Abstract
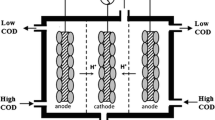
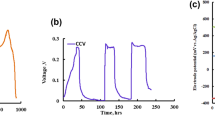
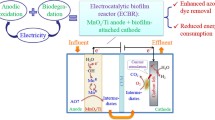
A distinctive process (BCE-MFC) was developed to explore the methyl orange (MO) degradation and simultaneous bioelectricity generation based on the functional collaboration of biofilm, electrolysis, constructed wetland, and microbial fuel cell. The biofilm-cathode electrode–microbial fuel cell (BCE-MFC) was capable of sustaining an excellent MO removal (100%) and bioelectricity production (0.63 V). BCE significantly enhanced MO biodegradability, thus resulting in a 56.3% improvement of COD removal in subsequent MFC. Bacillus was dominant in biofilm on cathode in BCE. In MFC, Proteobacteria phylum (64.84%) and Exiguobacterium genus (13.30%) were predominated in the anode region, probably basically responsible for electricity generation. Interestingly, relatively high content of Heliothrix sp. (9.94%) was found in the MFC designed here, which was likely to participate in electricity production as well. The proposed functional collaboration may be an effective strategy in refractory wastewater treatment and power production.







Similar content being viewed by others
References
APHA (2005) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, American Water Works Association and Water Environment Federation, Washington
Balapure KH, Jain K, Chattaraj S, Bhatt NS, Madamwar D (2014) Co-metabolic degradation of diazo dye-reactive blue 160 by enriched mixed cultures BDN. J Hazard Mater 279:85–95
Basu A, Suresh Kumar G (2014) Targeting proteins with toxic azo dyes: a microcalorimetric characterization of the interaction of the food colorant amaranth with serum proteins. J Agric Food Chem 62:7955–7962
Cerrillo M, Viñas M, Bonmatí A (2017) Unravelling the active microbial community in a thermophilic anaerobic digester-microbial electrolysis cell coupled system under different conditions. Water Res 110:192–201
Chen B, Ma C, Han K, Yueh P, Qin L, Hsueh C (2016) Influence of textile dye and decolorized metabolites on microbial fuel cell-assisted bioremediation. Bioresour Technol 200:1033–1038
Evelyn LY, Marshall A, Gostomski PA (2014) Gaseous pollutant treatment and electricity generation in microbial fuel cells (MFCs) utilising redox mediators. Rev Environ Sci Biotechnol 13:35–51
Fang Z, Song H, Yu R, Li X (2016) A microbial fuel cell-coupled constructed wetland promotes degradation of azo dye decolorization products. Ecol Eng 94:455–463
Hamad H, Bassyouni D, El-Ashtoukhy ES, Amin N, Abd El-Latif M (2018) Electrocatalytic degradation and minimization of specific energy consumption of synthetic azo dye from wastewater by anodic oxidation process with an emphasis on enhancing economic efficiency and reaction mechanism. Ecotoxicol Environ Saf 148:501–512
Horst A, Mangold KM, Holtmann D (2015) Application of gas diffusion electrodes in bioelectrochemical syntheses and energy conversion. Biotechnol Bioeng 113:260–267
Hou Y, Zhang R, Yu Z, Huang L, Liu Y, Zhou Z (2017) Accelerated azo dye degradation and concurrent hydrogen production in the single-chamber photocatalytic microbial electrolysis cell. Bioresour Technol 224:63–68
Hu M, Wang X, Wen X, Xia Y (2012) Microbial community structures in different wastewater treatment plants as revealed by 454-pyrosequencing analysis. Bioresour Technol 117:72–79
Huang J, Zhu N, Cao Y, Peng Y, Wu P, Dong W (2015) Exoelectrogenic bacterium phylogenetically related to citrobacter freundii, isolated from anodic biofilm of a microbial fuel cell. Appl Biochem Biotechnol 175:1879–1891
Khongkliang P, Kongjan P, Utarapichat B, Reungsang A, O-Thong S (2017) Continuous hydrogen production from cassava starch processing wastewater by two-stage thermophilic dark fermentation and microbial electrolysis. Int J Hydrog Energy 42:27584–27592
Kim KY, Zikmund E, Logan BE (2017) Impact of catholyte recirculation on different 3-dimensional stainless steel cathodes in microbial electrolysis cells. Int J Hydrog Energy 42:29708–29715
Liu ST, Song HL, Wei S, Liu Q, Li XN, Qian X (2015) Effect of direct electrical stimulation on decolorization and degradation of azo dye reactive brilliant red X-3B in biofilm-electrode reactors. Biochem Eng J 93:294–302
Mohanakrishna G, Abu-Reesh IM, Al-Raoush RI, He Z, Mohanakrishna G (2018) Cylindrical graphite based microbial fuel cell for the treatment of industrial wastewaters and bioenergy generation. Bioresour Technol 247:753–758
Mu Y, Rabaey K, Rozendal RA, Yuan Z, Keller J (2009) Decolorization of azo dyes in bioelectrochemical systems. Environ Sci Technol 43:5137–5143
Ooi J, Lee LY, Hiew BYZ, Thangalazhy-Gopakumar S, Lim SS, Gan S (2017) Assessment of fish scales waste as a low cost and eco-friendly adsorbent for removal of an azo dye: equilibrium, kinetic and thermodynamic studies. Bioresour Technol 245:656–664
Oturkar CC, Patole MS, Gawai KR, Madamwar D (2013) Enzyme based cleavage strategy of Bacillus lentus BI377 in response to metabolism of azoic recalcitrant. Bioresour Technol 130:360–365
Park HI, Ji SK, Dong KK, Choi YJ, Pak D (2006) Nitrate-reducing bacterial community in a biofilm-electrode reactor. Enzym Microb Technol 39:453–458
Pearce C (2003) The removal of colour from textile wastewater using whole bacterial cells: a review. Dyes Pigments 58:179–196
Shabbir S, Faheem M, Ali N, Kerr PG, Wu Y (2017) Evaluating role of immobilized periphyton in bioremediation of azo dye amaranth. Bioresour Technol 225:395–401
Tee HC, Lim PE, Seng CE, Nawi MM, Adnan R (2015) Enhancement of azo dye Acid Orange 7 removal in newly developed horizontal subsurface-flow constructed wetland. J Environ Manag 147:349–355
Thrash JC, Coates JD (2008) Review: direct and indirect electrical stimulation of microbial metabolism. Environ Sci Technol 42:3921–3931
Thung W, Ong S, Ho L, Wong Y, Ridwan F, Oon Y, Lehl HK (2015) A highly efficient single chambered up-flow membrane-less microbial fuel cell for treatment of azo dye Acid Orange 7-containing wastewater. Bioresour Technol 197:284–288
Van Der Zee FP, Bouwman RH, Strik DP, Lettinga G, Field JA (2001) Application of redox mediators to accelerate the transformation of reactive azo dyes in anaerobic bioreactors. Biotechnol Bioeng 75:691–701
Wang J, Wang Y, Bai J, Liu Z, Song X, Yan D, Abiyu A, Zhao Z, Yan D (2017) High efficiency of inorganic nitrogen removal by integrating biofilm-electrode with constructed wetland: autotrophic denitrifying bacteria analysis. Bioresour Technol 227:7–14
Xiao L, He Z (2014) Applications and perspectives of phototrophic microorganisms for electricity generation from organic compounds in microbial fuel cells. Renew Sust Energ Rev 37:550–559
Yang S, Xiao T, Zhang J, Chen Y, Li L (2015) Activated carbon fiber as heterogeneous catalyst of peroxymonosulfate activation for efficient degradation of Acid Orange 7 in aqueous solution. Sep Purif Technol 143:19–26
Zeng X, Collins MA, Borole AP, Pavlostathis SG (2017) The extent of fermentative transformation of phenolic compounds in the bioanode controls exoelectrogenic activity in a microbial electrolysis cell. Water Res 109:299–309
Zhang G, Jiao Y, Lee D (2015) A lab-scale anoxic/oxic-bioelectrochemical reactor for leachate treatments. Bioresour Technol 186:97–105
Zhang S, Song H, Yang X, Yang Y, Yang K, Wang X (2016) Fate of tetracycline and sulfamethoxazole and their corresponding resistance genes in microbial fuel cell coupled constructed wetlands. RSC Adv 6:95999–96005
Zhang X, Huang W, Wang X, Li H, Lu H, Lin H (2011) Biofilm-electrode process with high efficiency for degradation of 2, 4-dichlorophenol. Environ Chem Lett 9:383–388
Zhang Y, Angelidaki I (2014) Microbial electrolysis cells turning to be versatile technology: recent advances and future challenges. Water Res 56:11–25
Zhong Y, Li X, Yang Q, Wang D, Yao F (2016) Complete bromate and nitrate reduction using hydrogen as the sole electron donor in a rotating biofilm-electrode reactor. J Hazard Mater 307:82–90
Zhou M, Fu W, Gu H, Lei L (2007) Nitrate removal from groundwater by a novel three-dimensional electrode biofilm reactor. Electrochim Acta 52:6052–6059
Zou HM, Lu XW, Abualhail S, Shi J, Gu Q (2014) Enrichment of PAO and DPAO responsible for phosphorus removal at low temperature. Environ Prot Eng 40:67–82
Zou HM, Wang Y (2017) Azo dyes wastewater treatment and simultaneous electricity generation in a novel process of electrolysis cell combined with microbial fuel cell. Bioresour Technol 235:167–175
Funding
This research was supported by the Research Items from the Department of Science and Technology of Anhui Province, China (1604f0704047), and the Special Foundation for Young Scientists of Anhui province, China (gxyqZD2016212).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Bingcai Pan
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zou, H., Wang, Y. Functional collaboration of biofilm-cathode electrode and microbial fuel cell for biodegradation of methyl orange and simultaneous bioelectricity generation . Environ Sci Pollut Res 26, 23061–23069 (2019). https://doi.org/10.1007/s11356-019-05617-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05617-w