient and reversible apoptotic response. Mol Biol Cell 23:2240–2252
Thayyullathil F, Chathoth S, Hago A, Wernery U, Patel M, Galadari S (2008) Investigation of heat stress response in the camel fibroblast cell line Dubca. Ann N Y Acad Sci 1138:376–384
Warda M, Prince A, Kim HK, Khafaga N, Scholkamy T, Linhardt RJ, Jin H (2014) Proteomics of old world camelid (Camelus dromedarius): better understanding the interplay between homeostasis and desert environment. J Adv Res 5:219–242
Warters RL, Henle KJ (1982) DNA-degradation in Chinese-hamster ovary cells after exposure to hyperthermia. Cancer Res 42:4427–4432
Wu H, Guang X, al-Fageeh MB, Cao J, Pan S, Zhou H, Zhang L, Abutarboush MH, Xing Y, Xie Z, Alshanqeeti AS, Zhang Y, Yao Q, al-Shomrani BM, Zhang D, Li J, Manee MM, Yang Z, Yang L, Liu Y, Zhang J, Altammami MA, Wang S, Yu L, Zhang W, Liu S, Ba L, Liu C, Yang X, Meng F, Wang S, Li L, Li E, Li X, Wu K, Zhang S, Wang J, Yin Y, Yang H, al-Swailem AM, Wang J (2014) Camelid genomes reveal evolution and adaptation to desert environments. Nat Commun 5:5188
Yarmolenko PS, Moon EJ, Landon C, Manzoor A, Hochman DW, Viglianti BL, Dewhirst MW (2011) Thresholds for thermal damage to normal tissues: an update. Int J Hyperth 27:320–343
Zhang Y, Zhao H, Liu T, Wan C, Liu X, Gao Z, Hou X, Jiang L, Liu F (2015) Activation of transcription factor AP-1 in response to thermal injury in rat small intestine and IEC-6 cells. BMC Gastroenterol 15:83
Acknowledgments
We thank Feriel Yasmine Mahiddine for editing the manuscript.
Funding
This work received funding from the Deanship of Scientific Research at King Saud University through research group RG-1438-018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saadeldin, I.M., Swelum, A.AA., Tukur, H.A. et al. Thermotolerance of camel (Camelus dromedarius) somatic cells affected by the cell type and the dissociation method. Environ Sci Pollut Res 26, 29490–29496 (2019). https://doi.org/10.1007/s11356-019-06208-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06208-5