Abstract
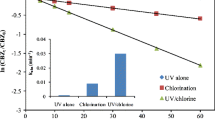
UV/chlorine, as a novel disinfection method, has attracted great interest due to its effective removal for pathogenic microorganism and degradation of trace organic contaminants existed in water environment. This paper investigated the degradation kinetics and pathways of Bezafibrate (BZF), a typical antilipemic drug, during UV/chlorine process. The results showed that 92.3% of BZF was degraded after 20 min in UV/chlorine process. This indicated HO• and reactive chlorine species (RCSs) formed in UV/chlorine played the dominant role in degrading BZF. Observed rate constants of BZF degradation (k obs,BZF) in UV/chlorine process increased linearly in a wide chlorine dosage from 0.1 to 1.0 mM, which implied that ClO• generated from the reactions of chlorine with HO• and Cl• could react with BZF rapidly. The steady-state kinetic modeling result proved this deduction and the rate constant of ClO• with BZF was fitted to be 5.0 × 108 M−1 s−1. k obs,BZF was affected by Cl− and HA. The total contribution of RCSs (including Cl•, Cl2•−, and ClO•) to the degradation of BZF was determined to be ~ 80%, which is much higher than that of HO•. Thirteen degradation products of BZF were identified by LC-MS/MS. Initial degradation products were arisen from hydroxylation, chlorine substitution and cyclization by HO• and RCSs, and then further oxidized to generate acylamino cleavage and demethylation products.









Similar content being viewed by others
References
Beretta M, Britto V, Tavares TM, da Silva SMT, Pletsch AL (2014) Occurrence of pharmaceutical and personal care products (PPCPs) in marine sediments in the Todos os Santos Bay and the north coast of Salvador, Bahia, Brazil. J Soils Sediments 14:1278–1286
Buxton GV, Greenstock CL, Helman WP, Ross AB (1988) Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (·OH/·O−) in aqueous solution. J Phys Chem Ref Data 17:513–886
Cai M-Q, Feng L, Jiang J, Qi F, Zhang L-Q (2013) Reaction kinetics and transformation of antipyrine chlorination with free chlorine. Water Res 47:2830–2842
Carranzo IV (2012) APHA, AWWA, WEF. “standard methods for examination of water and wastewater.” Anales de Hidrología Médica 5
Cermola M, DellaGreca M, Iesce M, Previtera L, Rubino M, Temussi F, Brigante M (2005) Phototransformation of fibrate drugs in aqueous media. Environ Chem Lett 3:43–47
Dantas RF, Canterino M, Marotta R, Sans C, Esplugas S, Andreozzi R (2007) Bezafibrate removal by means of ozonation: primary intermediates, kinetics, and toxicity assessment. Water Res 41:2525–2532
Fang J, Fu Y, Shang C (2014) The roles of reactive species in micropollutant degradation in the UV/free chlorine system. Environ Sci-Wat Res 48:1859–1868
Gibs J, Stackelberg PE, Furlong ET, Meyer M, Zaugg SD, Lippincott RL (2007) Persistence of pharmaceuticals and other organic compounds in chlorinated drinking water as a function of time. Sci Total Environ 373:240–249
Glassmeyer S, Shoemaker J (2005) Effects of chlorination on the persistence of pharmaceuticals in the environment. Bull Environ Contam Toxicol 74:24–31
Gonçalves A, Órfão JJ, Pereira MFR (2013) Ozonation of bezafibrate promoted by carbon materials. Appl Catal B Environ 140:82–91
Gonçalves AG, JJ Ó, Pereira MF (2012) Catalytic ozonation of sulphamethoxazole in the presence of carbon materials: catalytic performance and reaction pathways. J Hazard Mater 239-240:167–174
Guan YH, Ma J, Li XC, Fang JY, Chen LW (2011) Influence of pH on the formation of sulfate and hydroxyl radicals in the UV/peroxymonosulfate system. Environ Sci-Wat Res 45:9308
Guo Z-B, Lin Y-L, Xu B, Huang H, Zhang T-Y, Tian F-X, Gao N-Y (2016) Degradation of chlortoluron during UV irradiation and UV/chlorine processes and formation of disinfection by-products in sequential chlorination. Chem Eng J 283:412–419
Hessler DP, Gorenflo V, Frimmel FH (2010) Degradation of aqueous atrazine and metazachlor solutions by UV and UV/H2O2—influence of pH and herbicide concentration Abbau von Atrazin und metazachlor in wäßriger Lösung durch UV und UV/H2O2—Einfluß von pH und Herbizid-Konzentration. CLEAN Soil Air Water 21:209–214
Huerta-Fontela M, Galceran MT, Ventura F (2011) Occurrence and removal of pharmaceuticals and hormones through drinking water treatment. Water Res 45:1432–1442
Kim I, Tanaka H (2009) Photodegradation characteristics of PPCPs in water with UV treatment. Environ Int 35:793–802
Kim I, Yamashita N, Tanaka H (2009) Photodegradation of pharmaceuticals and personal care products during UV and UV/H2O2 treatments. Chemosphere 77:518–525
Kim SD, Cho J, Kim IS, Vanderford BJ, Snyder SA (2007) Occurrence and removal of pharmaceuticals and endocrine disruptors in South Korean surface, drinking, and waste waters. Water Res 41:1013–1021
Kong X, Jiang J, Ma J, Yang Y, Liu W, Liu Y (2016) Degradation of atrazine by UV/chlorine: efficiency, influencing factors, and products. Water Res 90:15–23
Kosma CI, Lambropoulou DA, Albanis TA (2010) Occurrence and removal of PPCPs in municipal and hospital wastewaters in Greece. J Hazard Mater 179:804–817
Lambropoulou D, Hernando M, Konstantinou I, Thurman E, Ferrer I, Albanis T, Fernández-Alba A (2008) Identification of photocatalytic degradation products of bezafibrate in TiO 2 aqueous suspensions by liquid and gas chromatography. J Chromatogr A 1183:38–48
Lee W, Westerhoff P, Yang X, Shang C (2007) Comparison of colorimetric and membrane introduction mass spectrometry techniques for chloramine analysis. Water Res 41:3097–3102
Lee Y, von Gunten U (2010) Oxidative transformation of micropollutants during municipal wastewater treatment: comparison of kinetic aspects of selective (chlorine, chlorine dioxide, ferrate VI, and ozone) and non-selective oxidants (hydroxyl radical). Water Res 44:555–566
Li H, Xu B, Qi F, Sun D, Chen Z (2014) Degradation of bezafibrate in wastewater by catalytic ozonation with cobalt doped red mud: efficiency, intermediates and toxicity. Appl Catal B Environ 152–153:342–351
Lyon BA, Milsk RY, Deangelo AB, Simmons JE, Moyer MP, Weinberg HS (2014) Integrated chemical and toxicological investigation of UV-chlorine/chloramine drinking water treatment. Environ Sci-Wat Res 48:6743–6753
Mártire DO, Rosso JA, Bertolotti S, Le Roux GC, Braun AM, Gonzalez MC (2001) Kinetic study of the reactions of chlorine atoms and Cl2 •-radical anions in aqueous solutions. II. Toluene, benzoic acid, and chlorobenzene. J Phys Chem A 105:5385–5392
Martino M, Rosal R, Sastre H, Díez FV (1999) Hydrodechlorination of dichloromethane, trichloroethane, trichloroethylene and tetrachloroethylene over a sulfided Ni/Mo–γ-alumina catalyst. Appl Catal B Environ 20:301–307
Mitch WA, Sedlak DL (2004) Characterization and fate of N-nitrosodimethylamine precursors in municipal wastewater treatment plants. Environ Sci-Wat Res 38:1445–1454
Mompelat S, Le Bot B, Thomas O (2009) Occurrence and fate of pharmaceutical products and by-products, from resource to drinking water. Environ Int 35:803–814. https://doi.org/10.1016/j.envint.2008.10.008
Rahn RO, Stefan MI, Bolton JR, Goren E, Shaw PS, Lykke KR (2003) Quantum yield of the iodide-iodate chemical actinometer: dependence on wavelength and concentrations. Photochem Photobiol 78:146–152
Razavi B, Song W, Cooper WJ, Greaves J, Jeong J (2009) Free-radical-induced oxidative and reductive degradation of fibrate pharmaceuticals: kinetic studies and degradation mechanisms. J Phys Chem A 113:1287–1294
Simazaki D, Fujiwara J, Manabe S, Matsuda M, Asami M, Kunikane S (2008) Removal of selected pharmaceuticals by chlorination, coagulation–sedimentation and powdered activated carbon treatment. Water Sci Technol 58:1129–1135
Szabó RK (2010) Decomposition of some pharmaceuticals by advanced oxidation processes University of Szeged Doctoral School of Environmental Sciences
Trovó AG, Melo SAS, Nogueira RFP (2008) Photodegradation of the pharmaceuticals amoxicillin, bezafibrate and paracetamol by the photo-Fenton process—application to sewage treatment plant effluent. J Photochem Photobiol A Chem 198:215–220
Wang D, Bolton JR, Hofmann R (2012) Medium pressure UV combined with chlorine advanced oxidation for trichloroethylene destruction in a model water. Water Res 46:4677–4686
Wang WL, Wu QY, Huang N, Wang T, Hu HY (2016) Synergistic effect between UV and chlorine (UV/chlorine) on the degradation of carbamazepine: influence factors and radical species. Water Res 98:190–198
Watkinson A, Murby E, Kolpin D, Costanzo S (2009) The occurrence of antibiotics in an urban watershed: from wastewater to drinking water. Sci Total Environ 407:2711–2723
Watts MJ, Linden KG (2007) Chlorine photolysis and subsequent OH radical production during UV treatment of chlorinated water. Water Res 41:2871–2878
Wols BA, Harmsen DJH, Beerendonk EF, Hofman-Caris CHM (2015) Predicting pharmaceutical degradation by UV (MP)/H2O2 processes: a kinetic model. Chem Eng J 263:336–345
Wu Y, Bianco A, Brigante M, Dong W, Sainteclaire PD, Hanna K, Mailhot G (2015) Sulfate radical photogeneration using Fe-EDDS: influence of critical parameters and naturally occurring scavengers. Environ Sci-Wat Res 49:14343
Wu Z, Fang J, Xiang Y, Shang C, Li X, Meng F, Yang X (2016) Roles of reactive chlorine species in trimethoprim degradation in the UV/chlorine process: kinetics and transformation pathways. Water Res 104:272–282
Xiang Y, Fang J, Shang C (2016) Kinetics and pathways of ibuprofen degradation by the UV/chlorine advanced oxidation process. Water Res 90:301
Xu B, Qi F, Sun D, Chen Z, Robert D (2016) Cerium doped red mud catalytic ozonation for bezafibrate degradation in wastewater: efficiency, intermediates, and toxicity. Chemosphere 146:22–31
Yang X et al (2016) PPCP degradation by UV/chlorine treatment and its impact on DBP formation potential in real waters. Water Res 98:309–318
Yuan H, Zhang Y, Zhou X (2012) Degradation of bezafibrate with UV/H2O2 in surface water and wastewater treatment plant effluent. CLEAN Soil Air Water 40:239–245
Z S, C L, M B, JR B, MG E (2014) Application of a solar UV/chlorine advanced oxidation process to oil sands process-affected water remediation. Environ Sci-Wat Res 48:9692–9701
Zhang X, Li W, Rd BE, Wang X, Ren P (2015) UV/chlorine process for ammonia removal and disinfection by-product reduction: comparison with chlorination. Water Res 68:804–811
Zhao X, Chen Z-L, Wang X-C, Shen J-M, Xu H (2014) PPCPs removal by aerobic granular sludge membrane bioreactor. Appl Microbiol Biotechnol 98:9843–9848
Funding
This work was supported by the National Natural Science Foundation of China (51578066 and 51608036), the Beijing Natural Science Foundation (No. 8152022), and the Fundamental Research Funds for the Central Universities (No. 2015ZCQ-HJ-02).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Bingcai Pan
Electronic supplementary material
ESM 1
(DOCX 285 kb)
Rights and permissions
About this article
Cite this article
Shi, XT., Liu, YZ., Tang, YQ. et al. Kinetics and pathways of Bezafibrate degradation in UV/chlorine process. Environ Sci Pollut Res 25, 672–682 (2018). https://doi.org/10.1007/s11356-017-0461-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0461-9