Abstract
Background
This study investigated the effects of endurance exercise training and zoledronic acid (Zol) treatment on ovariectomy-induced osteopenia.
Methods
Middle-aged female Wistar rats were subjected to sham and ovariectomy surgeries and assigned to seven different groups according to Zol treatment doses and treadmill endurance exercise (Ex) training; these were the Sham, Ovx, Ovx + LowZ, Ovx + HighZ, Ovx + Ex, Ovx + LowZ + Ex, Ovx + HighZ + Ex groups. The subcutaneous doses for the low- and high-Zol-treated ovariectomized rats were 20 and 100 µg/kg, respectively. Total intervention period was 10 weeks.
Results
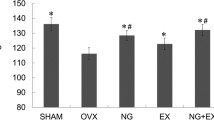
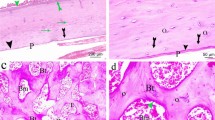
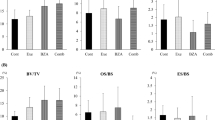
When comparing the Ovx groups to the Sham group, ovariectomy resulted in multiple compromises in bone histomorphometry, size-related measurements, and biomechanical properties. Two-way ANOVA was adopted to investigate the effects of Zol treatments and Ex on the six ovariectomized groups. In spongy bone histomorphometry, Zol treatment resulted in comprehensive preservation for all parameters, while exercise demonstrated benefits for osteoclastic number and mineralization activity. In serum marker assays, the Zol-treated rats had lower serum osteocalcin and IGF-1/IGFBP-3 molar ratio when compared to the non-treated rats. Exercise training significantly benefited metabolism through lower serum triglycerides, cholesterol, leptin, IGF-1 and IGF-1/IGFBP-3 molar ratio, along with increased IGFBP-3 and phosphorus (p < 0.05). In bone-size-related measurements and biomechanical property analyses, data were normalized to body weight. For whole bone strength, Zol treatments demonstrated higher bone fracture load (FL) and FL energy, while Ex showed benefits to yield load, FL, post-yield load energy, and stiffness. In tissue-level biomechanical properties, rats treated with the higher Zol dose were higher in yield stress and yield toughness. Exercise benefited most bone-size-related measurements, while the high Zol-treated rats showed smaller medullar area.
Conclusion
When combined with Zol treatments, exercise training did not demonstrate significant collaborative or modifying effects. Exercise and bisphosphonate tend to affect to bone through distinct pathways. A lower dose or different timing of Zol treatment or during a later phase of bisphosphonate treatment (drug holiday) might allow the occurrence of synergistic effects between Zol treatment and exercise.




Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- BMD:
-
Bone mineral density
- BMC:
-
Bone mineral content
- Zol:
-
Zoledronic acid
- IGF-1:
-
Insulin-like growth factor-1
- LI-IGF-1− / − :
-
Liver-specific IGF-1 inactivation
- LowZ and HighZ:
-
Zol treatment dose at 20 and 100 µg/kg, respectively
- EXE:
-
Endurance running training
- MMA:
-
Methyl methacrylate
- EDTA:
-
Ethylenediaminetetraacetic acid
- BV/TV:
-
Bone volume ratio (%), Tb.Th.: trabecular thickness (μm)
- Tb.Sp.:
-
Space between trabeculae (μm)
- Tb.No.:
-
Trabeculae number (1/mm)
- MS/BS:
-
Bone mineralization over bone surface (%)
- TRAP:
-
Tartrate-resistant acid phosphatase
- IGFBP-3:
-
Insulin-like growth factor binding protein-3
- YL:
-
Yield load
- FL:
-
Fracture load
- FFDW:
-
Fat-free dry weight
- Ct.Th.:
-
Cortical thickness (mm)
- CS.Ar.:
-
Total cross-sectional area (mm2)
- Ct.Ar.:
-
Cortical area (mm2)
- Me.Ar.:
-
Medullar area (mm2)
- CSMI:
-
Cross-sectional moment of inertia (mm4)
- YS:
-
Yield stress
- YT:
-
Yield toughness
- EM:
-
Elastic modulus
References
Nancollas GH, Tang R, Phipps RJ, Henneman Z, Gulde S, Wu W, Mangood A, Russell RG, Ebetino FH (2006) Novel insights into actions of bisphosphonates on bone: differences in interactions with hydroxyapatite. Bone 38:617–627
Lewiecki EM (2008) Intravenous zoledronic acid for the treatment of osteoporosis. Current Osteoporos Rep 6:17–23
Grey A, Bolland M, Wattie D, Horne A, Gamble G, Reid IR (2010) Prolonged antiresorptive activity of zoledronate: a randomized, controlled trial. J Bone Miner Res 25:2251–2255
Reid IR, Horne AM, Mihov B, Stewart A, Garratt E, Wong S, Wiessing KR, Bolland MJ, Bastin S, Gamble GD (2018) Fracture prevention with zoledronate in older women with osteopenia. N Engl J Med 379:2407–2416
Gasser JA, Ingold P, Venturiere A, Shen V, Green JR (2008) Long-term protective effects of zoledronic acid on cancellous and cortical bone in the ovariectomized rat. J Bone Miner Res 23:544–551
Khajuria DK, Razdan R, Mahapatra DR, Bhat MR (2013) Osteoprotective effect of propranolol in ovariectomized rats: a comparison with zoledronic acid and alfacalcidol. J Orthop Sci 18:832–842
Khajuria DK, Razdan R, Mahapatra DR (2014) The combination therapy with zoledronic Acid and propranolol improves the trabecular microarchitecture and mechanical property in an rat model of postmenopausal osteoporosis. J Osteoporos 2014:586431
Khajuria DK, Razdan R, Mahapatra DR (2015) Additive effects of zoledronic acid and propranolol on bone density and biochemical markers of bone turnover in osteopenic ovariectomized rats. Rev Bras Reumatol 55:103–112
Orwoll ES, Miller PD, Adachi JD, Brown J, Adler RA, Kendler D, Bucci-Rechtweg C, Readie A, Mesenbrink P, Weinstein RS (2010) Efficacy and safety of a once-yearly i.v. infusion of zoledronic acid 5 mg versus a once-weekly 70-mg oral alendronate in the treatment of male osteoporosis: a randomized, multicenter, double-blind, active-controlled study. J Bone Miner Res 25:2239–2250
Allen MR, Burr DB (2011) Bisphosphonate effects on bone turnover, microdamage, and mechanical properties: what we think we know and what we know that we don’t know. Bone 49:56–65
Lee JK (2009) Bilateral atypical femoral diaphyseal fractures in a patient treated with alendronate sodium. Int J Rheum Dis 12:149–154
Shane E, Burr D, Ebeling PR et al (2010) Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 25:2267–2294
Rogers LF, Taljanovic M (2010) FDA statement on relationship between bisphosphonate use and atypical subtrochanteric and femoral shaft fractures: a considered opinion. AJR Am J Roentgenol 195:563–566
Shane E, Burr D, Abrahamsen B et al (2014) Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 29:1–23
Lespessailles E, Jaffre C, Beaupied H, Nanyan P, Dolleans E, Benhamou CL, Courteix D (2009) Does exercise modify the effects of zoledronic acid on bone mass, microarchitecture, biomechanics, and turnover in ovariectomized rats? Calcif Tissue Int 85:146–157
Tamaki H, Akamine T, Goshi N, Kurata H, Sakou T (1998) Effects of exercise training and etidronate treatment on bone mineral density and trabecular bone in ovariectomized rats. Bone 23:147–153
Uusi-Rasi K, Kannus P, Cheng S et al (2003) Effect of alendronate and exercise on bone and physical performance of postmenopausal women: a randomized controlled trial. Bone 33:132–143
Uusi-Rasi K, Sievanen H, Heinonen A, Kannus P, Vuori I (2004) Effect of discontinuation of alendronate treatment and exercise on bone mass and physical fitness: 15-month follow-up of a randomized, controlled trial. Bone 35:799–805
Chilibeck PD, Davison KS, Whiting SJ, Suzuki Y, Janzen CL, Peloso P (2002) The effect of strength training combined with bisphosphonate (etidronate) therapy on bone mineral, lean tissue, and fat mass in postmenopausal women. Can J Physiol Pharmacol 80:941–950
Fuchs RK, Shea M, Durski SL, Winters-Stone KM, Widrick J, Snow CM (2007) Individual and combined effects of exercise and alendronate on bone mass and strength in ovariectomized rats. Bone 41:290–296
Waltman NL, Twiss JJ, Ott CD, Gross GJ, Lindsey AM, Moore TE, Berg K, Kupzyk K (2010) The effect of weight training on bone mineral density and bone turnover in postmenopausal breast cancer survivors with bone loss: a 24-month randomized controlled trial. Osteoporos Int 21:1361–1369
Paliologo T, Shimano RC, Shimano AC, Macedo AP, Falcai MJ, Issa JPM (2015) Effects of swimming associated with risedronate in osteopenic bones: an experimental study with ovariectomized rats. Micron 78:40–44
Shimano RC, Macedo AP, Falcai MJ, Ervolino E, Shimano AC, Issa JP (2014) Biomechanical and microstructural benefits of physical exercise associated with risedronate in bones of ovariectomized rats. Microsc Res Tech 77:431–438
Weitzmann MN, Pacifici R (2006) Estrogen deficiency and bone loss: an inflammatory tale. J Clin Investig 116:1186–1194
Weitzmann MN (2017) Bone and the immune system. Toxicol Pathol 45:911–924
Huang TH (2015) The skeleton modeled by endurance exercise and dietary manipulation: a perspective from the Interaction between bone metabolism and energy metabolism. Phys Educ J 48:1–17
Lindberg MK, Svensson J, Venken K, Chavoshi T, Andersson N, Moverare Skrtic S, Isaksson O, Vanderschueren D, Carlsten H, Ohlsson C (2006) Liver-derived IGF-I is permissive for ovariectomy-induced trabecular bone loss. Bone 38:85–92
Pichini S, Ventura R, Palmi I et al (2010) Effect of physical fitness and endurance exercise on indirect biomarkers of growth hormone and insulin misuse: Immunoassay-based measurement in urine samples. J Pharm Biomed Anal 53:1003–1010
Wieczorek-Baranowska A, Nowak A, Michalak E, Karolkiewicz J, Pospieszna B, Rutkowski R, Laurentowska M, Pilaczynska-Szczesniak L (2011) Effect of aerobic exercise on insulin, insulin-like growth factor-1 and insulin-like growth factor binding protein-3 in overweight and obese postmenopausal women. J Sports Med Phys Fit 51:525–532
Nishida Y, Matsubara T, Tobina T, Shindo M, Tokuyama K, Tanaka K, Tanaka H (2010) Effect of low-intensity aerobic exercise on insulin-like growth factor-I and insulin-like growth factor-binding proteins in healthy men. Int J Endocrinol 2010:452820
Grandys M, Majerczak J, Kuczek P, Sztefko K, Duda K, Zoladz JA (2017) Endurance training-induced changes in the GH-IGF-I axis influence maximal muscle strength in previously untrained men. Growth Horm IGF Res 32:41–48
Sengupta P (2013) The laboratory rat: relating its age with human’s. Int J Prev Med 4:624–630
Khajuria DK, Razdan R, Mahapatra DR (2012) Description of a new method of ovariectomy in female rats. Rev Bras Reumatol 52:462–470
Park SB, Lee YJ, Chung CK (2010) Bone mineral density changes after ovariectomy in rats as an osteopenic model : stepwise description of double dorso-lateral approach. J Korean Neurosurg Soc 48:309–312
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2:595–610
Janssen JA, Lamberts SW (2004) Igf-I and longevity. Horm Res 62(Suppl 3):104–109
Huang TH, Lin SC, Chang FL, Hsieh SS, Liu SH, Yang RS (2003) Effects of different exercise modes on mineralization, structure, and biomechanical properties of growing bone. J Appl Physiol 95:300–307
Honda A, Umemura Y, Nagasawa S (2001) Effect of high-impact and low-repetition training on bones in ovariectomized rats. J Bone Miner Res 16:1688–1693
Turner CH, Burr DB (1993) Basic biomechanical measurements of bone: a tutorial. Bone 14:595–608
Biewener AA (1991) Musculoskeletal design in relation to body size. J Biomech 24(Suppl 1):19–29
Wronski TJ, Cintron M, Dann LM (1988) Temporal relationship between bone loss and increased bone turnover in ovariectomized rats. Calcif Tissue Int 43:179–183
Riggs BL, Khosla S, Melton LJ 3rd (2002) Sex steroids and the construction and conservation of the adult skeleton. Endocr Rev 23:279–302
Rissanen JP, Suominen MI, Peng Z, Halleen JM (2008) Secreted tartrate-resistant acid phosphatase 5b is a Marker of osteoclast number in human osteoclast cultures and the rat ovariectomy model. Calcif Tissue Int 82:108–115
Kalu DN, Arjmandi BH, Liu CC, Salih MA, Birnbaum RS (1994) Effects of ovariectomy and estrogen on the serum levels of insulin-like growth factor-I and insulin-like growth factor binding protein-3. Bone Miner 25:135–148
Skedros JG, Dayton MR, Sybrowsky CL, Bloebaum RD, Bachus KN (2006) The influence of collagen fiber orientation and other histocompositional characteristics on the mechanical properties of equine cortical bone. J Exp Biol 209:3025–3042
Burr DB, Diab T, Koivunemi A, Koivunemi M, Allen MR (2009) Effects of 1 to 3 years’ treatment with alendronate on mechanical properties of the femoral shaft in a canine model: implications for subtrochanteric femoral fracture risk. J Orthop Res 27:1288–1292
Bajaj D, Geissler JR, Allen MR, Burr DB, Fritton JC (2014) The resistance of cortical bone tissue to failure under cyclic loading is reduced with alendronate. Bone 64:57–64
Fairey AS, Courneya KS, Field CJ, Bell GJ, Jones LW, Mackey JR (2003) Effects of exercise training on fasting insulin, insulin resistance, insulin-like growth factors, and insulin-like growth factor binding proteins in postmenopausal breast cancer survivors: a randomized controlled trial. Cancer Epidemiol Biomark Prev 12:721–727
Campbell PT, Gross MD, Potter JD, Schmitz KH, Duggan C, McTiernan A, Ulrich CM (2010) Effect of exercise on oxidative stress: a 12-month randomized, controlled trial. Med Sci Sports Exer 42:1448–1453
Pitocco D, Collina MC, Musella T, Ruotolo V, Caputo S, Manto A, Caradonna P, Galli M, Mancini L, Ghirlanda G (2008) Interaction between IGF-1, inflammation, and neuropathy in the pathogenesis of acute charcot neuroarthropathy: lessons from alendronate therapy and future perspectives of medical therapy. Horm Metab Res 40:163–164
Polidoulis I, Beyene J, Cheung AM (2012) The effect of exercise on pQCT parameters of bone structure and strength in postmenopausal women–a systematic review and meta-analysis of randomized controlled trials. Osteoporos Int 23:39–51
Kemmler W, von Stengel S, Bebenek M, Engelke K, Hentschke C, Kalender WA (2012) Exercise and fractures in postmenopausal women: 12-year results of the Erlangen Fitness and Osteoporosis Prevention Study (EFOPS). Osteoporos Int 23:1267–1276
Acknowledgements
Jae Cody is deeply appreciated for her editorial assistance.
Funding
This study was supported by a grant from the National Science Council (NSC 97–2410-H-006–083-MY2, TAIWAN).
Author information
Authors and Affiliations
Contributions
HS. L and TH. H prepared figures, tables and conducted the whole animal experiment; JL. L provided the expertise in calculations of material properties; MS. C provided expertise in serum analyses; AT. H conducted the tissue biomechanical test; HS. L, RS. Y, and TH. H wrote the draft of manuscript. JL. L contributed to review and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The procedures of animal experimentation throughout this study were approved by the Committee of Animal Study at National Cheng Kung University (Document Serial No. 97112).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, HS., Yang, RS., Lewis, J.L. et al. The effects of zoledronic acid treatment and endurance exercise on ovariectomy-induced osteopenia in Wistar rats. Sport Sci Health (2024). https://doi.org/10.1007/s11332-024-01181-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11332-024-01181-0