Abstract
This study evaluates the chemistry, kinetics, and mass transfer aspects of the removal of NO and SO2 simultaneously from flue gas induced by the combined heat and Fe2+ activation of aqueous persulfate. The work involves experimental studies and the development of a mathematical model utilizing a comprehensive reaction scheme for detailed process evaluation, and to validate the results of an experimental study at 30–70 °C, which demonstrated that both SO2 and Fe2+ improved NO removal, while the SO2 is almost completely removed. The model was used to correlate experimental data, predict reaction species and nitrogen-sulfur (N-S) product concentrations, to obtain new kinetic data, and to estimate mass transfer coefficient (KLa) for NO and SO2 at different temperatures. The model percent conversion results appear to fit the data remarkably well for both NO and SO2 in the temperature range of 30–70 °C. The conversions ranged from 43.2 to 76.5% and 98.9 to 98.1% for NO and SO2, respectively, in the 30–70 °C range. The model predictions at the higher temperature of 90 °C were 90.0 and 97.4% for NO and SO2, respectively. The model also predicted decrease in KLa for SO2 of 1.097 × 10−4 to 8.88 × 10−5 s−1 (30–90 °C) and decrease in KLa for NO of 4.79 × 10−2 to 3.67 × 10−2 s−1 (30–50 °C) but increase of 4.36 × 10−2 to 4.90 × 10−2 s−1 at higher temperatures (70–90 °C). This emerging sulfate-radical-based process could be applied to the treatment of flue gases from combustion sources.

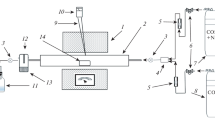
Graphical abstract











Similar content being viewed by others
References
Adewuyi YG (2005a) Sonochemistry in environmental remediation. 1. Combinative and hybrid sonophotochemical oxidation processes for the treatment of pollutants in water. Environ Sci Technol 39:3409–3420. https://doi.org/10.1021/es049138y
Adewuyi YG (2005b) Sonochemistry in environmental remediation. 2. Heterogeneous sonophotocatalytic oxidation processes for the treatment of pollutants in water. Environ Sci Technol 39:8557–8570. https://doi.org/10.1021/es0509127
Adewuyi YG, Carmichael GR (1982) A theoretical investigation of gaseous absorption by water droplets from SO2-HNO3-NH3-CO2-HCl mixtures. Atmos Environ 16:719–729. https://doi.org/10.1016/0004-6981(82)90389-4
Adewuyi YG, Khan NE (2012) Modeling the ultrasonic cavitation-enhanced removal of nitrogen oxide in a bubble column reactor. AIChE J 58:2397–2411. https://doi.org/10.1002/aic.12751
Adewuyi YG, Khan MA (2015) Nitric oxide removal by combined persulfate and ferrous–EDTA reaction systems. Chem Eng J 281:575–587. https://doi.org/10.1016/j.cej.2015.06.114
Adewuyi YG, Khan MA (2016) Nitric oxide removal from flue gas by combined persulfate and ferrous–EDTA solutions: effects of persulfate and EDTA concentrations, temperature, pH and SO2. Chem Eng J 304:793–807. https://doi.org/10.1016/j.cej.2016.06.071
Adewuyi YG, Owusu SO (2003) Aqueous absorption and oxidation of nitric oxide with oxone for the treatment of tail gases: process feasibility, stoichiometry, reaction pathways, and absorption rate. Ind Eng Chem Res 42:4084–4100. https://doi.org/10.1021/ie020709
Adewuyi YG, Owusu SO (2006) Ultrasound-induced aqueous removal of nitric oxide from flue gases: effects of sulfur dioxide, chloride, and chemical oxidant. J Phys Chem A 110:11098–11107. https://doi.org/10.1021/jp0631634
Adewuyi YG, Sakyi NY (2013a) Removal of nitric oxide by aqueous sodium persulfate simultaneously activated by temperature and Fe2+ in a lab-scale bubble reactor. Ind Eng Chem Res 52:14687–14697. https://doi.org/10.1021/ie4025177
Adewuyi YG, Sakyi NY (2013b) Simultaneous absorption and oxidation of nitric oxide and sulfur dioxide by aqueous solutions of sodium persulfate activated by temperature. Ind Eng Chem Res 52:11702–11711. https://doi.org/10.1021/ie401649s
Adewuyi YG, Cho S-Y, Tsay R-P, Carmichael GR (1984) Importance of formaldehyde in cloud chemistry. Atmos Environ 18:2413–2420. https://doi.org/10.1016/0004-6981(84)90011-8
Adewuyi Y, He X, Shaw H, Lolertpihop W (1999) Simultaneous absorption and oxidation of NO and SO2 by aqueous solutions of sodium chlorite. Chem Eng Commun 174:21–51. https://doi.org/10.1080/00986449908912788
Adewuyi YG, Khan MA, Sakyi NY (2014) Kinetics and modeling of the removal of nitric oxide by aqueous sodium persulfate simultaneously activated by temperature and Fe2+. Ind Eng Chem Res 53:828–839. https://doi.org/10.1021/ie402801b
Adewuyi YG, Sakyi NY, Khan MA (2018) Simultaneous removal of NO and SO2 from flue gas by combined heat and Fe2+ activated aqueous persulfate solutions. Chemosphere 193:1216–1225. https://doi.org/10.1016/j.chemosphere.2017.11.086
Aher A, Papp J, Colburn A, Wan H, Hatakeyama E, Prakash P, Weaver B, Bhattacharyya D (2017) Naphthenic acids removal from high TDS produced water by persulfate mediated iron oxide functionalized catalytic membrane, and by nanofiltration. Chem Eng J 327:573–583. https://doi.org/10.1016/j.cej.2017.06.128
Ajdari S, Normann F, Andersson K, Johnsson F (2015) Modeling the nitrogen and sulfur chemistry in pressurized flue gas systems. Ind Eng Chem Res 54:1216–1227. https://doi.org/10.1021/ie504038s
Ajdari S, Normann F, Andersson K, Johnsson F (2016) Reduced mechanism for nitrogen and sulfur chemistry in pressurized flue gas systems. Ind Eng Chem Res 55:5514–5525. https://doi.org/10.1021/acs.iecr.5b04670
Andreoni V, Miola A, Perujo A (2008) Cost effectiveness analysis of the emission abatement in the shipping sector emissions. European Commission Joint Research Centre Institute for Environment and Sustainability, Luxembourg. https://doi.org/10.2788/77899
Buxton GV, Greenstock CL, Helman WP, Ross AB (1988) Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (·OH/·O−) in aqueous solution. J Phys Chem Ref Data 17:513–886. https://doi.org/10.1063/1.555805
Cai X, Sun W, Xu C, Cao L, Yang J (2016) Highly selective catalytic reduction of NO via SO2/H2O-tolerant spinel catalysts at low temperature. Environ Sci Pollut Res 23:18609–18620. https://doi.org/10.1007/s11356-016-7061-y
Chiu C-H, Hsi H-C, Lin H-P (2015) Multipollutant control of Hg/SO2/NO from coal-combustion flue gases using transition metal oxide-impregnated SCR catalysts. Catal Today 245:2–9. https://doi.org/10.1016/j.cattod.2014.09.008
Dai Y, Deng T, Wang J, Xu K (2004) Enhancement of oxygen gas–liquid mass transfer with colloidal gas aphron dispersions. Colloids Surf A Physicochem Eng Asp 240:165–171. https://doi.org/10.1016/j.colsurfa.2004.03.018
Ding F, Chen H, Zhang S, Zhao T, Liu N (2017) Effect of chelating agents on Reactive Green 19 decolorization through Fe0-activated persulfate oxidation process. J Environ Manag 200:325–334. https://doi.org/10.1016/j.jenvman.2017.05.089
Drzewicz P, Perez-Estrada L, Alpatova A, Martin JW, Gamal El-Din M (2012) Impact of peroxydisulfate in the presence of zero valent iron on the oxidation of cyclohexanoic acid and naphthenic acids from oil sands process-affected water. Environ Sci Technol 46:8984–8991. https://doi.org/10.1021/es3011546
Fang G-D, Dionysiou DD, Wang Y, Al-Abed SR, Zhou D-M (2012) Sulfate radical-based degradation of polychlorinated biphenyls: effects of chloride ion and reaction kinetics. J Hazard Mater 227-228:394–401. https://doi.org/10.1016/j.jhazmat.2012.05.074
Fogler HS (2006) Elements of chemical reaction engineering, 4th edn. Prentice Hall, Upper Saddle River
Geißler M, Van Eldik R (1994) Product identification and kinetic data for the reaction of S(IV) with N(III) oxoacids in aqueous solution in the absence and presence of metal ions. Polyhedron 13:2983–2991. https://doi.org/10.1016/S0277-5387(00)83418-X
Gligorovski S, Strekowski R, Barbati S, Vione D (2015) Environmental implications of hydroxyl radicals (•OH). Chem Rev 115:13051–13092. https://doi.org/10.1021/cr500310b
Han D, Wan J, Ma Y, Wang Y, Li Y, Li D, Guan Z (2015) New insights into the role of organic chelating agents in Fe(II) activated persulfate processes. Chem Eng J 269:425–433. https://doi.org/10.1016/j.cej.2015.01.106
House DA (1962) Kinetics and mechanism of oxidations by peroxydisulfate. Chem Rev 62:185–203. https://doi.org/10.1021/cr60217a001
Huang K-C, Zhao Z, Hoag GE, Dahmani A, Block PA (2005) Degradation of volatile organic compounds with thermally activated persulfate oxidation. Chemosphere 61:551–560. https://doi.org/10.1016/j.chemosphere.2005.02.032
Hutson ND, Krzyzynska R, Srivastava RK (2008) Simultaneous removal of SO2, NOX, and Hg from coal flue gas using a NaClO2-enhanced wet scrubber. Ind Eng Chem Res 47:5825–5831. https://doi.org/10.1021/ie800339p
Ibrahim S (2016) Process evaluation of a SOx and NOx exhaust gas cleaning concept for marine application. Chalmers University of Technology, Gothenburg
Janik I, Bartels DM, Jonah CD (2007) Hydroxyl radical self-recombination reaction and absorption spectrum in water up to 350 °C. J Phys Chem A 111:1835–1843. https://doi.org/10.1021/jp065992v
Jethani KR, Suchak NJ, Joshi JB (1990) Selection of reactive solvent for pollution abatement of NOx. Gas Sep Purif 4:8–28. https://doi.org/10.1016/0950-4214(90)80023-E
Khan NE, Adewuyi YG (2010) Absorption and oxidation of nitric oxide (NO) by aqueous solutions of sodium persulfate in a bubble column reactor. Ind Eng Chem Res 49:8749–8760. https://doi.org/10.1021/ie100607u
Khan NE, Adewuyi YG (2011) A new method of analysis of peroxydisulfate using ion chromatography and its application to the simultaneous determination of peroxydisulfate and other common inorganic ions in a peroxydisulfate matrix. J Chromatogr A 1218:392–397. https://doi.org/10.1016/j.chroma.2010.11.038
Khan MA, Adewuyi YG (2017) High pressure reactive distillation simulation and optimization for the esterification of pyrolysis bio-oil. Process Eng J 1:73–85
Kolthoff IM, Miller IK (1951) The chemistry of persulfate. I. The kinetics and mechanism of the decomposition of the persulfate ion in aqueous medium. J Am Chem Soc 73:3055–3059. https://doi.org/10.1021/ja01151a024
Korell J, Paur H-R, Seifert H, Andersson S (2009) Simultaneous removal of mercury, PCDD/F, and fine particles from flue gas. Environ Sci Technol 43:8308–8314. https://doi.org/10.1021/es901289g
Krzyzynska R, Hutson ND (2012) Effect of solution pH on SO2, NOx, and Hg removal from simulated coal combustion flue gas in an oxidant-enhanced wet scrubber. J Air Waste Manage Assoc 62:212–220. https://doi.org/10.1080/10473289.2011.642951
Liang C, Guo Y-Y (2010) Mass transfer and chemical oxidation of naphthalene particles with zerovalent iron activated persulfate. Environ Sci Technol 44:8203–8208. https://doi.org/10.1021/es903411a
Liang C, Wang Z-S, Mohanty N (2006) Influences of carbonate and chloride ions on persulfate oxidation of trichloroethylene at 20 °C. Sci Total Environ 370:271–277. https://doi.org/10.1016/j.scitotenv.2006.08.028
Liang C, Huang C-F, Chen Y-J (2008) Potential for activated persulfate degradation of BTEX contamination. Water Res 42:4091–4100. https://doi.org/10.1016/j.watres.2008.06.022
Liang C, Chen Y-J, Chang K-J (2009) Evaluation of persulfate oxidative wet scrubber for removing BTEX gases. J Hazard Mater 164:571–579. https://doi.org/10.1016/j.jhazmat.2008.08.056
Littlejohn D, Chang SG (1984) Identification of species in a wet flue gas desulfurization and denitrification system by laser Raman spectroscopy. Environ Sci Technol 18:305–310. https://doi.org/10.1021/es00123a004
Littlejohn D, Chang SG (1986) Determination of nitrogen-sulfur compounds by ion chromatography. Anal Chem 58:158–160. https://doi.org/10.1021/ac00292a038
Littlejohn D, Chang SG (1994) Oxidative decomposition of nitrogen-sulfur oxides. Ind Eng Chem Res 33:515–518. https://doi.org/10.1021/ie00027a007
Littlejohn D, Hu KY, Chang SG (1986) Kinetics of the reaction of nitric oxide with sulfite and bisulfite ions in aqueous solution. Inorg Chem 25:3131–3135. https://doi.org/10.1021/ic00238a007
Liu Y, Adewuyi YG (2016) A review on removal of elemental mercury from flue gas using advanced oxidation process: chemistry and process. Chem Eng Res Des 112:199–250. https://doi.org/10.1016/j.cherd.2016.06.024
Liu Y, Zhang J (2017) Removal of NO from flue gas using UV/S2O82− process in a novel photochemical impinging stream reactor. AIChE J 63:2968–2980. https://doi.org/10.1002/aic.15633
Matzek LW, Carter KE (2016) Activated persulfate for organic chemical degradation: a review. Chemosphere 151:178–188. https://doi.org/10.1016/j.chemosphere.2016.02.055
Neta P, Huie RE (1986) Rate constants for reactions of nitrogen oxide (NO3) radicals in aqueous solutions. J Phys Chem 90:4644–4648. https://doi.org/10.1021/j100410a035
Normann F, Jansson E, Petersson T, Andersson K (2013) Nitrogen and sulphur chemistry in pressurised flue gas systems: a comparison of modelling and experiments. Int J Greenhouse Gas Control 12:26–34. https://doi.org/10.1016/j.ijggc.2012.11.012
Oblath SB, Markowitz SS, Novakov T, Chang SG (1981) Kinetics of the formation of hydroxylamine disulfonate by reaction of nitrite with sulfites. J Phys Chem 85:1017–1021. https://doi.org/10.1021/j150608a018
Oblath SB, Markowitz SS, Novakov T, Chang SG (1982) Kinetics of the initial reaction of nitrite ion in bisulfite solutions. J Phys Chem 86:4853–4857. https://doi.org/10.1021/j100222a005
Owusu SO, Adewuyi YG (2006) Sonochemical removal of nitric oxide from flue gases. Ind Eng Chem Res 45:4475–4485. https://doi.org/10.1021/ie0509692
Palash SM, Kalam MA, Masjuki HH, Masum BM, Rizwanul Fattah IM, Mofijur M (2013) Impacts of biodiesel combustion on NOx emissions and their reduction approaches. Renew Sust Energ Rev 23:473–490. https://doi.org/10.1016/j.rser.2013.03.003
Qian Y, Guo X, Zhang Y, Peng Y, Sun P, Huang C-H, Niu J, Zhou X, Crittenden JC (2016) Perfluorooctanoic acid degradation using uv–persulfate process: modeling of the degradation and chlorate formation. Environ Sci Technol 50:772–781. https://doi.org/10.1021/acs.est.5b03715
Rezaei F, Rownaghi AA, Monjezi S, Lively RP, Jones CW (2015) SOx/NOx removal from flue gas streams by solid adsorbents: a review of current challenges and future directions. Energy Fuel 29:5467–5486. https://doi.org/10.1021/acs.energyfuels.5b01286
Seinfeld JH, Pandis SN (2006) Atmospheric chemistry and physics: from air pollution to climate change. Second edn. Wiley, New Jersey
Skalska K, Miller JS, Ledakowicz S (2010) Trends in NOx abatement: a review. Sci Total Environ 408:3976–3989. https://doi.org/10.1016/j.scitotenv.2010.06.001
Susianto PM, Zoulalian A (2001) Influence of the pH on the interactions between nitrite and sulfite ions. Kinetic of the reaction at pH 4 and 5. Ind Eng Chem Res 40:6068–6072. https://doi.org/10.1021/ie010016d
Susianto PM, Pétrissans A, Zoulalian A (2005) Experimental study and modelling of mass transfer during simultaneous absorption of SO2 and NO2 with chemical reaction. Chem Eng Process Process Intensif 44:1075–1081. https://doi.org/10.1016/j.cep.2005.03.001
Tsitonaki A, Petri B, Crimi M, Mosbaek H, Siegrist RL, Bjerg PL (2010) In situ chemical oxidation of contaminated soil and groundwater using persulfate: a review. Crit Rev Environ Sci Technol 40:55–91. https://doi.org/10.1080/10643380802039303
Turšič J, Grgić I, Bizjak M (2001) Influence of NO2 and dissolved iron on the S(IV) oxidation in synthetic aqueous solution. Atmos Environ 35:97–104. https://doi.org/10.1016/S1352-2310(00)00283-1
Wu B, Xiong Y, Ru J, Feng H (2016) Removal of NO from flue gas using heat-activated ammonium persulfate aqueous solution in a bubbling reactor. RSC Adv 6:33919–33930. https://doi.org/10.1039/C6RA01524G
Xiao R, Luo Z, Wei Z, Luo S, Spinney R, Yang W, Dionysiou DD (2018) Activation of peroxymonosulfate/persulfate by nanomaterials for sulfate radical-based advanced oxidation technologies. Curr Opin Chem Eng 19:51–58. https://doi.org/10.1016/j.coche.2017.12.005
Xu W, Adewuyi YG, Liu Y, Wang Y (2018) Removal of elemental mercury from flue gas using CuOx and CeO2 modified rice straw chars enhanced by ultrasound. Fuel Process Technol 170:21–31. https://doi.org/10.1016/j.fuproc.2017.10.017
Acknowledgments
The authors wish to acknowledge the contribution of the National Science Foundation (NSF) for the funding received via Grant CBET-0651811.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Marcus Schulz
Highlights
• An advanced oxidation process for the simultaneous removal of SO2 and NO was developed
• The process involved combined heat and Fe2+ activation of Na2S2O8
• Existing and newly proposed reaction pathways for the combined NO/SO2 removal are discussed
• A comprehensive model was developed and used to predict species concentration and kinetic parameters
Electronic supplementary material
ESM 1
(DOCX 31 kb)
Rights and permissions
About this article
Cite this article
Adewuyi, Y.G., Khan, M.A. Simultaneous NO and SO2 removal by aqueous persulfate activated by combined heat and Fe2+: experimental and kinetic mass transfer model studies. Environ Sci Pollut Res 27, 1186–1201 (2020). https://doi.org/10.1007/s11356-018-2453-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2453-9