Abstract
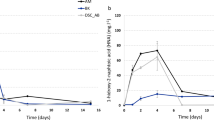
A natural phenanthrene-degrading consortium CON was inoculated with an exogenous strain Sphingobium sp. (ex Sp. paucimobilis) 20006FA yielding the consortium called I-CON, in order to study ecological interactions into the bacterial community. DGGE and proteomic profiles and analyses by HTS (High-Throughput Sequencing) technologies demonstrated inoculant establishment and changes on CON composition. Inoculation increased degradation efficiency in I-CON and prevented intermediate HNA accumulation. This could be explained not only by the inoculation, but also by enrichment in Achromobacter genus at expense of a decrease in Klebsiella genus. After inoculation, cooperation between Sphingobium and Achromobacter genera were improved, thereby, some competition could have been generated, and as a consequence, species in minor proportion (cheaters), as Inquilinus sp. and Luteibacter sp., were not detected. Sequences of Sphingobium (corresponding to the inoculated strain) did not vary. PICRUSt predicted a network with bacterial phylotypes connected with enzymes, showing functional redundancy in the phenanthrene pathway, with exception of the first enzymes biphenyl-2,3-diol 1,2-dioxygenase and protocatechuate 4,5-dioxygenase that were only encoded in Sphingobium sp. This is the first report where a natural consortium that has been characterized by HTS technologies is inoculated with an exogenous strain in order to study competitiveness and interactions.





Similar content being viewed by others
References
Balashova NV, Stolz A, Knackmuss HJ, Kosheleva IA, Naumov AV, Boronin AM (2001) Purification and characterization of a salicylate hydroxylase involved in 1-hydroxy-2-naphthoic acid hydroxylation from the naphthalene and phenanthrene-degrading bacterial strain Pseudomonas putida BS202-P1. Biodegradation 12:179–188. https://doi.org/10.1023/A:1013126723719
Bohorquez LC, Delgado-Serrano L, López G, Osorio-Forero C, Klepac-Ceraj V, Kolter R, Junca H, Baena S, Zambrano MM (2012) In-depth characterization via complementing culture-independent approaches of the microbial Community in an Acidic hot Spring of the Colombian Andes Laura. Microb Ecol 63:103–115. https://doi.org/10.1007/s00248-011-9943-3ENVIRONMENTAL
Brenner K, You L, Arnold FH (2008) Engineering microbial consortia: a new frontier in synthetic biology. Trends Biotechnol 26:483–489. https://doi.org/10.1016/j.tibtech.2008.05.004
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high- throughput community sequencing data intensity normalization improves color calling in SOLiD sequencing. Nat Publ Group 7:335–336. https://doi.org/10.1038/nmeth0510-335
Cerniglia CE (1993) Biodegradation of polycyclic aromatic hydrocarbons. 4:331–338. https://doi.org/10.1016/0958-1669(93)90104-5
Cerniglia CE, Yang SK (1984) Stereoselective metabolism of anthracene and phenanthrene by the fungus Cunninghamella elegans. Appl Environ Microbiol 47:119–124
Chiu CH, Chao A (2014) Distance-based functional diversity measures and their decomposition: a framework based on hill numbers. PLoS One 9:e100014. https://doi.org/10.1371/journal.pone.0100014
Comeau AM, Douglas GM, Langille MGI (2017) Microbiome helper: a custom and streamlined workflow for microbiome research. mSystems 2:e00127–e00116. https://doi.org/10.1128/mSystems.00127-16
Coppotelli BM, Ibarrolaza A, Del Panno MT, Morelli IS (2008) Effects of the inoculant strain Sphingomonas paucimobilis 20006FA on soil bacterial community and biodegradation in phenanthrene-contaminated soil. Microb Ecol 55:173–183. https://doi.org/10.1007/s00248-007-9265-7
Coppotelli BM, Ibarrolaza A, Dias RL, del Panno MT, Berthe-Corti L, Morelli IS (2010) Study of the degradation activity and the strategies to promote the bioavailability of phenanthrene by Sphingomonas paucimobilis strain 20006FA. Microb Ecol 59:266–276. https://doi.org/10.1007/s00248-009-9563-3
De Lorenzo V, Pieper D, Ramos JL (2013) From the test tube to the environment-and back. Environ Microbiol 15:6–11. https://doi.org/10.1111/j.1462-2920.2012.02896.x
De Roy K, Marzorati M, Van den Abbeele P et al (2014) Synthetic microbial ecosystems: an exciting tool to understand and apply microbial communities. Environ Microbiol 16:1472–1481. https://doi.org/10.1111/1462-2920.12343
Desai C, Pathak H, Madamwar D (2010) Advances in molecular and “-omics” technologies to gauge microbial communities and bioremediation at xenobiotic/anthropogen contaminated sites. Bioresour Technol 101:1558–1569. https://doi.org/10.1016/j.biortech.2009.10.080
Dressler MD, Clark CJ, Thachettu CA, Zakaria Y, Eldakar OT, Smith RP (2017) Synthetically engineered microbes reveal interesting principles of cooperation. Front Chem Sci Eng 11:3–14. https://doi.org/10.1007/s11705-016-1605-z
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Festa S, Coppotelli BM, Morelli IS (2013) Bacterial diversity and functional interactions between bacterial strains from a phenanthrene-degrading consortium obtained from a chronically contaminated-soil. Int Biodeterior Biodegrad 85:42–51. https://doi.org/10.1016/j.ibiod.2013.06.006
Festa S, Coppotelli BM, Morelli IS (2016) Comparative bioaugmentation with a consortium and a single strain in a phenanthrene-contaminated soil: impact on the bacterial community and biodegradation. Appl Soil Ecol 98:8–19. https://doi.org/10.1016/j.apsoil.2015.08.025
Festa S, Coppotelli BM, Madueño L, Loviso CL, Macchi M, Neme Tauil RM, Valacco MP, Morelli IS (2017) Assigning ecological roles to the populations belonging to a phenanthrene-degrading bacterial consortium using omic approaches. PLoS One 12:1–21. https://doi.org/10.1371/journal.pone.0184505
Friedman J, Gore J (2017) Ecological systems biology: The dynamics of interacting populations. Curr Opin Syst Biol 1:114–121. https://doi.org/10.1016/j.coisb.2016.12.001
Gómez JP, Bravo GA, Brumfield RT, Tello JG, Cadena CD (2010) A phylogenetic approach to disentangling the role of competition and habitat filtering in community assembly of Neotropical forest birds. J Anim Ecol 79:1181–1192. https://doi.org/10.1111/j.1365-2656.2010.01725.x
Gotelli NJ, Chao A (2013) Measuring and estimating species richness, species diversity, and biotic similarity from sampling data. Elsevier Ltd
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–432. https://doi.org/10.2307/1934352
Hoek TA, Axelrod K, Biancalani T, Yurtsev EA, Liu J, Gore J (2016) Resource availability modulates the cooperative and competitive nature of a microbial cross-feeding mutualism. PLoS Biol 14:1–17. https://doi.org/10.1371/journal.pbio.1002540
Hua X, Wu Z, Zhang H, Lu D, Wang M, Liu Y, Liu Z (2010) Degradation of hexadecane by Enterobacter cloacae strain TU that secretes an exopolysaccharide as a bioemulsifier. Chemosphere 80:951–956. https://doi.org/10.1016/j.chemosphere.2010.05.002
Kazunga C, Aitken MD (2000) Products from the incomplete metabolism of pyrene by polycyclic aromatic hydrocarbon-degrading bacteria. Appl Environ Microbiol 66:1917–1922. https://doi.org/10.1128/AEM.66.5.1917-1922.2000
Khara P, Roy M, Chakraborty J, Ghosal D, Dutta TK (2014) Functional characterization of diverse ring-hydroxylating oxygenases and induction of complex aromatic catabolic gene clusters in Sphingobium sp. PNB. FEBS Open Bio 4:290–300. https://doi.org/10.1016/j.fob.2014.03.001
Kopylova E, Noé L, Touzet H (2012) SortMeRNA: fast and accurate filtering of ribosomal RNAs in metatranscriptomic data. Bioinformatics 28:3211–3217. https://doi.org/10.1093/bioinformatics/bts611
Langille MGI, Zaneveld J, Caporaso JG, McDonald D, Knights D, Reyes JA, Clemente JC, Burkepile DE, Vega Thurber RL, Knight R, Beiko RG, Huttenhower C (2013) Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol 31:814–821. https://doi.org/10.1038/nbt.2676
Macchi M, Martinez M, Tauil RMN, Valacco MP, Morelli IS, Coppotelli BM (2018) Insights into the genome and proteome of Sphingomonas paucimobilis strain 20006FA involved in the regulation of polycyclic aromatic hydrocarbon degradation. World J Microbiol Biotechnol 34:7. https://doi.org/10.1007/s11274-017-2391-6
Madueño L, Coppotelli BM, Alvarez HM, Morelli IS (2011) Isolation and characterization of indigenous soil bacteria for bioaugmentation of PAH contaminated soil of semiarid Patagonia, Argentina. Int Biodeterior Biodegrad 65:345–351. https://doi.org/10.1016/j.ibiod.2010.12.008
Mallon CA, Van Elsas JD, Salles JF (2015) Microbial invasions: the process, patterns, and mechanisms. Trends Microbiol 23:719–729. https://doi.org/10.1016/j.tim.2015.07.013
Mccully AL, Lasarre B, Mckinlay JB (2017) Recipient-biased competition for an intracellularly generated cross-fed nutrient is required for coexistence of microbial mutualists. MBio 8:1–13. https://doi.org/10.1128/mBio.01620-17
Megharaj M, Ramakrishnan B, Venkateswarlu K, Sethunathan N, Naidu R (2011) Bioremediation approaches for organic pollutants: a critical perspective. Environ Int 37:1362–1375. https://doi.org/10.1016/j.envint.2011.06.003
Mendes LW, Tsai SM, Navarrete AA, de Hollander M, van Veen JA, Kuramae EE (2015) Soil-borne microbiome: linking diversity to function. Microb Ecol 70:255–265. https://doi.org/10.1007/s00248-014-0559-2
Mercier C, Boyer F, Bonin A, Coissac E (2013) SUMATRA and SUMACLUST: fast and exact comparison and clustering of sequences. Available: https://git.metabarcoding.org/obitools/sumaclust
Muyzer G, Smalla K (1998) Application of denaturing gradient gel electrophoresis (DGGE) and temperature gradient gel electrophoresis (TGGE) in microbial ecology. Antonie Van Leewenhoek 73:127–141. https://doi.org/10.1023/A:1000669317571
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Pavlopoulos GA, Secrier M, Moschopoulos CN, Soldatos TG, Kossida S, Aerts J, Schneider R, Bagos PG (2011) Using graph theory to analyze biological networks. BioData Min 4:1–27. https://doi.org/10.1186/1756-0381-4-10
Pavlopoulos GA, Kontou PI, Pavlopoulou A et al (2018) Bipartite graphs in systems biology and medicine: a survey of methods and applications. GigaScience 7(4):giy014. https://doi.org/10.1093/gigascience/giy014
Quast C, Pruesse E, Yilmaz P, et al (2012) The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res 41:590–596. https://doi.org/10.1093/nar/gks1219
Reasoner DJ, Geldreich EE (1985) A new medium for the enumeration and subculture of bacteria from potable water. Appl Environ Microbiol 49:1–7. https://doi.org/10.1016/s0016-7037(99)00322-1
Rognes T, Flouri T, Nichols B, Quince C, Mahé F (2016) VSEARCH: a versatile open source tool for metagenomics. PeerJ 4:e2584. https://doi.org/10.7717/peerj.2584
Rungrassamee W, Klanchui A, Chaiyapechara S, Maibunkaew S, Tangphatsornruang S, Jiravanichpaisal P, Karoonuthaisiri N (2013) Bacterial population in intestines of the black tiger shrimp (Penaeus monodon) under different growth stages. PLoS One 8:e60802. https://doi.org/10.1371/journal.pone.0060802
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Stadie J, Gulitz A, Ehrmann MA, Vogel RF (2013) Metabolic activity and symbiotic interactions of lactic acid bacteria and yeasts isolated from water kefir. Food Microbiol 35:92–98. https://doi.org/10.1016/j.fm.2013.03.009
Stolz A (2009) Molecular characteristics of xenobiotic-degrading sphingomonads. Appl Microbiol Biotechnol 81:793–811. https://doi.org/10.1007/s00253-008-1752-3
Su Z, Dai T, Tang Y, Tao Y, Huang B, Mu Q, Wen D (2018) Sediment bacterial community structures and their predicted functions implied the impacts from natural processes and anthropogenic activities in coastal area. Mar Pollut Bull 131:481–495. https://doi.org/10.1016/j.marpolbul.2018.04.052
Takeuchi M, Hamana K, Hiraishi A (2001) Proposal of the genus Sphingomonas sensu stricto and three new genera. Int J Syst Evol Microbiol 51:1405–1417. https://doi.org/10.1099/00207713-51-4-1405
Tamaki H, Wright CL, Li X, Lin Q, Hwang C, Wang S, Thimmapuram J, Kamagata Y, Liu WT (2011) Analysis of 16S rRNA amplicon sequencing options on the roche/454 next-generation titanium sequencing platform. PLoS One 6:6–8. https://doi.org/10.1371/journal.pone.0025263
Tao K, Liu X, Chen X, Hu X, Cao L, Yuan X (2017) Biodegradation of crude oil by a defined co-culture of indigenous bacterial consortium and exogenous Bacillus subtilis. Bioresour Technol 224:327–332. https://doi.org/10.1016/j.biortech.2016.10.073
Timmis KN, Pieper DH (1999) Bacteria designed for bioremediation. Trends Biotechnol 17:201–204. https://doi.org/10.1016/S0167-7799(98)01295-5
Toledo FL, Gonzalez-Lopez J, Calvo C (2008) Production of bioemulsifier by Bacillus subtilis, Alcaligenes faecalis and Enterobacter species in liquid culture. Bioresour Technol 99:8470–8475. https://doi.org/10.1016/j.biortech.2007.08.055
Vecchioli GI, Del Panno MT, Painceira MT (1990) Use of selected autochthonous soil bacteria to enhanced degradation of hydrocarbons in soil. Environ Pollut 67:249–258. https://doi.org/10.1016/0269-7491(90)90190-N
Větrovský T, Baldrian P (2013) The variability of the 16S rRNA gene in bacterial genomes and its consequences for bacterial community analyses. PLoS One 8:1–10. https://doi.org/10.1371/journal.pone.0057923
Waigi MG, Kang F, Goikavi C, Ling W, Gao Y (2015) Phenanthrene biodegradation by sphingomonads and its application in the contaminated soils and sediments: a review. Int Biodeterior Biodegrad 104:333–349. https://doi.org/10.1016/j.ibiod.2015.06.008
Waite AJ, Shou W (2012) Adaptation to a new environment allows cooperators to purge cheaters stochastically. Proc Natl Acad Sci U S A 109:19079–19086. https://doi.org/10.1073/pnas.1210190109
Wanapaisan P, Laothamteep N, Vejarano F, Chakraborty J, Shintani M, Muangchinda C, Morita T, Suzuki-Minakuchi C, Inoue K, Nojiri H, Pinyakong O (2018) Synergistic degradation of pyrene by five culturable bacteria in a mangrove sediment-derived bacterial consortium. J Hazard Mater 342:561–570. https://doi.org/10.1016/j.jhazmat.2017.08.062
Wang H, Wang B, Dong W, Hu X (2016) Co-acclimation of bacterial communities under stresses of hydrocarbons with different structures. Sci Rep 6:1–12. https://doi.org/10.1038/srep34588
Wickham H (2016) ggplot2 - elegant graphics for data analysis. Springer-Verlag, New York 77. https://doi.org/10.1007/978-3-319-24277-4
Wrenn BA, Venosa AD (1996) Selective enumeration of aromatic and aliphatic hydrocarbon degrading bacteria by a most-probable-number procedure. Can J Microbiol 42:252–258. https://doi.org/10.1139/m96-037
Yang T, Wei Z, Friman VP, Xu Y, Shen Q, Kowalchuk GA, Jousset A (2017) Resource availability modulates biodiversity-invasion relationships by altering competitive interactions. Environ Microbiol 19:2984–2991. https://doi.org/10.1111/1462-2920.13708
Yuan SY, Wei SH, Chang BV (2000) Biodegradation of polycyclic aromatic hydrocarbons by a mixed culture. Chemosphere 41:1463–1468
Zafra G, Absalón ÁE, Anducho-Reyes MÁ, Fernandez FJ, Cortés-Espinosa DV (2017) Construction of PAH-degrading mixed microbial consortia by induced selection in soil. Chemosphere 172:120–126. https://doi.org/10.1016/j.chemosphere.2016.12.038
Zhang J, Kobert K, Flouri T, Stamatakis A (2014) PEAR: a fast and accurate Illumina paired-end reAd mergeR. Bioinformatics 30:614–620. https://doi.org/10.1093/bioinformatics/btt593
Zhao HP, Wang L, Ren JR, Li Z, Li M, Gao HW (2008) Isolation and characterization of phenanthrene-degrading strains Sphingomonas sp. ZP1 and Tistrella sp. ZP5. J Hazard Mater 152:1293–1300. https://doi.org/10.1016/j.jhazmat.2007.08.008
Zhao JK, Li XM, Ai GM, Deng Y, Liu SJ, Jiang CY (2016) Reconstruction of metabolic networks in a fluoranthene-degrading enrichments from polycyclic aromatic hydrocarbon polluted soil. J Hazard Mater 318:90–98. https://doi.org/10.1016/j.jhazmat.2016.06.055
Zhao Q, Yue S, Bilal M, et al (2017) Comparative genomic analysis of 26 Sphingomonas and Sphingobium strains: Dissemination of bioremediation capabilities, biodegradation potential and horizontal gene transfer. Sci Total Environ 609:1238–1247. https://doi.org/10.1016/j.scitotenv.2017.07.249
Funding
This research was partially supported by the Agencia Nacional de Promoción Científica y Tecnológica (PICT 2013–0103). Festa S. and Macchi M. have doctoral and postdoctoral fellowships supported by CONICET. Morelli I.S. is a research member of CIC-PBA. Coppotelli B.M. is a research member of CONICET.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Robert Duran
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 699 kb)
Rights and permissions
About this article
Cite this article
Macchi, M., Festa, S., Vega-Vela, N.E. et al. Assessing interactions, predicting function, and increasing degradation potential of a PAH-degrading bacterial consortium by effect of an inoculant strain. Environ Sci Pollut Res 26, 25932–25944 (2019). https://doi.org/10.1007/s11356-019-05760-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05760-4