Abstract
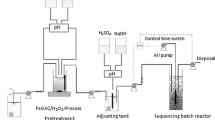
Fenton oxidation and ultrasound-based pretreatment have been applied to improve the treatment of real industrial wastewater based on the use of biological oxidation. The effect of operating parameters such as Fe2+ loading, contact time, initial pH, and hydrogen peroxide loading on the extent of chemical oxygen demand (COD) reduction and change in biochemical oxygen demand (BOD5)/COD ratio has been investigated. The optimum operating conditions established for the pretreatment were initial pH of 3.0, Fe2+ loading of 2.0, and 2.5 g L−1 for the US/Fenton/stirring and Fenton approach, respectively, and temperature of 25 °C with initial H2O2 loading of 1.5 g L−1. The use of pretreatment resulted in a significant increase in the BOD5/COD ratio confirming the production of easily digestible intermediates. The effect of the type of sludge in the aerobic biodegradation was also investigated based on the use of primary activated sludge (PAS), modified activated sludge (MAS), and activated sludge (AS). Enhanced removal of the pollutants as well as higher biomass yield was observed for MAS as compared to PAS and AS. The use of US/Fenton/stirring pretreatment under the optimized conditions followed by biological oxidation using MAS resulted in maximum COD removal at 97.9 %. The required hydraulic retention time for the combined oxidation system was also significantly lower as compared to only biological oxidation operation. Kinetic studies revealed that the reduction in the COD followed a first-order kinetic model for advanced oxidation and pseudo first-order model for biodegradation. The study clearly established the utility of the combined technology for the effective treatment of real industrial wastewater.















Similar content being viewed by others
References
Abdulkadir M, Waliyu S (2012) Screening and isolation of the soil bacteria for ability to produce antibiotics. Eur J Appl Sci 4:211–215
Aiba S, Humphrey AE, Millis NF (1973) Biochemical engineering. Academic, New York
APHA, AWWA (1998) Standard methods for the examination of water and wastewater. WPCF, 20th ed., Washington, 1998.
Bautista P, Mohedano AF, Gilarranz MA, Casas JA, Rodriguez JJ (2007) Application of Fenton oxidation to cosmetic wastewater treatment. J Haz Mat 143:128–134
Beltran JH, Torregrosa J, Dominguez JR, Gracia J (2000) Treatment of black-olive wastewater by ozonation and aerobic biological degradation. Water Res 34:3515–3522
Benitez FJ, Acero JL, Real FJ, Rubio FJ, Leal AI (2001) The role of hydroxyl radicals for the decomposition of p-hydroxy phenylacetic acid in aqueous solutions. Water Res 35:1338–1343
Chan YJ, Chong MF, Law CL, Hassell DG (2009) A review on anaerobic–aerobic treatment of industrial and municipal wastewater. Chem Eng J 155:1–18
Contreras S, Rodriguez M, Al Momani F, Sans C, Esplugas S (2003) Contribution of the ozonation pre-treatment to the biodegradation of aqueous solutions of 2,4-dichlorophenol. Water Res 37:3164–3171
Cortez S, Teixeira P, Oliveira R, Mota M (2010) Ozonation as polishing treatment of mature landfill leachate. J Haz Mat 182:730–734
Cravotto G, Di Carlo S, Tumiatti V, Roggero C, Bremner HD (2005) Degradation of persistent organic pollutants by Fenton reagent facilitated by microwave of high-intensity ultrasound. Environ Tech 26:721–724
Elbeshbishy E, Nakhla G (2011) Comparative study of the effect of ultrasonication on the anaerobic biodegradability of food waste in single and two-stage systems. Biores Tech 102:6449–6457
Eskelinen K, Sarkka H, Kurniawan TA, Sillanpaa MET (2010) Removal of recalcitrant contaminants from bleaching effluents in pulp and paper mills using ultrasonic irradiation and Fenton-like oxidation, electrochemical treatment, and/or chemical precipitation: a comparative study. Desalination 255:179–187
Garcia-Montano J, Torrades F, Garcia-Hortal JA, Domenech X, Peral J (2006) Degradation of Procion Red H-E7B reactive dye by coupling a photo-Fenton system with a sequencing batch reactor. J Haz Mat 134:220–229
Ghaly MY, Hartel G, Haseneder R (2009) Integrated coupled systems as biodegradability enhancement of textile wastewater by photo-Fenton process. Afinidad 66:420–428
Gogate PR (2008) Treatment of wastewater streams containing phenolic compounds using hybrid techniques based on cavitation: a review of current status and the way forward. Ultrason Sonochem 15:1–15
Gogate PR, Pandit AB (2004) A review of imperative technologies for wastewater treatment I: oxidation technologies at ambient conditions. Adv Env Res 8:501–551
Grau P, Dohanyas M, Chudoba J (1975) Kinetics of multicomponent substrate removal by activated sludge. Water Res 9:337–342
Jamil TS, Ghaly MY, El-Seesy IE, Souaya ER, Nasr RA (2011) A comparative study among different photochemical oxidation processes to enhance the biodegradability of paper mill wastewater. J Haz Mat 185:353–358
Karthikeyan S, Titus A, Gnanamani A, Mandal AB, Sekaran G (2011) Treatment of textile wastewater by homogeneous and heterogeneous Fenton oxidation processes. Desalination 281:438–445
Lafi WK, Shannak B, Shannag MA, Anber ZA, Hasan MA (2009) Treatment of olive mill wastewater by combined advanced oxidation and biodegradation. Sep Pur Tech 70:141–146
Lodha B, Chaudhari S (2007) Optimization of Fenton-biological treatment scheme for the treatment of aqueous dye solutions. J Haz Mat 148:459–466
Mandal T, Maity S, Dasgupta D, Datta S (2010) Advanced oxidation process and biotreatment: their roles in combined industrial wastewater treatment. Desalination 250:87–94
Metcalf I, Eddy (1979) Wastewater engineering: treatment, disposal, reuse. 2nd ed. Tata McGraw Hill, New Delhi.
Oller I, Malato S, Sanchez-Perez JA (2011) Combination of advanced oxidation processes and biological treatments for wastewater decontamination—a review. Sci Total Environ 409:4141–4166
Ramteke LP, Gogate PR (2015a) Synthesis and characterization of modified activated sludge for biological oxidation with isolation studies and performance evaluation. Sep Pur Tech 141:179–187
Ramteke LP, Gogate PR (2015b) Treatment of toluene, benzene, naphthalene and xylene (BTNXs) containing wastewater using improved biological oxidation with pretreatment using Fenton/ultrasound based processes. J Ind Eng Chem 28:247–260
Ramteke LP, Gogate PR (2015c) Removal of ethylbenzene and p-nitrophenol using combined approach of advanced oxidation with biological oxidation based on the use of novel modified prepared activated sludge. Process Saf Environ Protection 95:146–158
Rathi AKA (2002) Chemical industry wastewater treatment using adsorption. J Sci Ind Res 61:53–60
Rehorek A, Tauber M, Guebitz G (2004) Application of power ultrasound for azo dye degradation. Ultrason Sonochem 11:177–182
Rivas FJ, Beltran FJ, Gimeno O, Alvarez P (2003) Optimization of Fenton’s reagent usage as a pre-treatment for fermentation brines. J Haz Mat 96:277–290
San Sebastian N, Figuls J, Font X, Sanchez A (2003) Pre-oxidation of an extremely polluted industrial wastewater by the Fenton’s reagent. J Haz Mat 101:315–322
Silverstein RMC, Basdler GC, Morrill GC (1991) Spectrophotometric identification of organic compounds. Wiley, New York, p 1991
Stasinakis AS (2008) Use of selected advanced oxidation process (AOPs) for wastewater treatment—a mini review. Global NEST J 10:376–385
Symons JM (1960) A procedure for determination of the biological treatability of industrial wastes. J Water Pollut Control Fed 32:841–51
Walling C (1998) Intermediates in the reactions of Fenton type reagents. Accounts Chem Res 31:155–157
Zazo JA, Casas JA, Mohedano AF, Gilarranz MA, Rodriguez JJ (2005) Chemical pathway and kinetics of phenol oxidation by Fenton’s reagent. Env Sci Tech 39:9295–9302
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Gerald Thouand
Rights and permissions
About this article
Cite this article
Ramteke, L.P., Gogate, P.R. Treatment of real industrial wastewater using the combined approach of advanced oxidation followed by aerobic oxidation. Environ Sci Pollut Res 23, 9712–9729 (2016). https://doi.org/10.1007/s11356-016-6156-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-016-6156-9