Abstract
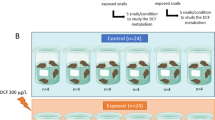
Human pharmaceuticals, such as nonsteroidal anti-inflammatory drugs (NSAIDs), are an emerging threat to marine organisms. NSAIDs act through inhibition of cyclooxygenase (COX) conversion of arachidonic acid into prostaglandins. One experiment was carried out whereby marine mussels were exposed for 72 h to 1 and 100 μg/L diclofenac (DCF). A specific and sensitive method using liquid chromatography high-resolution tandem mass spectrometry was developed to quantify DCF in mussel tissues. The developed method could also clearly identify and quantify COX products, i.e., prostaglandin levels, and be used to assess their modulation following DCF exposure. Prostaglandin-D2 (PGD2) was always found below the detection limit (20 μg/kg dry weight (dw)). Basal prostaglandin-E2 (PGE2) concentrations ranged from below the detection limit to 202 μg/kg dw. Exposure of 100 μg/L resulted in a significant reduction in PGE2 levels, whereas a downward trend was observed at 1 μg/L exposure. No difference was observed for prostaglandin-F2α (PGF2α) levels between controls and exposed organisms.


Similar content being viewed by others
References
Afonso-Olivares C, Torres-Padron ME, Sosa-Ferrera Z, Santana-Rodriguez JJ (2013) Assessment of the presence of pharmaceutical compounds in seawater samples from coastal area of gran Canaria Island (Spain). Antibiotics (Basel) 2:274–287. doi:10.3390/antibiotics2020274
Arpin-Pont L, Bueno MJM, Gomez E, Fenet H (2016) Occurrence of PPCPs in the marine environment: a review. Environ Sci Pollut Res Int 23:4978–4991. doi:10.1007/s11356-014-3617-x
Bhandari K, Venables B (2011) Ibuprofen bioconcentration and prostaglandin {E2} levels in the bluntnose minnow Pimephales notatus. Comp Biochem Physiol C: Toxicol Pharmacol 153:251–257. doi:10.1016/j.cbpc.2010.11.004
Boillot C, Martinez Bueno MJ, Munaron D et al (2015) In vivo exposure of marine mussels to carbamazepine and 10-hydroxy-10,11-dihydro-carbamazepine: Bioconcentration and metabolization. Sci Total Environ 532:564–570. doi:10.1016/j.scitotenv.2015.05.067
Bonnefille B, Arpin-Pont L, Gomez E et al (2017) Metabolic profiling identification of metabolites formed in Mediterranean mussels (Mytilus galloprovincialis) after diclofenac exposure. Sci Total Environ 583:257–268. doi:10.1016/j.scitotenv.2017.01.063
Chakraborty K, Chakkalakal SJ, Joseph D (2014) Response of pro-inflammatory prostaglandin contents in anti-inflammatory supplements from green mussel Perna Viridis L. in a time-dependent accelerated shelf-life study. J Funct Foods 7:527–540. doi:10.1016/j.jff.2014.01.003
Commission Implementing Decision (EU) 2015/495 of 20 March 2015, establishing a watch list of substances for Union-wide monitoring in the field of water policy pursuant to Directive 2008/105/EC of the European Parliament and of the Council
Courant F, Antignac J-P, Dervilly-Pinel G, Le Bizec B (2014) Basics of mass spectrometry based metabolomics. Proteomics 14:2369–2388. doi:10.1002/pmic.201400255
Daniele G, Fieu M, Joachim S et al (2016) Development of a multi-residue analysis of diclofenac and some transformation products in bivalves using QuEChERS extraction and liquid chromatography-tandem mass spectrometry. Application to samples from mesocosm studies. Talanta 155:1–7. doi:10.1016/j.talanta.2016.04.016
Efosa NJ, Kleiner W, Kloas W, Hoffmann F (2017) Diclofenac can exhibit estrogenic modes of action in male Xenopus laevis, and affects the hypothalamus-pituitary-gonad axis and mating vocalizations. Chemosphere 173:69–77. doi:10.1016/j.chemosphere.2017.01.030
Ericson H, Thorsen G, Kumblad L (2010) Physiological effects of diclofenac, ibuprofen and propranolol on Baltic Sea blue mussels. Aquat Toxicol 99:223–231. doi:10.1016/j.aquatox.2010.04.017
Freas W, Grollman S (1980) Ionic and osmotic influence on prostaglandin release from the gill tissue of a marine bivalve, Modiolus demissus. J Exp Biol 84:169–185
Gagne F, Berube E, Fournier M, Blaise C (2005) Inflammatory properties of municipal effluents to Elliptio complanata mussels lack of effects from anti-inflammatory drugs. Comp Biochem Physiol C: Toxicol Pharmacol 141:332–337. doi:10.1016/j.cca.2005.06.006
Gaw S, Thomas KV, Hutchinson TH (2014) Sources, impacts and trends of pharmaceuticals in the marine and coastal environment. Philos Trans R Soc Lond Ser B Biol Sci. doi:10.1098/rstb.2013.0572
Gibbons MC, Castagna M (1984) Serotonin as an inducer of spawning in six bivalve species. Aquaculture 40:189–191. doi:10.1016/0044-8486(84)90356-9
Gonzalez-Rey M, Bebianno MJ (2012) Does non-steroidal anti-inflammatory (NSAID) ibuprofen induce antioxidant stress and endocrine disruption in mussel Mytilus galloprovincialis? Environ Toxicol Pharmacol 33:361–371. doi:10.1016/j.etap.2011.12.017
Gonzalez-Rey M, Bebianno MJ (2014) Effects of non-steroidal anti-inflammatory drug (NSAID) diclofenac exposure in mussel Mytilus galloprovincialis. Aquat Toxicol 148:221–230. doi:10.1016/j.aquatox.2014.01.011
Groner F, Hohne C, Kleiner W, Kloas W (2017) Chronic diclofenac exposure affects gill integrity and pituitary gene expression and displays estrogenic activity in nile tilapia (Oreochromis niloticus). Chemosphere 166:473–481. doi:10.1016/j.chemosphere.2016.09.116
Kallio J-M, Lahti M, Oikari A, Kronberg L (2010) Metabolites of the aquatic pollutant diclofenac in fish bile. Environ Sci Technol 44:7213–7219. doi:10.1021/es903402c
Maruya KA, Vidal-Dorsch DE, Bay SM et al (2012) Organic contaminants of emerging concern in sediments and flatfish collected near outfalls discharging treated wastewater effluent to the Southern California bight. Environ Toxicol Chem 31:2683–2688. doi:10.1002/etc.2003
Matsutani T, Nomura T (1986) Pharmacological observations on the mechanism of spawning in the scallop Patinopecten yessoensis. Nippon Suisan Gakkaishi 52:1589–1594. doi:10.2331/suisan.52.1589
Matsutani T, Nomura T (1987) In vitro effects of serotonin and prostaglandins on release of eggs from the ovary of the scallop, Patinopecten Yessoensis. Gen Comp Endocrinol 67:111–118
McEneff G, Barron L, Kelleher B et al (2014) A year-long study of the spatial occurrence and relative distribution of pharmaceutical residues in sewage effluent, receiving marine waters and marine bivalves. Sci Total Environ 476–477:317–326. doi:10.1016/j.scitotenv.2013.12.123
Mehinto AC, Hill EM, Tyler CR (2010) Uptake and biological effects of environmentally relevant concentrations of the nonsteroidal anti-inflammatory pharmaceutical diclofenac in rainbow trout (Oncorhynchus mykiss). Environ Sci Technol 44:2176–2182. doi:10.1021/es903702m
Morthorst JE, Lister A, Bjerregaard P, Van der Kraak G (2013) Ibuprofen reduces zebrafish PGE(2) levels but steroid hormone levels and reproductive parameters are not affected. Comp Biochem Physiol C: Toxicol Pharmacol 157:251–257. doi:10.1016/j.cbpc.2012.12.001
Nodler K, Voutsa D, Licha T (2014) Polar organic micropollutants in the coastal environment of different marine systems. Mar Pollut Bull 85:50–59. doi:10.1016/j.marpolbul.2014.06.024
Osada M, Nishikawa M, Nomura T (1989) Involvement of prostaglandins in the spawning of the scallop, Patinopecten Yessoensis. Comp Biochem Physiol C: Comp Pharmacol 94:595–601. doi:10.1016/0742-8413(89)90119-9
Rowley AF, Vogan CL, Taylor GW, Clare AS (2005) Prostaglandins in non-insectan invertebrates: recent insights and unsolved problems. J Exp Biol 208:3–14. doi:10.1242/jeb.01275
Saintsing DG, Dietz TH (1983) Modification of sodium transport in freshwater mussels by prostaglandins, cyclic AMP and 5-hydroxytryptamine: effects of inhibitors of prostaglandin synthesis. Comp Biochem Physiol C 76:285–290
Schmidt W, O’Rourke K, Hernan R, Quinn B (2011) Effects of the pharmaceuticals gemfibrozil and diclofenac on the marine mussel (Mytilus spp.) and their comparison with standardized toxicity tests. Mar Pollut Bull 62:1389–1395. doi:10.1016/j.marpolbul.2011.04.043
Viant MR (2007) Metabolomics of aquatic organisms: the new ‘omics’ on the block. Mar Ecol Prog Ser 332:301–306. doi:10.3354/meps332301
Acknowledgments
Funding support was obtained from the The French National Research Program for Environmental and Occupational Health of Anses (AMeCE 2015/1/091) and the Agence Nationale de la Recherche (IMAP ANR-16-CE34-0006-01). This research benefited from the support of the Chair Veolia Environnement-HydroSciences: Risk analysis relating to emerging contaminants in water bodies. The doctoral fellowship of Bénilde Bonnefille is financially supported by a grant from the Université de Montpellier and Sanofi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Courant, F., Arpin-Pont, L., Bonnefille, B. et al. Exposure of marine mussels to diclofenac: modulation of prostaglandin biosynthesis. Environ Sci Pollut Res 25, 6087–6094 (2018). https://doi.org/10.1007/s11356-017-9228-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9228-6