Abstract
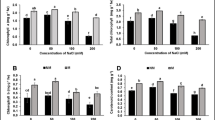
Agriculture accounts for ~ 70% of all water use and the world population is increasing annually; soon more people will need to be fed, while also using less water. The use of plant-associated bacteria (PAB) is an eco-friendly alternative that can increase crop water use efficiency. This work aimed to study the effect of some PAB on increasing soybean tolerance to drought stress, the mechanisms of the drought tolerance process, and the effect of the PAB on promoting plant growth and on the biocontrol of Sclerotinia sclerotiorum. PAB were isolated from soybean rhizosphere and S. sclerotiorum sclerotia. The strains identified as UFGS1 (Bacillus subtilis), UFGS2 (Bacillus thuringiensis), UFGRB2 and UFGRB3 (Bacillus cereus) were selected on their ability to grow in media with reduced water activity. Soybean plants were inoculated with the PAB and evaluated for growth promotion, physiological and molecular parameters, after drought stress. Under drought stress, UFGS2 and UFGRB2 sustained potential quantum efficiency of PSII (Fv/Fm), while a decrease was found in the control plants. Moreover, UFGS2 and UFGRB3 maintained the photosynthetic rates in non-stressed conditions compared to the control. UFGS2-treated plants showed a higher stomatal conductance and higher transpiration than the control, after drought stress. Some PAB-treated plants also had other beneficial phenotypes, such as increases in fresh and dried biomass relative to the control. Differential gene expression analysis of genes involved in plant stress pathways shows changes in expression in PAB-treated plants. Results from this study suggest that PAB can mitigate drought stress in soybean and may improve water efficiency under certain conditions.







Similar content being viewed by others
References
Berendsen RL, Pieterse CM, Bakker PA (2012) The rhizosphere microbiome and plant health. Trends Plant Sci 17:478–486. https://doi.org/10.1016/j.tplants.2012.04.001
Berg G, Rybakova D, Grube M, Köberl M (2016) The plant microbiome explored: implications for experimental botany. J Exp Bot 67:995–1002. https://doi.org/10.1093/jxb/erv466
Boiteux LS, Fonseca MEN, Simon PW (1999) Effects of plant tissue and DNA purification method on randomly amplified polymorphic DNA-based genetic fingerprinting analysis in carrot. J Am Soc Hort Sci 124:32–38
Chen Y, Chen P, de los Reyes B (2006) Differential responses of the cultivated and wild species of soybean to dehydration stress. Crop Sci 46:2041–2046
Chen W, Yao Q, Patil GB, Agarwal G, Deshmukh RK, Lin L, Wang B, Wang Y, Prince SJ, Song L, Xu D, An YC, Valliyodan B, Varshney RK, Nguyen HT (2016) Identification and comparative analysis of differential gene expression in soybean leaf tissue under drought and flooding stress revealed by RNA-seq. Front Plant Sci. https://doi.org/10.3389/fpls.2016.01044
Cohen D, Bogeat-Triboulot MB, Tisserant E, Balzergue S, Martin-Magniette ML, Lelandais G, Ningre N, Renou JP, Tamby JP, le Thiec D, Hummel I (2010) Comparative transcriptomics of drought responses in Populus: a meta-analysis of genome-wide expression profiling in mature leaves and root apices across two genotypes. BMC Genomics 11:630. https://doi.org/10.1186/1471-2164-11-630
Ferreira DF (2011) Program SISVAR a Computer statistic analysis system. Ciência e Agrotecnologia 35:1039–1042. https://doi.org/10.1590/S1413-70542011000600001
Firoz MJ, Xiao X, Zhu FX, Fu YP, Jiang DH, Schnabel G, Luo CX (2016) Exploring mechanisms of resistance to dimethachlone in Sclerotinia sclerotiorum. Pest Manag Sci 72:770–779. https://doi.org/10.1002/ps.4051
Gamalero E, Berta G, Glick BR (2009) The use of microorganisms to facilitate the growth of plants in saline soils. In: Khan MS, Zaidi A, Musarrat J (eds) Microbial strategies for crop improvement. Springer, Berlin, pp 1–22
Genty B, Briantais J, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica et Biphysica Acta 990:87–92. https://doi.org/10.1016/S0304-4165(89)80016-9
Geraldine AM, Lopes FAC, Carvalho DDC, Barbosa ET, Rodrigues AR, Brandão RS, Ulhoa CJ, Lobo Junior M (2013) Cell wall-degrading enzymes and parasitism of sclerotia are key factors on field biocontrol of white mold by Trichoderma spp. Biol Control 67:308–316. https://doi.org/10.1016/j.biocontrol.2013.09.013
Heydari A, Pessarakli M (2010) A review on biological control of fungal plant pathogens using microbial antagonists. J Biol Sci 10:273–290
Hu X, Roberts DP, Maul JE, Emche SE, Liao X, Guo X, Liu Y, McKenna LF, Buyer JS, Liu S (2011) Formulations of the endophytic bacterium Bacillus subtilis Tu-100 suppress Sclerotinia sclerotiorum on oilseed rape and improve plant vigor in field trials conducted at separate locations. Can J Microbiol 57:539–546. https://doi.org/10.1139/W11-041
Hu X, Roberts DP, Xie L, Maul JE, Yu C, Li Y, Jiang M, Liao X, Che Z, Liao X (2014) Formulations of Bacillus subtilis BY-2 suppress Sclerotinia sclerotiorum on oilseed rape in the field. Biol Control 70:54–64. https://doi.org/10.1016/j.biocontrol.2013.12.005
Jetiyanon K (2007) Defensive-related enzyme response in plants treated with a mixture of Bacillus strains (IN937a and IN937b) against different pathogens. Biol Control 42:178–185. https://doi.org/10.1016/j.biocontrol.2007.05.008
Kamal MM, Savocchia S, Lindbeck KD, Ash GJ (2016) Biology and biocontrol of Sclerotinia sclerotiorum (Lib.) de Bary in oilseed Brassicas. Australas Plant Pathol 45:1–14. https://doi.org/10.1007/s13313-015-0391-2
Kim YC, Glick B, Bashan Y, Ryu CM (2013) Enhancement of plant drought tolerance by microbes. In: Aroca R (ed) Plant responses to drought stress. Springer Verlag, Berlin, pp 383–413
Krannich CT, Maletzki L, Kurowsky C, Horn R (2015) Network candidate genes in breeding for drought tolerant crops. Int J Mol Sci 16:16378–16400. https://doi.org/10.3390/ijms160716378
Mahmood A, Turgay OC, Farooq M, Hayat R (2016) Seed biopriming with plant growth promoting rhizobacteria: a review. FEMS Microbiol Ecol 92. https://doi.org/10.1093/femsec/fiw112
Martins SJ, Medeiros FHV, Souza RM, Resende MLV, Ribeiro Junior PM (2013) Biological control of bacterial wilt of common bean by plant growth-promoting rhizobacteria. Biol Control 66:65–71
Martins SJ, Medeiros FHV, Souza RM, Vilela LAF (2014) Is the curtobacterium-wilt biocontrol temperature-dependent? Acta Sci Agron 36:409–415. https://doi.org/10.4025/actasciagron.v36i4.18018
Martins SJ, Medeiros FHV, de Souza RM, Faria AF, Cancellier EL, Silveira HRO, Rezende MLV, Guilherme LRG (2015a) Common bean growth and health promoted by rhizobacteria and the contribution of magnesium to the observed responses. Appl Soil Ecol 87:49–55. https://doi.org/10.1016/j.apsoil.2014.11.005
Martins SJ, Soares AC, Medeiros FHV, Santos DBC, Pozza EA (2015b) Contribution of host and environmental factors to the hyperparasitism of coffee rust under field conditions. Australas Plant Pathol 44:605–610. https://doi.org/10.1007/s13313-015-0375-2
Medeiros FHV, Martins SJ, Zucchi TD, Melo IS, Batista LR, Machado JC (2012) Biological control of mycotoxin-producing molds. Ciência e Agrotecnologia 36:483–497. https://doi.org/10.1590/S1413-70542012000500001
Mishra KB, Iannacone R, Petrozza A, Mishra A, Armentano N, la Vecchia G, Trtílek M, Cellini F, Nedbal L (2012) Engineered drought tolerance in tomato plants is reflected in chlorophyll fluorescence emission. Plant Sci 182:79–86. https://doi.org/10.1016/j.plantsci.2011
Muller B, Pantin F, Génard M, Turc O, Freixes S, Piques M, Gibon Y (2011) Water deficits uncouple growth from photosynthesis, increase C content, and modify the relationships between C and growth in sink organs. J Exp Bot 62:1715–1729. https://doi.org/10.1093/jxb/erq438
Nakaya A, Isobe SN (2012) Will genomic selection be a practical method for plant breeding? Ann Bot 110:13003–13016. https://doi.org/10.1093/aob/mcs109
Niu DD, Liu HX, Jiang CH, Wang YP, Wang QY, Jin HL, Guo JH (2011) The plant growth-promoting rhizobacterium Bacillus cereus AR156 induces systemic resistance in Arabidopsis thaliana by simultaneously activating salicylate–and jasmonate/ethylene-dependent signaling pathways. Mol Plant-Microbe Interact 24:533–542. https://doi.org/10.1094/MPMI-09-10-0213
Peres AP, Nasser LCB, Machado JC (2002) Use of semi-seletive media for detection of Sclerotinia sclerotiorum on bean and soybean seeds. Fitopatol Bras 27:123–127. https://doi.org/10.1590/S0100-41582002000200001
Prasad SR, Kamble UR, Sripathy KV, Bhaskar KU, Singh DP (2016) Seed bio-priming for biotic and abiotic stress management. In: Singh DP, Singh HB, Prabha R (eds) Microbial inoculants in sustainable agricultural productivity. Springer, Berlin, pp 211–228
Rahbarian R, Khavari-Nejad R, Ganjeali A, Bagheri A, Najafi F (2011) Drought stress effects on photosynthesis, chlorophyll fluorescence and water relations in tolerant and susceptible chickpea (Cicer arietinum L.) genotypes. Acta Biologica Cracoviensia 53:47–56
Sadeghipour O, Abbasi S (2012) Soybean response to drought and seed inoculation. World Appl Sci J 17:55–60
Shinozaki A, Yamaguchi-Shinozaki K (2007) Gene networks involved in drought stress response and tolerance. J Exp Bot 58:221–227. https://doi.org/10.1093/jxb/erl164
Staudinger C, Mehmeti-Tershani V, Gil-Quintana E, Gonzalez EM, Hofhansl F, Bachmann G (2016) Evidence for a rhizobia-induced drought stress response strategy in Medicago truncatula. J Proteome 136:202–213. https://doi.org/10.1016/j.jprot.2016.01.006
Stolf-Moreira R, Medri ME, Neumaier N, Lemos NG, Pimenta JA, Tobita S, Brogin RL, Marcelino-Guimaraes FC, Oliveira MCN, Farias JRB, Abdelnoor RV, Nepomuceno AL (2010) Soybean physiology and gene expression during drought. Genet Mol Res 9:1946–1956. https://doi.org/10.4238/vol9-4gmr851
Timmusk S, Behers L (2012) Rhizobacterial application for sustainable water management on the areas of limited water resources. Irrig Drain Syst Eng. https://doi.org/10.4172/2168-9768.1000e111
Timmusk S, Timmusk K, Behers L (2013) Rhizobacterial plant drought stress tolerance enhancement: towards sustainable water resource management and food security. J Food Security 1:6–10. https://doi.org/10.12691/jfs-1-1-2
Turdi M, Yang L (2016) Trace elements contamination and human health risk assessment in drinking water from the agricultural and pastoral areas of Bay County, Xinjiang, China. Int J Environ Res Public Health 13. https://doi.org/10.3390/ijerph13100938
Wang CJ, Yang W, Wang C, Gu C, Niu DD, Liu H-X (2012) Induction of drought tolerance in cucumber plants by a consortium of three plant growth- promoting rhizobacterium strains. PLoS One 7:e52565. https://doi.org/10.1371/journal.pone.0052565
Yoon S, Ha S, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Yu S, Liao F, Wang F, Wen W, Li J, Mei H, Luo L (2012) Identification of rice transcription factors associated with drought tolerance using ecotilling method. PLoS One 7:e30765. https://doi.org/10.1371/journal.pone.0030765
Acknowledgements
The authors express their appreciation to the Brazilian Agricultural Research Corporation (EMBRAPA) for kindly providing the Sclerotinia sclerotiorum strain BRM 29673 to be used in this work. We also thank Caroline Elizabeth Martins for providing an English review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Diane Purchase
Electronic supplementary material
Supplementary 1
Maximum Likelihood tree of 16S rRNA of subject strains. The tree was produced using the HKY85 model and complete deletion of missing data. The level of bootstrap support was calculated from 1500 replicates. The bar is equal to 0.01 substitutions per bp. (PNG 110 kb)
Rights and permissions
About this article
Cite this article
Martins, S.J., Rocha, G.A., de Melo, H.C. et al. Plant-associated bacteria mitigate drought stress in soybean. Environ Sci Pollut Res 25, 13676–13686 (2018). https://doi.org/10.1007/s11356-018-1610-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1610-5