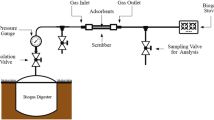
Abstract
Acid gases such as carbon dioxide and hydrogen sulfide are common contaminants in oil and gas operations, landfill gases, and exhaust stacks from power plants. While there are processes currently used to treat these effluents (e.g., amine absorption and adsorption using zeolite), many of these processes require high energy, space, and hazardous chemicals. Removal using biochar derived from the fast pyrolysis of forestry residues represents a more sustainable option. In this study, adsorption using CO2 as a surrogate for acid gases was investigated using various biochars produced from fast pyrolysis of sawmill residues. Response surface methodology was used to determine operating conditions for maximum adsorption and assess interaction of the adsorption parameters, i.e., temperature, inlet feed flow rate, and CO2 concentration, on biochar adsorption capacity. The Freundlich isotherm best represented the equilibrium adsorption, and the kinetic model was pseudo first-order. Thermodynamic analysis indicated the adsorption process was spontaneous and exothermic. The biochar had better adsorption capacity relative to commercial zeolite. Our results suggested that biochar could be used as a sustainable and cost-effective option for contaminant removal from acid gases produced in landfill gas treatment, fossil fuel extraction, and/or combustion.











Similar content being viewed by others
References
Abnisa F, Arami-Niya A, Daud WMAW, Sahu JN (2013) Characterization of bio-oil and bio-char from pyrolysis of palm oil wastes. Bioenergy Res 6:830–840. https://doi.org/10.1007/s12155-013-9313-8
Ammendola P, Raganati F, Chirone R (2017) CO2 adsorption on a fine activated carbon in a sound assisted fluidized bed: thermodynamics and kinetics. Chem Eng J 322:302–313. https://doi.org/10.1016/j.cej.2017.04.037
Anderson MJ, Whitcomb PJ (2013) DOE Simplified: practical tools for effective experimentation, vol 53. https://doi.org/10.1017/CBO9781107415324.004
Auta M, Hameed BH (2014) Adsorption of carbon dioxide by diethanolamine activated alumina beads in a fixed bed. Chem Eng J 253:350–355. https://doi.org/10.1016/j.cej.2014.05.018
Bamdad H, Hawboldt K (2016) Comparative study between physicochemical characterization of biochar and metal organic frameworks (MOFs) as gas adsorbents. Can J Chem Eng 9999:1–7. https://doi.org/10.1002/cjce.22595
Bamdad H, Hawboldt K, Macquarrie S (2018a) Nitrogen functionalized biochar as a renewable adsorbent for efficient CO2 removal. Energy Fuel 32:11742–11748. https://doi.org/10.1021/acs.energyfuels.8b03056
Bamdad H, Hawboldt K, MacQuarrie S (2018b) A review on common adsorbents for acid gases removal: focus on biochar. Renew Sust Energ Rev 81:1705–1720
Baroutaji A, Gilchrist MD, Smyth D, Olabi AG (2015) Crush analysis and multi-objective optimization design for circular tube under quasi-static lateral loading. Thin-Walled Struct 86:121–131. https://doi.org/10.1016/j.tws.2014.08.018
Baş D, Boyacı İH, Bas D et al (2007) Modeling and optimization I: usability of response surface methodology. J Food Eng 78:836–845. https://doi.org/10.1016/j.jfoodeng.2005.11.024
Box GEP, Wilson KB (1951) On the experimental attainment of optimum conditions. J R Stat Soc 13:1–45. https://doi.org/10.1007/978-1-4612-4380-9_23
Bruce PC (2016) Introductory statistics and analytics: a resampling perspective. John Wiley, Inc., Hoboken
Chatterjee R, Sajjadi B, Mattern DL, Chen WY, Zubatiuk T, Leszczynska D, Leszczynski J, Egiebor NO, Hammer N (2018) Ultrasound cavitation intensified amine functionalization: a feasible strategy for enhancing CO2capture capacity of biochar. Fuel 225:287–298. https://doi.org/10.1016/j.fuel.2018.03.145
Chen Y, Zhang D (2014) Adsorption kinetics, isotherm and thermodynamics studies of flavones from Vaccinium Bracteatum Thunb leaves on NKA-2 resin. Chem Eng J 254:579–585. https://doi.org/10.1016/j.cej.2014.05.120
Chen CP, Chuang MT, Hsiao YH, Yang YK, Tsai CH (2009a) Simulation and experimental study in determining injection molding process parameters for thin-shell plastic parts via design of experiments analysis. Expert Syst Appl 36:10752–10759. https://doi.org/10.1016/j.eswa.2009.02.017
Chen S, Shen W, Yu F, Wang H (2009b) Kinetic and thermodynamic studies of adsorption of Cu2+ and Pb2+ onto amidoximated bacterial cellulose. Polym Bull 63:283–297. https://doi.org/10.1007/s00289-009-0088-1
Creamer AE, Gao B, Zhang M (2014) Carbon dioxide capture using biochar produced from sugarcane bagasse and hickory wood. Chem Eng J 249:174–179. https://doi.org/10.1016/j.cej.2014.03.105
Draper NR, Smith H (1998) Applied regression analysis. Technometrics 47:706. https://doi.org/10.1198/tech.2005.s303
Espejel-Ayala F, Corella RC, Pérez AM et al (2014) Carbon dioxide capture utilizing zeolites synthesized with paper sludge and scrap-glass. Waste Manag Res 32:1219–1226. https://doi.org/10.1177/0734242X14554643
Freundlich HM (1926) New conception in colloidal chemistry, colloid and capillary chemistry. Methuen 45:970–984. https://doi.org/10.3390/ijerph9030970
Gallucci F, Van Sint Annaland M (2015) Process intensification for sustainable energy conversion. https://doi.org/10.1002/9781118449394
García S, Gil MV, Martín CF et al (2011) Breakthrough adsorption study of a commercial activated carbon for pre-combustion CO2 capture. Chem Eng J 171:549–556. https://doi.org/10.1016/j.cej.2011.04.027
Geethakarthi A, Phanikumar BR (2011) Adsorption of reactive dyes from aqueous solutions by tannery sludge developed activated carbon: kinetic and equilibrium studies. Int J Environ Sci Technol 8:561–570. https://doi.org/10.1007/BF03326242
Gereli G, Seki Y, Murat Kuşoǧlu I, Yurdakoç K (2006) Equilibrium and kinetics for the sorption of promethazine hydrochloride onto K10 montmorillonite. J Colloid Interface Sci 299:155–162. https://doi.org/10.1016/j.jcis.2006.02.012
Ghorai S, Pant KK (2005) Equilibrium, kinetics and breakthrough studies for adsorption of fluoride on activated alumina. Sep Purif Technol 42:265–271. https://doi.org/10.1016/j.seppur.2004.09.001
Gil MV, Álvarez-Gutiérrez N, Martínez M, Rubiera F, Pevida C, Morán A (2015) Carbon adsorbents for CO2 capture from bio-hydrogen and biogas streams: breakthrough adsorption study. Chem Eng J 269:148–158. https://doi.org/10.1016/j.cej.2015.01.100
Giles CH, Smith D, Huitson A (1974) A general treatment and classification of the solute adsorption isotherm. I. Theoretical. J Colloid Interface Sci 47:755–765. https://doi.org/10.1016/0021-9797(74)90252-5
Goel C, Kaur H, Bhunia H, Bajpai PK (2016) Carbon dioxide adsorption on nitrogen enriched carbon adsorbents: experimental, kinetics, isothermal and thermodynamic studies. J CO2 Util 16:50–63. https://doi.org/10.1016/j.jcou.2016.06.002
González AS, Plaza MG, Rubiera F, Pevida C (2013) Sustainable biomass-based carbon adsorbents for post-combustion CO2 capture. Chem Eng J 230:456–465. https://doi.org/10.1016/j.cej.2013.06.118
Guerrero M, Ruiz MP, Alzueta MU, Bilbao R, Millera A (2005) Pyrolysis of eucalyptus at different heating rates: studies of char characterization and oxidative reactivity. J Anal Appl Pyrolysis 74:307–314. https://doi.org/10.1016/j.jaap.2004.12.008
Halsey G (1948) Physical adsorption on non uniform surfaces. J Chem Phys 16:931–937. https://doi.org/10.1063/1.1747042
Heidari A, Younesi H, Rashidi A, Ghoreyshi AA (2014a) Evaluation of CO2adsorption with eucalyptus wood based activated carbon modified by ammonia solution through heat treatment. Chem Eng J 254:503–513. https://doi.org/10.1016/j.cej.2014.06.004
Heidari A, Younesi H, Rashidi A, Ghoreyshi AA (2014b) Evaluation of CO2 adsorption with eucalyptus wood based activated carbon modified by ammonia solution through heat treatment. Chem Eng J 254:503–513. https://doi.org/10.1016/j.cej.2014.06.004
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Khan TA, Khan EA, Shahjahan (2015) Removal of basic dyes from aqueous solution by adsorption onto binary iron-manganese oxide coated kaolinite: non-linear isotherm and kinetics modeling. Appl Clay Sci 107:70–77. https://doi.org/10.1016/j.clay.2015.01.005
Kim KH, Kim JY, Cho TS, Choi JW (2012) Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Bioresour Technol 118:158–162. https://doi.org/10.1016/j.biortech.2012.04.094
Kim KH, Kim TS, Lee SM, Choi D, Yeo H, Choi IG, Choi JW (2013) Comparison of physicochemical features of biooils and biochars produced from various woody biomasses by fast pyrolysis. Renew Energy 50:188–195. https://doi.org/10.1016/j.renene.2012.06.030
Langmuir I (1916) The constitution and fundamental properties of solids and liquids (part I). J Am Chem Soc 38:2221–2295. https://doi.org/10.1021/ja02268a002
Li J, Dai J, Liu G, Zhang H, Gao Z, Fu J, He Y, Huang Y (2016) Biochar from microwave pyrolysis of biomass: a review. Biomass Bioenergy 94:228–244
Liu Y (2009) Is the free energy change of adsorption correctly calculated? J Chem Eng Data 54:1981–1985. https://doi.org/10.1021/je800661q
Liu H, Cai X, Wang Y, Chen J (2011) Adsorption mechanism-based screening of cyclodextrin polymers for adsorption and separation of pesticides from water. Water Res 45:3499–3511. https://doi.org/10.1016/j.watres.2011.04.004
Lua AC, Yang T (2009) Theoretical and experimental SO2 adsorption onto pistachio-nut-shell activated carbon for a fixed-bed column. Chem Eng J 155:175–183. https://doi.org/10.1016/j.cej.2009.07.031
Lua AC, Yang T, Guo J (2004) Effects of pyrolysis conditions on the properties of activated carbons prepared from pistachio-nut shells. J Anal Appl Pyrolysis 72:279–287. https://doi.org/10.1016/j.jaap.2004.08.001
Monazam ER, Spenik J, Shadle LJ (2013) Fluid bed adsorption of carbon dioxide on immobilized polyethylenimine (PEI): kinetic analysis and breakthrough behavior. Chem Eng J 223:795–805. https://doi.org/10.1016/j.cej.2013.02.041
Morero B, Groppelli ES, Campanella EA (2016) Evaluation of biogas upgrading technologies using a response surface methodology for process simulation. J Clean Prod 141:978–988. https://doi.org/10.1016/j.jclepro.2016.09.167
Mulgundmath VP, Jones RA, Tezel FH, Thibault J (2012) Fixed bed adsorption for the removal of carbon dioxide from nitrogen: breakthrough behaviour and modelling for heat and mass transfer. Sep Purif Technol 85:17–27. https://doi.org/10.1016/j.seppur.2011.07.038
O’Mahony T, Guibal E, Tobin JM (2002) Reactive dye biosorption by Rhizopus arrhizus biomass. Enzym Microb Technol 31:456–463. https://doi.org/10.1016/S0141-0229(02)00110-2
Papari S, Hawboldt K, Helleur R (2015) Pyrolysis: a theoretical and experimental study on the conversion of softwood sawmill residues to biooil. Ind Eng Chem Res 54:605–611. https://doi.org/10.1021/ie5039456
Papari S, Hawboldt K, Helleur R (2017) Production and characterization of pyrolysis oil from sawmill residues in an auger reactor. Ind Eng Chem Res 56:1920–1925. https://doi.org/10.1021/acs.iecr.6b04405
Plaza MG, Pevida C, Arenillas A, Rubiera F, Pis JJ (2007) CO2 capture by adsorption with nitrogen enriched carbons. Fuel 86:2204–2212. https://doi.org/10.1016/j.fuel.2007.06.001
Plaza MG, González AS, Pis JJ, Rubiera F, Pevida C (2014) Production of microporous biochars by single-step oxidation: effect of activation conditions on CO2 capture. Appl Energy 114:551–562. https://doi.org/10.1016/j.apenergy.2013.09.058
Raganati F, Alfe M, Gargiulo V, et al (2018) Isotherms and thermodynamics of CO2 adsorption on a novel carbon-magnetite composite sorbent. Chem Eng Res Des 134:540–552. https://doi.org/10.1016/j.cherd.2018.04.037
Rajapaksha AU, Vithanage M, Zhang M, Ahmad M, Mohan D, Chang SX, Ok YS (2014) Pyrolysis condition affected sulfamethazine sorption by tea waste biochars. Bioresour Technol 166:303–308. https://doi.org/10.1016/j.biortech.2014.05.029
Rouquerol J, Rouquerol F, Llewellyn P, Maurin G, Sing KSW (2013) Adsorption by powders and porous solids: principles, methodology and applications: second edition. https://doi.org/10.1016/C2010-0-66232-8.
Ryu Z, Zheng J, Wang M, Zhang B (1999) Characterization of pore size distributions on carbonaceous adsorbents by DFT. Carbon 37:1257–1264. https://doi.org/10.1016/S0008-6223(98)00322-4
Schaefer M (1991) Measurement of Adsorption-Isotherms by Means of Gas Chromatography
Seyhi B, Drogui P, Buelna G, Blais JF (2011) Modeling of sorption of bisphenol A in sludge obtained from a membrane bioreactor process. Chem Eng J 172:61–67. https://doi.org/10.1016/j.cej.2011.05.065
Shafeeyan MS, Daud WMAW, Houshmand A, Shamiri A (2010) A review on surface modification of activated carbon for carbon dioxide adsorption. J Anal Appl Pyrolysis 89:143–151. https://doi.org/10.1016/j.jaap.2010.07.006
Shafeeyan MS, Daud WMAW, Shamiri A, Aghamohammadi N (2015) Modeling of carbon dioxide adsorption onto ammonia-modified activated carbon: kinetic analysis and breakthrough behavior. Energy Fuel 29:6565–6577. https://doi.org/10.1021/acs.energyfuels.5b00653
Sing KSW, Everett DH, Haul RAW et al (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (recommendations 1984). Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Singh J, Bhunia H, Basu S (2018) CO2 adsorption on oxygen enriched porous carbon monoliths: Kinetics, isotherm and thermodynamic studies. J Ind Eng Chem 60:321–332. https://doi.org/10.1016/j.jiec.2017.11.018
Spokas KA, Novak JM, Stewart CE et al (2011) Qualitative analysis of volatile organic compounds on biochar. Chemosphere 85:869–882. https://doi.org/10.1016/j.chemosphere.2011.06.108
Srivastava VC, Mall ID, Mishra IM (2007) Adsorption thermodynamics and isosteric heat of adsorption of toxic metal ions onto bagasse fly ash (BFA) and rice husk ash (RHA). Chem Eng J 132:267–278. https://doi.org/10.1016/j.cej.2007.01.007
Tamez Uddin M, Rukanuzzaman M, Maksudur Rahman Khan M, Akhtarul Islam M (2009) Adsorption of methylene blue from aqueous solution by jackfruit (Artocarpus heteropyllus) leaf powder: a fixed-bed column study. J Environ Manag 90:3443–3450. https://doi.org/10.1016/j.jenvman.2009.05.030
Thouchprasitchai N, Pintuyothin N, Pongstabodee S (2017) Optimization of CO2 adsorption capacity and cyclical adsorption/desorption on tetraethylenepentamine-supported surface-modified hydrotalcite. J Environ Sci 65:293–305. https://doi.org/10.1016/j.jes.2017.02.015
Tiwari D, Goel C, Bhunia H, Bajpai PK (2017) Dynamic CO2 capture by carbon adsorbents: Kinetics, isotherm and thermodynamic studies. Sep Purif Technol 181:107–122. https://doi.org/10.1016/j.seppur.2017.03.014
Valenciano R, Aylón E, Izquierdo MT (2015) A critical short review of equilibrium and kinetic adsorption models for VOCs breakthrough curves modelling. Adsorpt Sci Technol 33:851–869. doi: https://doi.org/10.1260/0263-6174.33.10.851
Wang Q, Luo J, Zhong Z, Borgna A (2011) CO2 capture by solid adsorbents and their applications: current status and new trends. Energy Environ Sci 4:42–55. https://doi.org/10.1039/C0EE00064G
Wang X, Chen L, Guo Q (2014) Development of hybrid amine-functionalized MCM-41 sorbents for CO2 capture. Chem Eng J 260:573–581. https://doi.org/10.1016/j.cej.2014.08.107
Wang J, Huang H, Wang M, Yao L, Qiao W, Long D, Ling L (2015) Direct capture of low-concentration CO<inf>2</inf> on mesoporous carbon-supported solid amine adsorbents at ambient temperature. Ind Eng Chem Res 54:5319–5327. https://doi.org/10.1021/acs.iecr.5b01060
Wu J, Zhou L, Sun Y, Su W, Zhou Y (2007) Measurement and prediction of adsorption equilibrium for a H2/N2/CH4/CO2 mixture. AICHE J 53:1178–1191. https://doi.org/10.1002/aic
Yaumi AL, Bakar MZA, Hameed BH (2018) Melamine-nitrogenated mesoporous activated carbon derived from rice husk for carbon dioxide adsorption in fixed-bed. Energy 155:46–55. https://doi.org/10.1016/j.energy.2018.04.183
Zhu T, Heo HJ, Row KH (2010) Optimization of crude polysaccharides extraction from Hizikia fusiformis using response surface methodology. Carbohydr Polym 82:106–110
Acknowledgments
We would like to express our gratitude to Dr. Andrew Carrier, Postdoctoral Fellow in Cape Breton University for productive comments and discussion.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 623 kb)
Rights and permissions
About this article
Cite this article
Bamdad, H., Hawboldt, K., MacQuarrie, S. et al. Application of biochar for acid gas removal: experimental and statistical analysis using CO2. Environ Sci Pollut Res 26, 10902–10915 (2019). https://doi.org/10.1007/s11356-019-04509-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04509-3