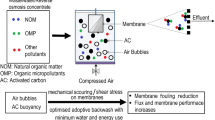
Abstract
Nitrate contamination of ground and surface waters causes environmental pollution and human health problems in many parts of the world. This study tests the nitrate removal efficiencies of two ion exchange resins (Dowex 21K XLT and iron-modified Dowex 21K XLT (Dowex-Fe)) and two chemically modified bio-adsorbents (amine-grafted corn cob (AG corn cob) and amine-grafted coconut copra (AG coconut copra)) using a dynamic adsorption treatment system. A submerged membrane (microfiltration) adsorption hybrid system (SMAHS) was used for the continuous removal of nitrate with a minimal amount of adsorbents. The efficiency of membrane filtration flux and replacement rate of adsorbent were studied to determine suitable operating conditions to maintain the effluent nitrate concentration below the WHO drinking standard limit of 11.3 mg N/L. The volume of water treated and the amount of nitrate adsorbed per gramme of adsorbent for all four flux tested were in the order Dowex-Fe > Dowex > AG coconut copra > AG corn cob. The volumes of water treated (L/g adsorbent) were 0.91 and 1.85, and the amount of nitrate removed (mg N/g adsorbent) were 9.8 and 22.2 for AG corn cob and Dowex-Fe, respectively, at a flux of 15 L/(m2/h).





Similar content being viewed by others
References
Bhatnagar A, Sillanpää M (2011) A review of emerging adsorbents for nitrate removal from water. Chem Eng J 168:493–504
Bryjak M, Wolska J, Kabay N (2008) Removal of boron from seawater by adsorption–membrane hybrid process: implementation and challenges. Desalination 223:57–62
Camargo JA, Alonso Á (2006) Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: a global assessment. Environ Int 32:831–849
Fewtrell L (2004) Drinking-water nitrate, methemoglobinemia, and global burden of disease: a discussion. Environ Health Perspect 112:1371–1374
Guo W, Shim W, Vigneswaran S, Ngo H (2005) Effect of operating parameters in a submerged membrane adsorption hybrid system: experiments and mathematical modeling. J Membr Sci 247:65–74
Johir M, Aryal R, Vigneswaran S, Kandasamy J, Grasmick A (2011) Influence of supporting media in suspension on membrane fouling reduction in submerged membrane bioreactor (SMBR). J Membr Sci 374:121–128
Johir M, Shanmuganathan S, Vigneswaran S, Kandasamy J (2013) Performance of submerged membrane bioreactor (SMBR) with and without the addition of the different particle sizes of GAC as suspended medium. Bioresour Technol 141:13–18
Johir M, Nguyen TT, Mahatheva K, Pradhan M, Ngo HH, Guo W, Vigneswaran S (2016) Removal of phosphorus by a high rate membrane adsorption hybrid system. Bioresour Technol 201:365–369
Kalaruban M, Loganathan P, Shim W, Kandasamy J, Naidu G, Nguyen TV, Vigneswaran S (2016a) Removing nitrate from water using iron-modified Dowex 21K XLT ion exchange resin: batch and fluidised-bed adsorption studies. Sep Purif Technol 158:62–70
Kalaruban M, Loganathan P, Shim W, Kandasamy J, Ngo H, Vigneswaran S (2016b) Enhanced removal of nitrate from water using amine-grafted agricultural wastes. Sci Total Environ 565:503–510
Kapoor A, Viraraghavan T (1997) Nitrate removal from drinking water—review. J Environ Eng 123:371–380
Lee JW, Choi SP, Thiruvenkatachari R, Shim WG, Moon H (2006) Submerged microfiltration membrane coupled with alum coagulation/powdered activated carbon adsorption for complete decolorization of reactive dyes. Water Res 40:435–444
Loganathan P, Vigneswaran S, Kandasamy J (2013) Enhanced removal of nitrate from water using surface modification of adsorbents—a review. J Environ Manag 131:363–374
Nguyen TAH, Ngo HH, Guo WS, Pham TQ, Li FM, Nguyen TV, Bui XT (2015) Adsorption of phosphate from aqueous solutions and sewage using zirconium loaded okra (ZLO): fixed-bed column study. Sci Total Environ 523:40–49
Nur T, Shim WG, Loganathan P, Vigneswaran S, Kandasamy J (2015) Nitrate removal using Purolite A520E ion exchange resin: batch and fixed-bed column adsorption modelling. Inter J Environ Sci Technol 12:1311–1320
Samatya S, Kabay N, Yüksel Ü, Arda M, Yüksel M (2006) Removal of nitrate from aqueous solution by nitrate selective ion exchange resins. React Funct Polym 66:1206–1214
Shanmuganathan S, Nguyen TV, Jeong S, Kandasamy J, Vigneswaran S (2015a) Submerged membrane–(GAC) adsorption hybrid system in reverse osmosis concentrate treatment. Sep Purif Technol 146:8–14
Shanmuganathan S, Johir MA, Nguyen TV, Kandasamy J, Vigneswaran S (2015b) Experimental evaluation of microfiltration–granular activated carbon (MF–GAC)/nano filter hybrid system in high quality water reuse. J Membr Sci 476:1–9
Smith P, Vigneswaran S (2009) Effect of backwash and powder activated carbon (PAC) addition on performance of side stream membrane filtration system (SSMFS) on treatment of biological treatment effluent. Desalin Water Treat 11:46–51
Vigneswaran S, Chaudhary D, Ngo H, Shim W, Moon H (2003) Application of a PAC-membrane hybrid system for removal of organics from secondary sewage effluent: experiments and modelling. Sep Sci Technol 38:2183–2199
WHO (2011) Guidelines for drinking-water quality, Fourth edn. World Health Organization, Geneva
Acknowledgements
This study was financially supported by the Cooperative Research Centre for Contamination Assessment and Remediation of the Environment (CRC CARE; project number 4.1.17-13/14).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Highlights
> SMAHS treatment of polluted water continuously maintained NO3 − below WHO limit.
> NO3 − adsorption capacity: Dowex-Fe > Dowex > coconut copra/amine > corn cob/amine.
> Treated water volume was in the same order as the adsorption capacity.
> Transmembrane pressure increased with flux and was highest for bio-adsorbents.
Rights and permissions
About this article
Cite this article
Kalaruban, M., Loganathan, P., Kandasamy, J. et al. Submerged membrane adsorption hybrid system using four adsorbents to remove nitrate from water. Environ Sci Pollut Res 25, 20328–20335 (2018). https://doi.org/10.1007/s11356-017-8905-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-8905-9