Abstract
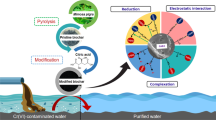
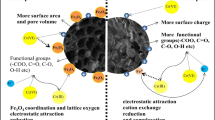
This study was conducted to understand the effects of ultraviolet (UV) irradiation on the physicochemical properties and the hexavalent chromium (Cr(VI)) removal ability of biochar. Structural and morphological analysis showed that UV irradiation increased the specific surface area of biochar and added a large amount of oxygen-containing functional groups (e.g., carboxyl, lactonic, and hydroxyl) to biochar’s surface. Batch sorption experimental results showed that UV-modified biochar (UVBC) produced at the pyrolysis temperature of 300 °C, the irradiated time of 24 h, and the irradiation distance of 40 mm exhibited excellent Cr(VI) removal ability (from 1.11 mg/g for BC to 20.04 mg/g for UVBC, a 18.1-fold increase). The adsorption kinetics and adsorption isotherm data agreed well with the pseudo-second-order model and Freundlich model, respectively. Experimental and modeling results suggest that the oxygen-containing functional groups and specific surface areas of biochars were notably increased by UV irradiation, which enhanced Cr(VI) adsorption by surface complexation. X-ray photoelectron spectroscopy (XPS) analysis of UVBC before and after reaction with Cr(VI) showed that reduction occurred during Cr(VI) adsorption. The energy consumption of UV modification is 2.7 MJ per gram of UVBC produced, which is comparable to that in activated carbon. The results showed that the method of UV modification of biochar is a very novel and effective method for the adsorption of Cr(VI) in solution.








Similar content being viewed by others
References
Ahmadi M, Kouhgardi E, Ramavandi B (2016) Physico-chemical study of dew melon peel biochar for chromium attenuation from simulated and actual wastewaters. Korean J Chem Eng 33(9):2589–2601. https://doi.org/10.1007/s11814-016-0135-1
Al-Turaif HA (2013) Surface morphology and chemistry of epoxy-based coatings after exposure to ultraviolet radiation. Prog Org Coat 76(4):677–681. https://doi.org/10.1016/j.porgcoat.2012.12.010
Al-Wabel MI, Al-Omran A, El-Naggar AH, Nadeem M, Usman AR (2013) Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour Technol 131:374–379. https://doi.org/10.1016/j.biortech.2012.12.165
Cao X, Harris W (2010) Properties of dairy-manure-derived biochar pertinent to its potential use in remediation. Bioresour Technol 101(14):5222–5228. https://doi.org/10.1016/j.biortech.2010.02.052
Chun Y, Sheng G, Chiou CT, Xing B (2004) Compositions and sorptive properties of crop residue-derived chars. Environ Sci Technol 38(17):4649–4655. https://doi.org/10.1021/es035034w
Dong X, Ma LQ, Li Y (2011) Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J Hazard Mater 190(1–3):909–915. https://doi.org/10.1016/j.jhazmat.2011.04.008
Dong X, Ma LQ, Zhu Y, Li Y, Gu B (2013) Mechanistic investigation of mercury sorption by Brazilian pepper biochars of different pyrolytic temperatures based on X-ray photoelectron spectroscopy and flow calorimetry. Environ Sci Technol 47(21):12156–12164. https://doi.org/10.1021/es4017816
Ellison MD, Buckley LK, Lewis GG, Smith CE, Siedlecka EM, Palchak CV, Malarchik JM (2009) Photochemical hydroboration−oxidation of single-walled carbon nanotubes. J Phys Chem C 113(43):18536–18541. https://doi.org/10.1021/jp905805k
Fan Y, Wang B, Yuan S, Wu X, Chen J, Wang L (2010) Adsorptive removal of chloramphenicol from wastewater by NaOH modified bamboo charcoal. Bioresour Technol 101(19):7661–7664. https://doi.org/10.1016/j.biortech.2010.04.046
Harikishore Kumar Reddy D, Lee S-M (2014) Magnetic biochar composite: facile synthesis, characterization, and application for heavy metal removal. Colloid Surf A 454:96–103. https://doi.org/10.1016/j.colsurfa.2014.03.105
Kim WK, Shim T, Kim YS, Hyun S, Ryu C, Park YK, Jung J (2013) Characterization of cadmium removal from aqueous solution by biochar produced from a giant Miscanthus at different pyrolytic temperatures. Bioresour Technol 138:266–270. https://doi.org/10.1016/j.biortech.2013.03.186
Le QT, Naumov S, Conard T, Franquet A, Muller M, Beckhoff B, Adelmann C, Struyf H, Gendt SD, Baklanov MR (2013) Mechanism of modification of fluorocarbon polymer by ultraviolet irradiation in oxygen atmosphere. Ecs J Solid State Sci Technol 2(5):N93–N98
Li Q, Ding W, Yong Y, Zeng X, Gao Y (2016) Effects of ultraviolet modification on physicochemical property and adsorption performance of biochar. Nanosci Nanotechnol Lett 8(11):978–984. https://doi.org/10.1166/nnl.2016.2184
Li Q, Jin BS, Huang YJ, Zhong ZP, Li B, Sun Y (2009) Preparation of biomass activated carbon by steam activation. J Southeast Univ 39(5):1008–1011
Li Z, Katsumi T, Inui T, Imaizumi S (2011) Woods charred at low temperatures and their modification for the adsorption of Cr(VI) ions from aqueous solution. Adsorpt Sci Technol 28(5):99–108
Lyu H, Gong Y, Gurav R, Tang J (2016a) Biochar application: essential soil microbial ecology. In: Ralebitso-Senior TK, Orr CH (eds) Potential application of biochar for bioremediation of contaminated systems. Elsevier Inc., Netherlands, pp 221–246
Lyu H, Gong Y, Tang J, Huang Y, Wang Q (2016b) Immobilization of heavy metals in electroplating sludge by biochar and iron sulfide. Environ Sci Pollut Res Int 23(14):14472–14488
Lyu HH, Tang JC, Huang Y, Gai LS, Zeng EY, Liber K, Gong YY (2017) Removal of hexavalent chromium from aqueous solutions by a novel biochar supported nanoscale iron sulfide composite. Chem Eng J 322:516–524. https://doi.org/10.1016/j.cej.2017.04.058
Ma Y, Liu WJ, Zhang N, Li YS, Jiang H, Sheng GP (2014) Polyethylenimine modified biochar adsorbent for hexavalent chromium removal from the aqueous solution. Bioresour Technol 169:403–408. https://doi.org/10.1016/j.biortech.2014.07.014
Maroušek J (2014) Significant breakthrough in biochar cost reduction. Clean Techn Environ Policy 16(8):1821–1825. https://doi.org/10.1007/s10098-014-0730-y
Maroušek J, Vochozka M, Plachý J, Žák J (2016) Glory and misery of biochar. Clean Techn Environ Policy 19(2):311–317
Mendez A, Tarquis AM, Saa-Requejo A, Guerrero F, Gasco G (2013) Influence of pyrolysis temperature on composted sewage sludge biochar priming effect in a loamy soil. Chemosphere 93(4):668–676. https://doi.org/10.1016/j.chemosphere.2013.06.004
Mimmo T, Panzacchi P, Baratieri M, Davies CA, Tonon G (2014) Effect of pyrolysis temperature on miscanthus (Miscanthus × giganteus) biochar physical, chemical and functional properties. Biomass Bioenergy 62:149–157. https://doi.org/10.1016/j.biombioe.2014.01.004
Nakamura T, Ohana T (2013) Photochemical modification of DLC films with oxygen functionalities and their chemical structure control. Diam Relat Mater 33:16–19. https://doi.org/10.1016/j.diamond.2012.12.013
Panda J, Sarkar P (2015) Biosorption of Cr(VI) by calcium alginate-encapsulated enterobacter aerogenes T2, in a semi-batch plug flow process. Water Air Soil Pollut 226(1):2157
Qiu M, Luo D, Ding X, Liu Y (2012) Comparison of the cost of chemical precipitation in the treatment of chromium containing wastewater. Environ Protect Circ Econ 3:61–62
Rajapaksha AU, Chen SS, Tsang DCW, Zhang M, Vithanage M, Mandal S, Gao B, Bolan NS, Ok YS (2016) Engineered/designer biochar for contaminant removal/immobilization from soil and water: potential and implication of biochar modification. Chemosphere 148:276–291. https://doi.org/10.1016/j.chemosphere.2016.01.043
Riverautrilla J, Sánchezpolo M, Gómezserrano V, Alvarez PM, Alvimferraz MC, Dias JM (2011) Activated carbon modifications to enhance its water treatment applications. An overview. J Hazard Mater 187(1–3):1–23. https://doi.org/10.1016/j.jhazmat.2011.01.033
Shen Y-S, Wang S-L, Tzou Y-M, Yan Y-Y, Kuan W-H (2012) Removal of hexavalent Cr by coconut coir and derived chars—the effect of surface functionality. Bioresour Technol 104:165–172. https://doi.org/10.1016/j.biortech.2011.10.096
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammes A, Jia Y, Wu Y, Nguyen ST, Ruoff RS (2007) Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45(7):1558–1565. https://doi.org/10.1016/j.carbon.2007.02.034
Sun K, Tang J, Gong Y, Zhang H (2015) Characterization of potassium hydroxide (KOH) modified hydrochars from different feedstocks for enhanced removal of heavy metals from water. Environ Sci Pollut Res Int 22(21):16640–16651. https://doi.org/10.1007/s11356-015-4849-0
Takagi H, Maruyama K, Yoshizawa N, Yamada Y, Sato Y (2004) XRD analysis of carbon stacking structure in coal during heat treatment. Fuel 83(17–18):2427–2433. https://doi.org/10.1016/j.fuel.2004.06.019
Tang J, Huang Y, Gong Y, Lyu H, Wang Q, Ma J (2016) Preparation of a novel graphene oxide/Fe-Mn composite and its application for aqueous Hg(II) removal. J Hazard Mater 316:151–158. https://doi.org/10.1016/j.jhazmat.2016.05.028
Tang J, Lv H, Gong Y, Huang Y (2015) Preparation and characterization of a novel graphene/biochar composite for aqueous phenanthrene and mercury removal. Bioresour Technol 196:355–363. https://doi.org/10.1016/j.biortech.2015.07.047
Tytlak A, Oleszczuk P, Dobrowolski R (2015) Sorption and desorption of Cr(VI) ions from water by biochars in different environmental conditions. Environ Sci Pollut Res Int 22(8):5985–5994. https://doi.org/10.1007/s11356-014-3752-4
Wang C, Gu L, Liu X, Zhang X, Cao L, Hu X (2016) Sorption behavior of Cr(VI) on pineapple-peel-derived biochar and the influence of coexisting pyrene. Int Biodeter Biodegr 111:78–84. https://doi.org/10.1016/j.ibiod.2016.04.029
Yan J, Han L, Gao W, Xue S, Chen M (2014) Biochar supported nanoscale zerovalent iron composite used as persulfate activator for removing trichloroethylene. Bioresour Technol 175C:269–274
Yi M, Shen ZG (2015) A review on mechanical exfoliation for the scalable production of graphene. J Mater Chem A 3(22):11700–11715. https://doi.org/10.1039/C5TA00252D
Yin QQ, Wang SR (2013) Analysis and activation of biomass rapid pyrolysis carbon. J Basic Sci Eng 21(1):45–53
Zelmanov G, Semiat R (2011) Iron (Fe+3) oxide/hydroxide nanoparticles-based agglomerates suspension as adsorbent for chromium (Cr+6) removal from water and recovery. Sep Purif Technol 80(2):330–337. https://doi.org/10.1016/j.seppur.2011.05.016
Zhou L, Liu Y, Liu S, Yin Y, Zeng G, Tan X, Hu X, Hu X, Jiang L, Ding Y, Liu S, Huang X (2016) Investigation of the adsorption-reduction mechanisms of hexavalent chromium by ramie biochars of different pyrolytic temperatures. Bioresour Technol 218:351–359. https://doi.org/10.1016/j.biortech.2016.06.102
Zhou Y, Gao B, Zimmerman AR, Fang J, Sun Y, Cao X (2013) Sorption of heavy metals on chitosan-modified biochars and its biological effects. Chem Eng J 231:512–518. https://doi.org/10.1016/j.cej.2013.07.036
Zhu N, Yan T, Qiao J, Cao H (2016) Adsorption of arsenic, phosphorus and chromium by bismuth impregnated biochar: adsorption mechanism and depleted adsorbent utilization. Chemosphere 164:32–40. https://doi.org/10.1016/j.chemosphere.2016.08.036
Acknowledgments
The research was supported by (1) Tianjin Science and Technology Program (16JCTPJC51100), (2) National Natural Science Foundation of China (41473070), and (3) The National Water Pollution Control and Treatment Science and Technology Major Project (2015ZX07203-011-06).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Guilherme L. Dotto
Rights and permissions
About this article
Cite this article
Peng, Z., Zhao, H., Lyu, H. et al. UV modification of biochar for enhanced hexavalent chromium removal from aqueous solution. Environ Sci Pollut Res 25, 10808–10819 (2018). https://doi.org/10.1007/s11356-018-1353-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1353-3